Choose The Compound That Matches The Ir Spectrum Shown.
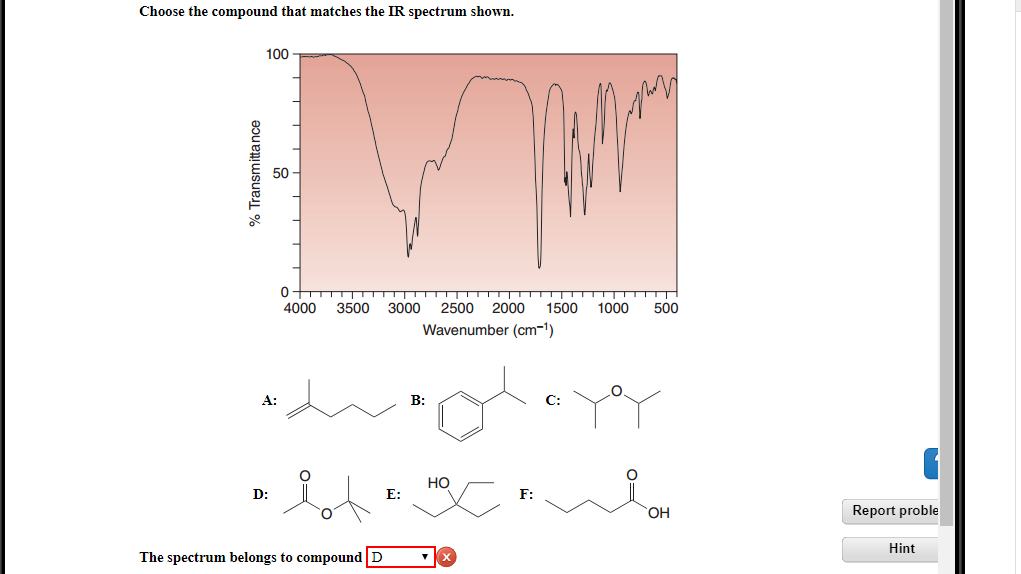
Hey there, science sleuths and spectral superheroes! Ever stare at a wiggly line graph and wonder, "What in the molecular mischief is going on here?" Well, buckle up, buttercups, because today we're diving headfirst into the wonderfully weird world of Infrared (IR) spectroscopy. Think of it as giving molecules a really good talking-to and listening to their unique "fingerprints" – those distinctive little wiggles in the spectrum. It’s like figuring out who’s singing the karaoke version of "Bohemian Rhapsody" just by listening to their voice! Fun, right?
So, imagine you've got this mystery compound, and someone hands you a beautiful, albeit slightly intimidating, IR spectrum. Your mission, should you choose to accept it (and let's be honest, you totally do because it's way more interesting than reorganizing your sock drawer), is to figure out which compound from a lineup of suspects is the culprit. It's like a molecular detective story, and we're about to become the Sherlock Holmeses of the lab!
Cracking the IR Code: The Basics (Without Making Your Brain Explode)
Alright, let's get down to the nitty-gritty, but don't worry, we're keeping it light and breezy. An IR spectrum basically shows us how much infrared light a molecule absorbs at different wavenumbers. Wavenumbers? Yeah, it's just a fancy way of measuring how many waves of light fit into a centimeter. Think of it as the "frequency" of the light, but for the IR realm. The higher the wavenumber, the higher the energy of the light. Simple enough, right? (Or as simple as organic chemistry gets, anyway!)
Why do molecules absorb IR light? Because they have bonds – you know, those little connections between atoms that hold everything together. And these bonds? They’re not just sitting there doing nothing. They're constantly vibrating. They stretch, they bend, they twist – it’s like a molecular dance party going on all the time! When a molecule encounters IR light with just the right amount of energy (which corresponds to a specific wavenumber), it can absorb that energy and get its bonds vibrating even more vigorously.
Each type of bond in a molecule vibrates at a characteristic frequency. A C-H bond vibrates differently than an O-H bond, which vibrates differently than a C=O bond. It’s like each bond has its own musical note. When you shine IR light through a sample, the molecule will only absorb the light that matches the vibrational frequencies of its bonds. Boom! That’s what creates those dips (or peaks, depending on how your instrument is set up – but usually, we see dips, so let's go with that) in your spectrum. These dips are the absorption bands, and they are our precious clues!
The "Fingerprint Region": Where the Magic Happens (and Gets Tricky)
Now, let's talk about the two main sections of an IR spectrum. Up on the left, typically above 1500 cm-1, we have what’s called the "functional group region." This is where the big hitters hang out. You’ll find characteristic peaks for things like:
- O-H bonds (like in alcohols or carboxylic acids): These usually show up as a broad, strong peak around 3300 cm-1. Think of it like a big, happy sigh of relief from the oxygen and hydrogen.
- C-H bonds (in alkanes, alkenes, and alkynes): These are super common and appear in the 2800-3100 cm-1 range. Alkyl C-H (single bonds) are usually below 3000 cm-1, while aromatic or vinylic C-H (double/triple bonds) are often above 3000 cm-1.
- C=O bonds (in aldehydes, ketones, esters, carboxylic acids): These are usually strong and sharp around 1700 cm-1. This is a HUGE clue for carbonyl compounds!
- C≡N bonds (nitriles): These show up as a sharp peak around 2250 cm-1.
- C≡C bonds (alkynes): These are usually weaker but can be seen around 2100-2260 cm-1.
These are your major giveaways. If you see a strong peak around 1700 cm-1, you immediately know you've got a carbonyl group in your molecule. Easy peasy, lemon squeezy! (Well, maybe not that easy, but you get the idea.)
Then, over on the right, usually below 1500 cm-1, we have the "fingerprint region." This is where things get really interesting, and frankly, a bit more challenging. Why? Because this region is packed with all sorts of vibrations: C-C stretches, C-O stretches, C-N stretches, C-H bends, and more. It's a complex tapestry of wiggles, and no two molecules have the exact same fingerprint here. It's like your unique collection of moles and freckles – nobody else has it quite like you!
The fingerprint region is incredibly useful for confirming a suspected structure or distinguishing between very similar compounds. If the functional groups match up, but the fingerprint region is different, you know they’re not the same molecule. It's the ultimate lie detector test for organic compounds!
Your Detective Toolkit: What to Look For
Okay, so you’ve got your spectrum. What’s your game plan? Here’s a step-by-step approach, like a seasoned gumshoe:
Step 1: Scan for the "Big Guys" in the Functional Group Region
Start on the left side of the spectrum (higher wavenumbers). Look for those prominent, easily identifiable peaks. Are there any broad, strong peaks in the 3200-3600 cm-1 range? That’s your clue for an O-H group. Any sharp, strong peaks around 1700 cm-1? Hello, carbonyl! Are there C-H stretches above or below 3000 cm-1? This tells you about the type of carbon the hydrogen is attached to.
Think of it this way: you're looking for the distinctive features of a person's face. Do they have a prominent nose? Big ears? A killer smile? These are your functional groups!
Step 2: Check for the "Absence of Evidence"
This is just as important as seeing what is there! If you don't see a peak where you expect one, that's a crucial piece of information. For example, if you suspect an alcohol, but there's no broad O-H stretch, then it’s probably not an alcohol (or it’s a very, very dry one!). If you’re looking at a compound that’s supposed to be an alkene, but there's no C=C stretch around 1650 cm-1, something’s up.
It's like interviewing witnesses. If someone didn't see the suspect, that's a vital piece of information too!
Step 3: Dive into the Fingerprint Region (with Caution!)
Once you’ve identified the major functional groups, it’s time to use the fingerprint region to fine-tune your suspicions. Compare the pattern of peaks in the 600-1500 cm-1 range with the spectra of your potential compounds. You're looking for a match, a perfect symphony of wiggles.
This is where having a good spectral database or reference spectra comes in handy. It’s like having a mugshot gallery to compare your suspect to. If the fingerprint region aligns perfectly, you’re probably on the right track!
Step 4: Consider the Intensity and Shape
Don’t just focus on the position of the peaks; their intensity (how tall or deep they are) and shape (broad or sharp) are also clues. For instance, hydrogen-bonded O-H groups (like in an alcohol that’s interacting with other molecules) are typically broader than free O-H groups. A very strong carbonyl peak suggests a typical ketone or aldehyde, while a slightly weaker one might indicate an ester or amide, which have resonance structures that affect the C=O bond.

Imagine a person’s voice. Is it loud and booming or soft and whispery? Is it a clear, sharp tone or a gravelly rasp? These details matter!
Putting It All Together: The "Choose the Compound" Challenge
So, let's say you're presented with this scenario:
The Spectrum: You see a strong, broad peak around 3300 cm-1. There's a strong peak around 1715 cm-1. And there are several sharp peaks in the 2850-2960 cm-1 range. The fingerprint region has a complex pattern with several peaks below 1500 cm-1.
The Suspects:
- Ethanol (C2H5OH): An alcohol.
- Acetone (CH3COCH3): A ketone.
- Acetic Acid (CH3COOH): A carboxylic acid.
- Ethyl Acetate (CH3COOCH2CH3): An ester.
Your Detective Work:
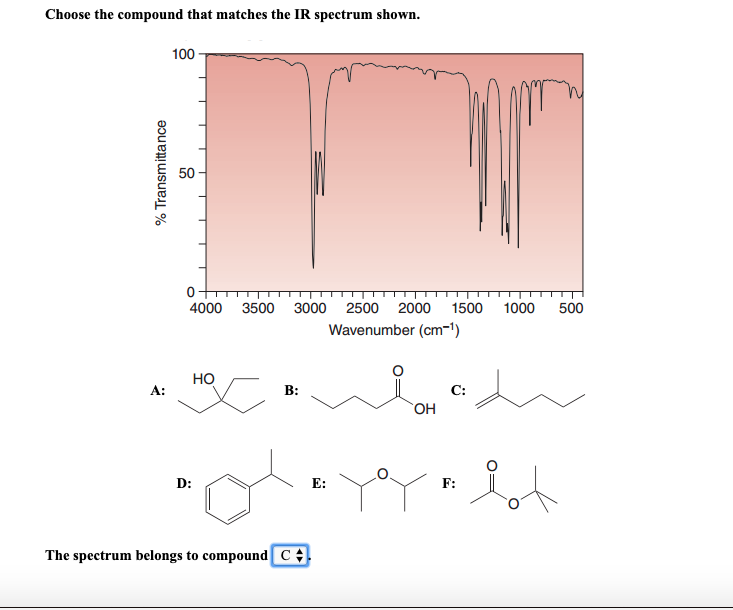
- The strong, broad peak at 3300 cm-1 screams O-H group! This immediately points towards a compound that has an alcohol or carboxylic acid functional group. Goodbye, acetone and ethyl acetate!
- The strong peak at 1715 cm-1 indicates a carbonyl group (C=O)! Hmm, this is interesting. Both acetic acid and ethyl acetate have carbonyls. Ethanol doesn't. So, acetone is out, and ethyl acetate is still in contention, but acetic acid is looking good!
- The peaks in the 2850-2960 cm-1 range are typical C-H stretches from alkyl groups. All our suspects have these, so they don't help us narrow it down further between acetic acid and ethyl acetate at this stage.
- Now, let’s combine the clues. We have BOTH an O-H and a C=O group. Which suspect has both? Acetic acid! Alcohols have O-H but no C=O. Ketones have C=O but no O-H. Esters have C=O but the oxygen is bonded to another carbon, not a hydrogen, so no broad O-H stretch.
Therefore, based on the presence of both a broad O-H stretch and a strong carbonyl stretch, the spectrum most likely belongs to acetic acid. The fingerprint region would then serve to confirm this identification by matching the complex pattern of peaks below 1500 cm-1 with the known spectrum of acetic acid.
A Little More Nuance: Carboxylic Acids vs. Alcohols
Let's just quickly touch on the subtle differences. Carboxylic acids have both a strong carbonyl peak (around 1700-1730 cm-1) and a very broad O-H stretch that often extends from around 2500 cm-1 all the way up to the O-H of alcohols (around 3300 cm-1). This broadness is due to strong hydrogen bonding in carboxylic acids. Alcohols, on the other hand, have a sharper O-H peak around 3300 cm-1 and no carbonyl peak. So, if you see that super wide, beefy O-H stretch plus a carbonyl, think carboxylic acid!
And What About Aldehydes vs. Ketones?
Both have carbonyls around 1700-1730 cm-1. The key difference lies in the C-H stretch of the aldehyde group. Aldehydes typically show a weak to medium intensity peak in the 2700-2850 cm-1 range, often accompanied by a slightly weaker one around 2700-2750 cm-1. These are your "aldehyde C-H stretches." Ketones, which only have C-C bonds to the carbonyl carbon, do not have these characteristic aldehyde C-H peaks.
The Joy of Discovery!
Honestly, isn't it just the coolest thing? You're taking this invisible, intangible light energy and using it to see into the very heart of molecules. You’re deciphering their secrets, one wiggly line at a time. It’s like being a detective, a scientist, and a bit of a wizard all rolled into one!
Every spectrum is a puzzle, and with a little practice and a good understanding of those key functional groups, you can become a master at solving them. So, the next time you’re faced with an IR spectrum, don’t feel intimidated. Smile, grab your detective hat, and get ready to enjoy the thrill of discovery. Because in the world of chemistry, understanding these spectral fingerprints is like unlocking a whole new language, and it’s a language that’s beautiful, powerful, and endlessly fascinating. Go forth and spectro-fy!
