Choose The Correct Compound For The Given Ir Spectrum.
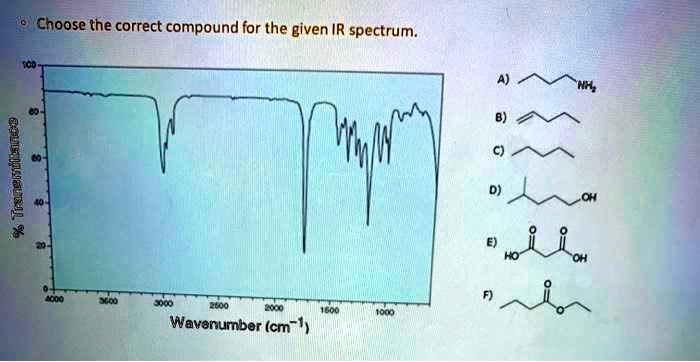
Imagine you're a detective, but instead of dusty fingerprints and shadowy alleyways, your clues are invisible waves of light! That's kind of what scientists do when they look at an Infrared (IR) Spectrum. Think of it as a special kind of fingerprint for molecules, a unique pattern that tells us exactly what kind of tiny building blocks make up a substance. Today, we’re going on a little adventure to see how we can use this cool tool to solve a molecular mystery. It’s like a fun puzzle, and the prize is knowing, with absolute certainty, what you’re holding!
Let's say you've stumbled upon a mysterious little vial. It could be anything! Maybe it’s a secret ingredient for a delicious cake, or perhaps a crucial component in making that special glue that holds your favorite toy together. The possibilities are endless, and frankly, a little bit exciting. Without any clues, it’s just a mystery liquid. But with our trusty IR Spectrometer, we can become molecular sleuths!
The IR Spectrometer works by shining infrared light through our mystery sample. Now, different types of bonds within a molecule, like the little arms that hold atoms together, vibrate at specific frequencies. It’s almost like each bond has its own tiny, secret song. When the infrared light hits these vibrating bonds, some of the light gets absorbed at those specific frequencies, kind of like how a tuning fork will vibrate if you play the right note. The spectrum is basically a graph that shows us which "notes" (frequencies) were absorbed and which ones sailed right on by.
So, how does this help us choose the correct compound? Well, imagine we have a few suspects for our mystery substance. Let’s call them Compound A, Compound B, and Compound C. Each of these compounds is made of different arrangements of atoms, with different kinds of bonds. For example, Compound A might be something simple and happy, like water (H2O). It’s got those lovely O-H bonds. Compound B could be something a bit more energetic, like ethanol (C2H5OH), which also has O-H bonds, but also those C-C and C-O bonds. And Compound C? Let's imagine it's something completely different, maybe like methane (CH4), which is all C-H bonds and no O-H!
Now, when we run our mystery sample through the IR Spectrometer, we get a spectrum – a wiggly line graph with peaks and valleys. This spectrum is like the compound's unique autobiography, written in the language of light absorption. If our mystery sample is actually water, its spectrum will have specific peaks that match the "songs" of the O-H bonds in water. These peaks are like its signature tune. If we compare our mystery spectrum to the known spectrum of water, and they match perfectly, bingo! We’ve found our culprit. It’s water!
But what if it’s not water? Let’s say we’re comparing our mystery spectrum to the known spectra of our suspects. If our mystery spectrum shows strong absorption in the region where the O-H bond usually sings its song, but also has other interesting "notes" that are characteristic of, say, the C-O bond, then we might be looking at Compound B, ethanol. It’s like finding the right combination of notes in a melody. Each bond contributes to the overall symphony of the molecule, and the IR spectrum captures this entire performance.
The really cool part is how specific this is. Even tiny differences in how atoms are arranged can create a completely different spectrum. It's like having a unique vocal fingerprint for every person on Earth – no two are exactly alike. So, if our mystery sample's spectrum doesn't match water, and it doesn't quite match ethanol, but it perfectly aligns with the spectrum of Compound C, methane, then we know we’ve got methane on our hands. It's this incredible precision that makes IR spectroscopy so powerful.
Think of it as a molecular ID card. You show up at the club (the lab), and you’ve got to prove who you are. Your IR spectrum is your ID. If it matches the ID of the person you claim to be (the known compound), then you’re in! If it doesn’t match any of the registered IDs, then you’re an imposter, or more likely, you’re a completely new mystery waiting to be identified.
The beauty of this technique lies in its ability to tell us not just that a molecule is present, but exactly what kind of molecule it is, all from its interaction with light. It’s a bit like magic, but it’s pure science!
So, the next time you hear about IR Spectroscopy, don’t think of complicated jargon. Think of it as a friendly detective, using the secret songs of molecular bonds to solve delicious mysteries in baking, to ensure the safety of your medicines, or even to understand the complex chemistry happening in the world around you. It’s a surprisingly accessible and incredibly fun way to peer into the hidden world of molecules and uncover their identities, one absorbed wave of light at a time!
