Choose The Compound Responsible For The Ir Spectrum Shown.
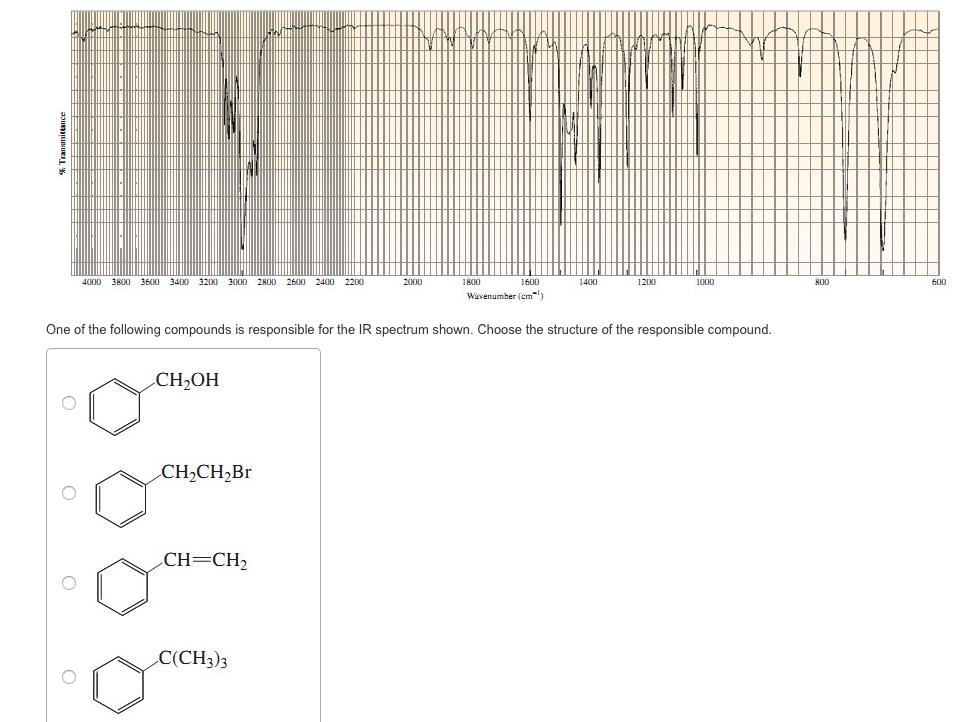
Alright, gather 'round, folks, and let me spin you a yarn about a little something called an IR spectrum. Now, I know what you're thinking: "IR spectrum? Is this some kind of fancy new dance move or a particularly aggressive yoga pose?" Nope, my friends, it's way cooler, and way more… invisible. Think of it as a chemical fingerprint, a secret handshake that molecules do when you shine a special kind of light on them. And today, we're going to be detectives, sniffing out the culprit behind a mysterious spectrum. Prepare for intrigue, suspense, and maybe a mild case of caffeine jitters from this imaginary café chat!
So, picture this: a bunch of scientists, probably fueled by copious amounts of espresso, are staring at a squiggly line on a screen. This squiggly line, my dear Watson, is our IR spectrum. It’s basically a graph showing how much infrared light a molecule eats. Different parts of the molecule, like little molecular taste buds, get excited by different wavelengths of this infrared light, and they slurp it up. The spectrum is just a record of which wavelengths got devoured.
Now, we’ve got a suspect list. Three chemical suspects are lined up, looking all innocent and innocent-like. We've got:
Suspect A: Ethanol
This guy is basically the life of the party. You know, the one with the slightly sweet smell and the tendency to make your karaoke debut legendary. Chemically speaking, it’s an alcohol, with an -OH group chilling out. This -OH group is pretty chatty, loves to bond with its buddies (hydrogen bonds, they call 'em), and it's got a particular craving for certain infrared wavelengths. Think of it as a picky eater who only eats broccoli if it’s perfectly steamed.
Suspect B: Acetic Acid
Ah, acetic acid! This is the stuff that gives vinegar its kick. It's got a bit of a sharp personality, and in the chemical world, it's a carboxylic acid. This means it has not only an -OH group, but also a C=O group (a carbon double-bonded to an oxygen). This duo is like a power couple, and they have very distinct dietary preferences when it comes to infrared light. They’re the kind of couple that goes to fancy restaurants and orders the most complicated dish on the menu.
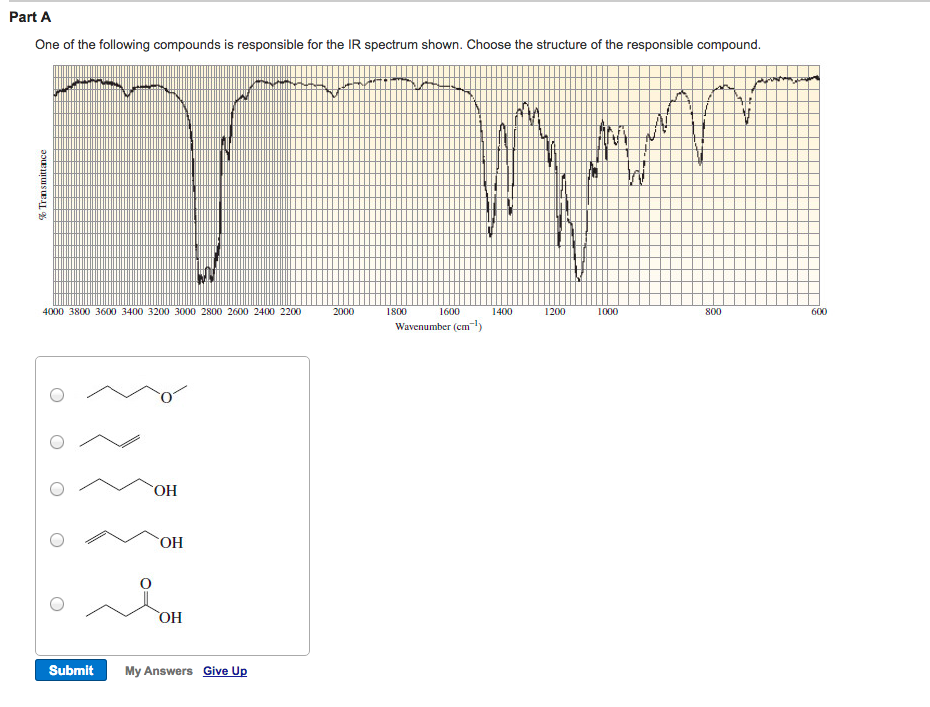
Suspect C: Acetone
And finally, acetone! The humble nail polish remover. This is a ketone, and its defining feature is that C=O group. No -OH here, just a simple, elegant double bond to oxygen. It’s like the minimalist of the group, with a very focused palate. It’s not trying to be fancy; it just knows what it likes, and it likes it specific. Think of it as the friend who always orders plain cheese pizza, no questions asked.
Our IR spectrum, the evidence we’ve gathered, is our witness. It’s got some key features that will help us nab the right suspect. We're looking for some specific peaks, these little dips in the spectrum. Each dip is like a shout-out from a particular functional group saying, "I'm here! And I just ate this specific wavelength of light!"
Let’s zoom in on the clues. One of the first things we’re looking for is a big, broad peak in the region of about 3200-3600 cm⁻¹. This is the tell-tale sign of an O-H stretch. Now, who has an O-H group? Both ethanol and acetic acid! But, aha, acetic acid's O-H is part of a carboxylic acid dimer, which is like two acetic acid molecules holding hands really tightly. This makes its O-H peak even broader and a bit shifted, usually appearing somewhere between 2500-3300 cm⁻¹, often looking like a fuzzy caterpillar instead of a sharp line. Ethanol’s O-H peak, being an alcohol, is generally sharper and in that 3200-3600 cm⁻¹ range, like a distinct finger pointing at you.
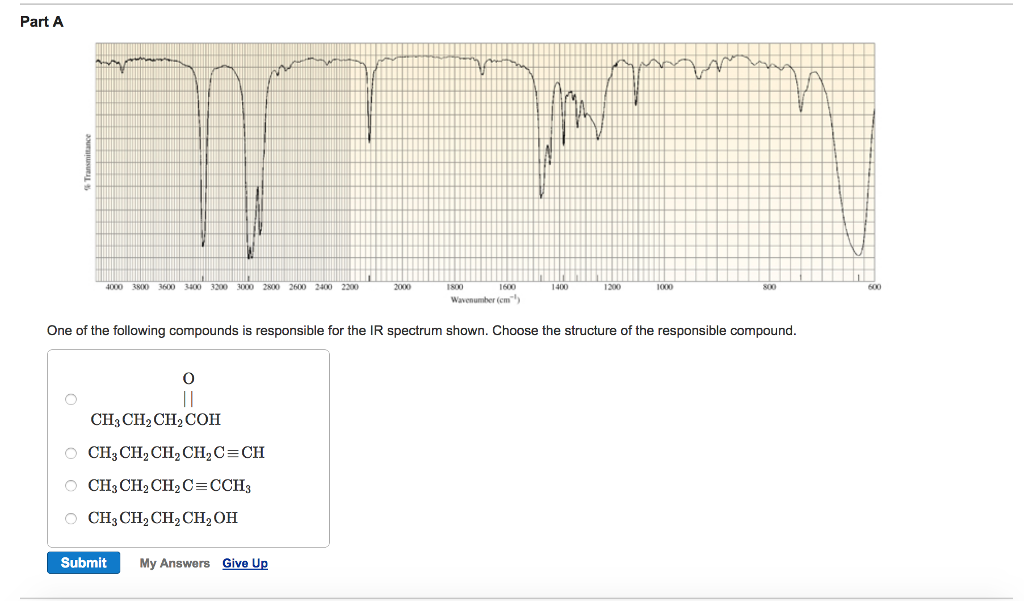
Next up, we’re on the hunt for a C=O stretch. This is a really strong signal, and it’s usually found in the fingerprint region, somewhere between 1650-1800 cm⁻¹. It’s like the molecule’s loudspeaker, announcing its presence with gusto. Who has a C=O group? Both acetic acid and acetone! But there’s a difference, a subtle nuance that separates the sophisticated from the… well, still pretty sophisticated, but in a different way.
Acetone, our ketone, has a C=O group that typically appears around 1715 cm⁻¹. It’s a nice, clear peak, saying, "Yep, I'm a ketone, deal with it." Acetic acid, being a carboxylic acid, also has a C=O, but because it’s bonded to two oxygens (one through the double bond, one through the single bond of the -OH), its C=O peak is usually a bit higher in frequency, often around 1710-1725 cm⁻¹, and sometimes it can look a bit wider because of all the interactions going on. It's like acetone is a solo artist, and acetic acid is part of a duet.
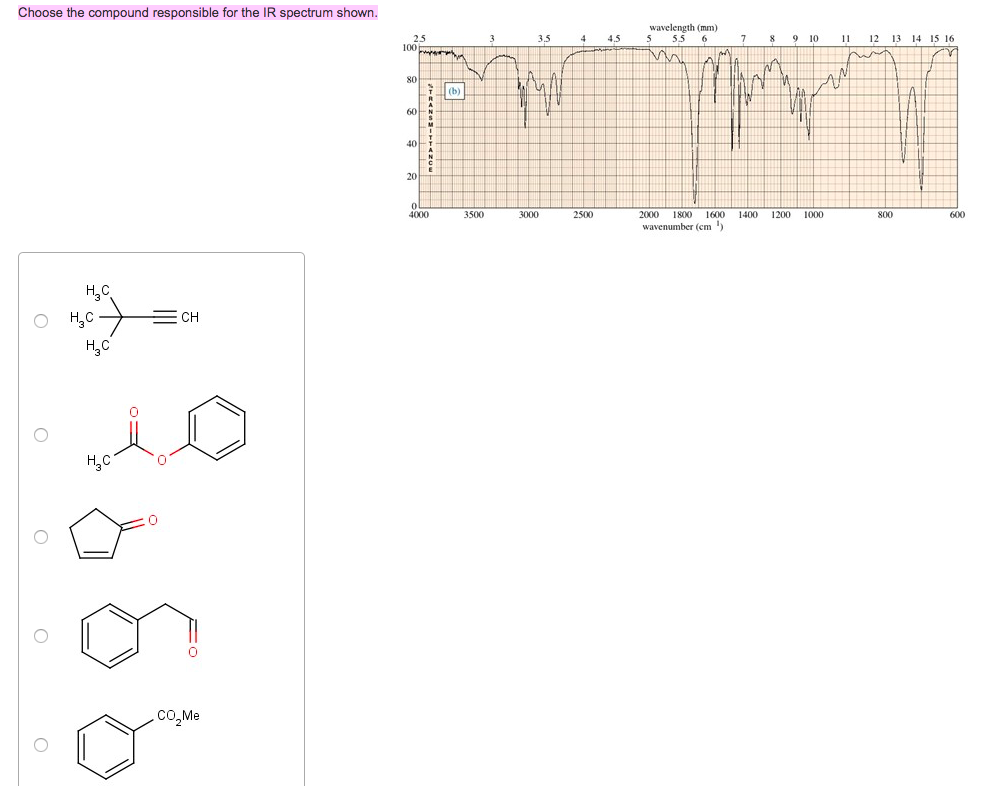
So, let’s put it all together. If we see that broad, fuzzy O-H stretch in the 2500-3300 cm⁻¹ range, AND a strong C=O stretch around 1710-1725 cm⁻¹, who do you think our culprit is? It’s not ethanol, because it doesn’t have that prominent C=O. It’s not acetone, because it’s missing that characteristic broad O-H.
It has to be Acetic Acid! It’s the only one on our suspect list that has both that distinctive, dimerized O-H stretch AND a C=O group in the right neighborhood. It’s the molecular equivalent of finding a single glove and a half-eaten donut at the scene of the crime – tells a pretty specific story!
Isn’t science grand? We take these invisible light waves, bounce 'em off molecules, and suddenly we can tell who’s who. It’s like a molecular identity parade. Next time you’re staring at a weird squiggle, remember, it’s not just random lines. It’s a story, a chemical confession, waiting to be deciphered. Now, who’s for another espresso? My detective work has made me thirsty!
