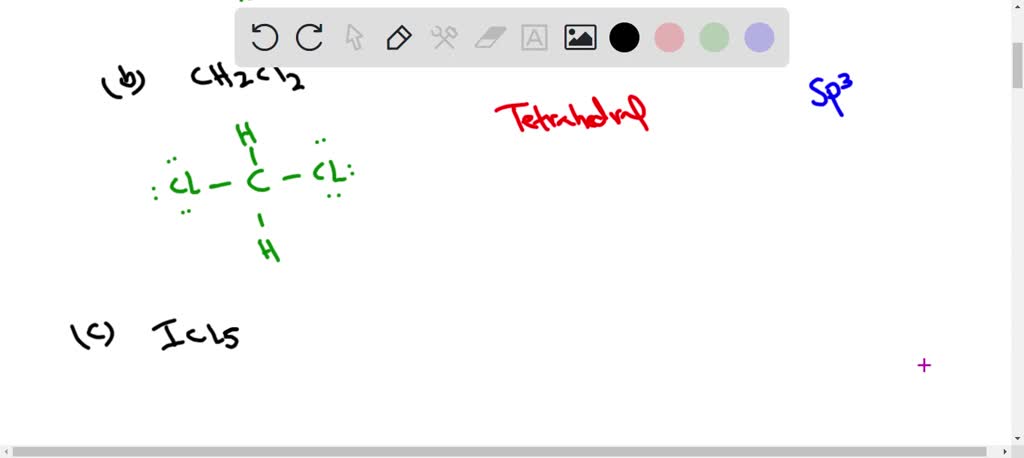
Choose The Best Electron-dot Structure For Ch2cl2.

Hey there, science adventurers! Ever find yourself staring at a molecule and thinking, "Hmm, how do those little fellas really hang out?" Well, today we're diving into the wonderfully whacky world of electron-dot structures, and we're going to pick the perfect one for a molecule called CH₂Cl₂. Don't let the fancy name scare you; it's just a fancy way of saying "dichloromethane," and it's a pretty common guy in the chemical world. Think of it like a molecular best friend, and we're about to figure out its favorite way to hold hands!
So, what's the big deal about electron-dot structures? Imagine you've got a group of friends, and they all have a certain number of "energy points" (those are electrons, by the way!). To be super stable and happy, they like to share these points in a way that makes everyone feel like they have a full set. Electron-dot structures, also known as Lewis structures, are like little diagrams showing exactly how these atoms are sharing their electrons. It’s like drawing a friendship circle with everyone holding hands!
Now, let's zoom in on our star, CH₂Cl₂. We’ve got one carbon atom (C), two hydrogen atoms (H), and two chlorine atoms (Cl). The carbon is the central player here, usually. Think of it as the life of the party, always trying to connect with everyone. Hydrogen is a bit of a minimalist; it's happy with just one electron to share. Chlorine, on the other hand, is a bit of a show-off with its seven valence electrons, always looking to complete that magic number eight.
To figure out the best electron-dot structure, we have a few rules, like secret handshake protocols for molecules. First, we count up all the valence electrons. Carbon has 4, each hydrogen has 1 (so 2 total for the two hydrogens), and each chlorine has 7 (so 14 total for the two chlorines). Add 'em all up, and you get a grand total of 4 + 2 + 14 = 20 valence electrons. That's the total number of "dots" we need to distribute.
Next, we arrange the atoms. Carbon is the least electronegative (meaning it's not super eager to hog electrons), so it's usually in the center. Our two hydrogens and two chlorines will be hanging off the carbon like ornaments on a molecular Christmas tree. We connect them with single bonds, which represent a shared pair of electrons. So, we've got C bonded to two H's and two Cl's. That uses up 4 bonds, which means 4 pairs of electrons, or 8 electrons. We've still got 20 - 8 = 12 electrons left to place.
The Dotting Dilemma
Now comes the fun part: filling in the rest of the electrons. We want to give each atom a full outer shell, usually meaning eight electrons (except for hydrogen, which is happy with two). We start by giving the outer atoms (the hydrogens and chlorines) their "lone pairs" – those electrons that aren't involved in bonding but just hang around the atom. Remember, we have 12 electrons left. The chlorines are a bit more electron-hungry, so they get the bulk of these lone pairs. Each chlorine needs 6 more electrons to complete its octet (it already shares 2 in the bond). So, we give each chlorine 3 lone pairs (6 dots). That uses up 6 + 6 = 12 electrons. And guess what? We've used all our electrons!
Let's check our work. Carbon has four bonds, meaning it's sharing 8 electrons (4 pairs). Perfect! Each hydrogen has one bond, sharing 2 electrons. Also perfect! And each chlorine has one bond (sharing 2 electrons) and 3 lone pairs (6 electrons), giving it a total of 8 electrons. Hooray! It looks like we've got a winner!

But wait, what if we considered other possibilities? Could we have double bonds? For example, what if one of the chlorines formed a double bond with the carbon? That would mean sharing 4 electrons. Then, the carbon would have two single bonds and one double bond, giving it 10 electrons. Uh oh, that's too many for carbon! Carbon is generally happy with just 8. So, that structure wouldn't be as stable or "happy." It’s like trying to cram too many people into a tiny car – it just doesn’t work well.
Another thing to consider is formal charge. This is a way to assign a "charge" to each atom in the structure, even if the overall molecule is neutral. It helps us find the most stable and likely arrangement. The formula is: (Number of valence electrons) - (Number of non-bonding electrons) - (1/2 * Number of bonding electrons). Let's apply this to our proposed CH₂Cl₂ structure. Carbon: 4 - 0 - (1/2 * 8) = 0. Hydrogen: 1 - 0 - (1/2 * 2) = 0. Chlorine: 7 - 6 - (1/2 * 2) = 0. All zeros! This is fantastic! It means the electrons are distributed perfectly, with no "extra" positive or negative charges hanging around on any individual atom. This is the hallmark of a truly best electron-dot structure.
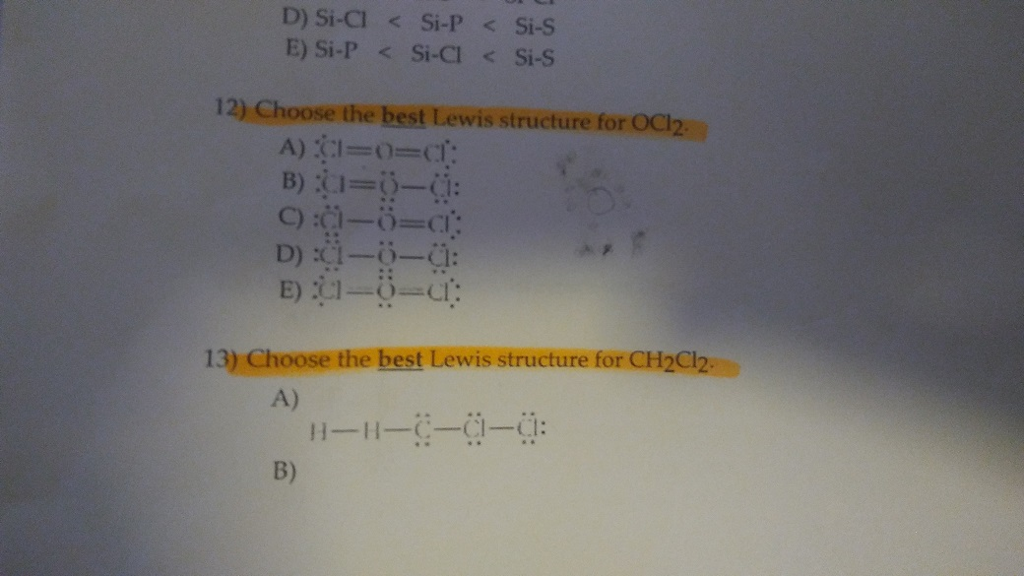
Now, imagine if we had a structure where, say, a chlorine had a +1 formal charge and another atom had a -1 formal charge. While the overall molecule might still be neutral, those formal charges create instability, like a slight tug-of-war that the atoms would prefer to avoid. So, the structure with all zero formal charges is undeniably the king of the hill, the champ, the best choice.
Why does this even matter? Well, understanding these electron arrangements helps us predict how molecules will behave. It’s like knowing your friend’s favorite snacks; you can guess what they’ll reach for at a party! This knowledge is crucial in everything from designing new medicines to creating new materials. It’s the foundation of chemistry, the secret language of the universe!

Think about it: every time you use a plastic bottle, wear a synthetic fabric, or even breathe, you're interacting with molecules and their electron structures. It’s not just abstract stuff in a textbook; it's the building blocks of everything around you! And learning to draw these structures is like gaining a superpower – the ability to visualize and understand the invisible world.
So, the next time you see CH₂Cl₂ or any other molecule, don't just see letters and numbers. See a story unfolding, a dance of electrons, a quest for stability. The best electron-dot structure for CH₂Cl₂ is the one where everyone is sharing nicely, no one is overloaded, and all is right in the molecular universe. It’s a beautiful illustration of how things work at the most fundamental level, and frankly, it’s pretty darn cool!
Embracing this kind of scientific curiosity can truly inject a dose of wonder into your day. It’s about asking "why?" and then going on a little adventure to find the answer. So, keep exploring, keep questioning, and remember that even the most complex-looking molecules are just collections of atoms playing a fascinating game of electron sharing. The world is full of molecular marvels waiting to be discovered, and you've got the perfect tools (your brain and a little bit of chemistry knowledge!) to start unraveling them. Go forth and be scientifically inspired!
