Chemwiki Ucdavis Edu Physical Chemistry Acids And Bases Ph Scale
Let's talk about something that sounds super serious but is actually kind of… fizzy. We're diving into the wild world of acids and bases, with a little help from Chemwiki Ucdavis Edu. You know, the place where all the smarty pants learn about the tiny stuff that makes everything else tick. And what’s the main attraction? The pH scale!
Now, I’ve got an unpopular opinion about pH. Ready? It’s not that complicated. Seriously. We’re taught to be terrified of it, like it’s some kind of pop quiz from a grumpy professor. But it’s just a way to measure how… well, how something feels. Like a sour gummy worm versus a creamy milkshake.
Think about it. You’ve got your acids. They’re the zingy, tangy ones. Lemons? Definitely acidic. Vinegar? That’s your go-to for a salad dressing that bites back. Even your own stomach uses hydrochloric acid to, you know, digest that questionable late-night snack. It’s basically the enthusiastic friend who’s always a little too loud.
Then, there are the bases. These are the slippery, sometimes bitter ones. Think of that feeling when you’re washing your hands with soap. A little bit slick, right? That’s a base at work. Baking soda? Your mom probably used it to calm down a bee sting. It’s the chill one, the one who says, "Let's all just relax."
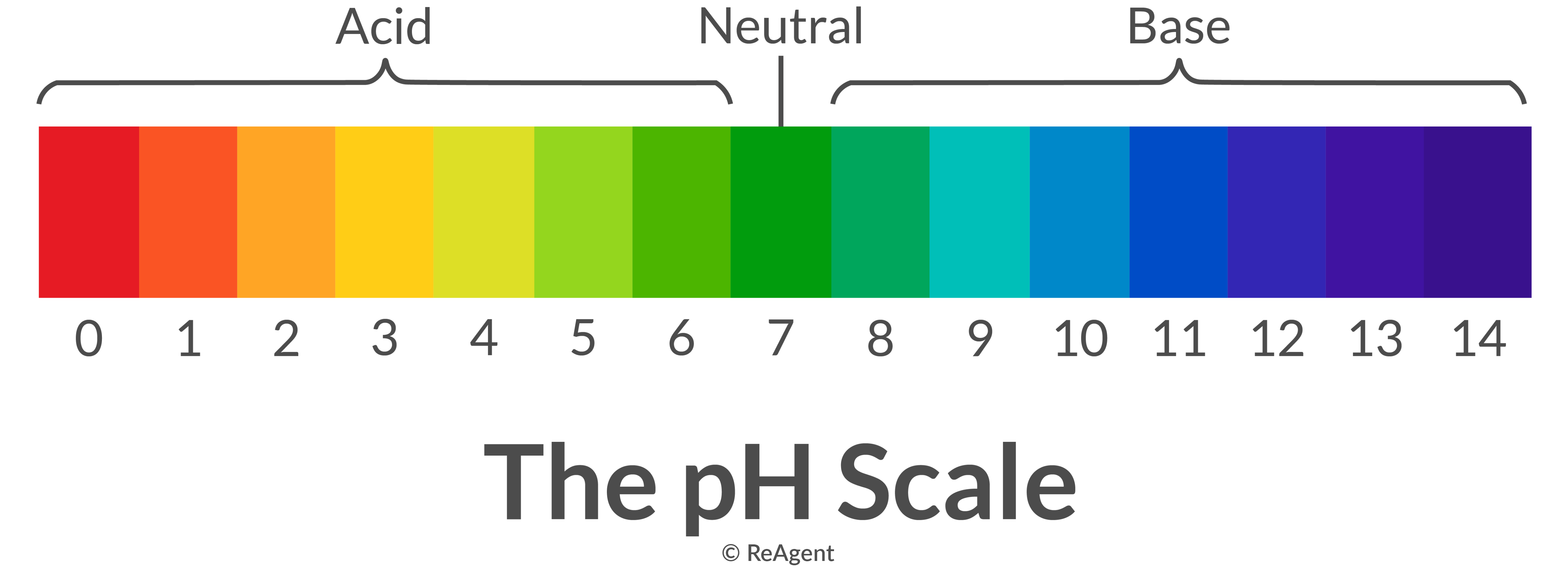
So, where does the pH scale come in? Imagine a ruler. But instead of inches, it measures… oomph. From 0 to 14.
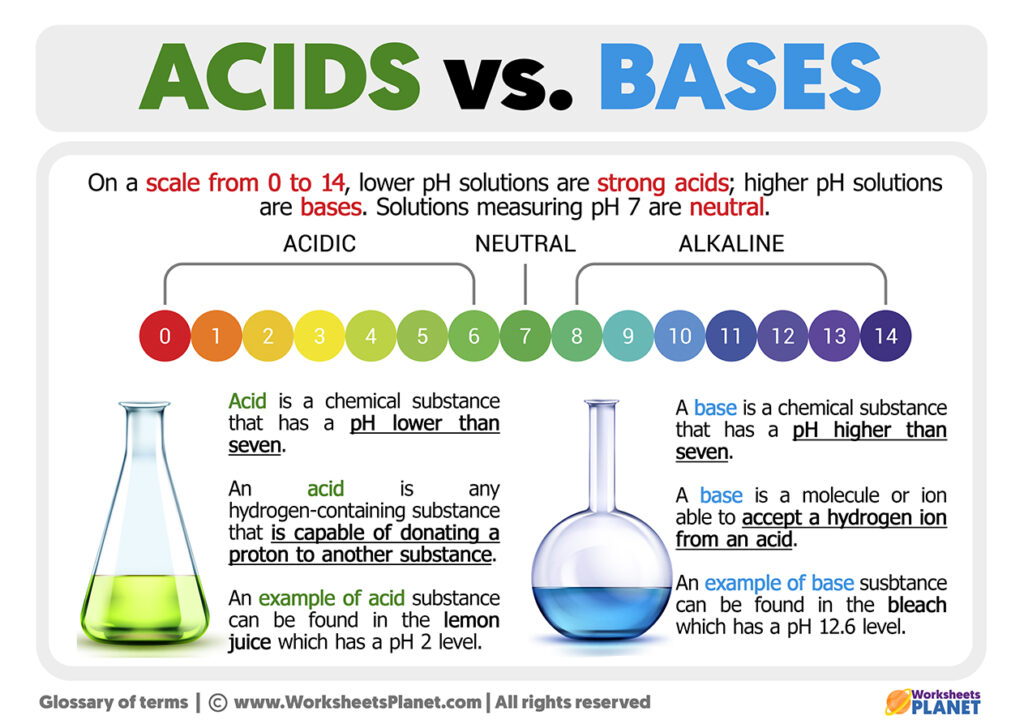
The magic number is 7. That’s the neutral zone. Like pure water. It’s not leaning one way or the other. It’s the Switzerland of chemistry.
Anything below 7 is acidic. The lower you go, the more acidic it gets. So, a pH of 1 is like a super-sour candy that makes your eyes water. A pH of 6 is more like… well, a slightly tart apple. You get the picture.
And anything above 7 is basic. The higher you go, the more basic it is. A pH of 8 is like mild soap. A pH of 14? That’s your really strong cleaning stuff. The kind you wear gloves for. It’s like the opposite end of that super-sour candy – it’s the super-smooth, almost too-gentle giant.

Chemwiki Ucdavis Edu probably goes into all the nitty-gritty details. All the chemical formulas and reactions. But at its heart, it’s about this simple concept of balance. Or imbalance, depending on what you’re looking at.
Why do we care? Because this stuff is everywhere! It’s in the rain that falls, the soil that grows our food, and even in our own bodies. Your blood has a very specific pH it needs to maintain. Too acidic or too basic, and things get… unpleasant. Really, really unpleasant. Your body is basically a master chef, constantly adjusting the ingredients to keep everything just right.

Think about a swimming pool. You don't want it to be too acidic, or it'll sting your eyes. You also don't want it to be too basic, or it'll feel slimy. The lifeguards are secretly pH wizards.
And what about science experiments? Ever seen those colourful reactions where liquids change colour? Often, it’s because you’re adding an acid or a base. It’s like a magic show, but with chemicals. And it’s all thanks to the pH scale.
So, next time you hear about acids and bases and the pH scale, don't get intimidated. Just think about sourness and slipperiness. Think about that ruler of oomph. And remember that even the most complex scientific concepts can be broken down into something… well, something you can actually understand. And maybe even chuckle about. Because who knew that chemistry could be so… relatable? It’s not just for textbooks; it’s for understanding why that lemon makes your mouth pucker. And that, my friends, is pretty cool. Even if it sounds a bit like something from a very organized, very informative, and let's be honest, slightly intimidating website. But hey, UCDavis knows its stuff! They're just sharing the secrets of the universe, one pH unit at a time. And it's all pretty darn logical when you stop overthinking it. It’s just a scale. A really, really useful scale.
