Chemistry Worksheet Oxidation Reduction Reactions 1 Answers

So, you’ve been staring at a chemistry worksheet, right? Specifically, one about oxidation-reduction reactions. Maybe you’re a little… mystified. Totally get it. It sounds super fancy, like something from a sci-fi movie. But guess what? It’s actually a lot cooler than it sounds. And the answers? Well, they’re the key to unlocking this whole electrifying world.
Let’s be real, sometimes those worksheets can feel like a cryptic puzzle. Numbers, arrows, weird symbols everywhere. Your brain might be doing a little happy dance to escape. But stick with me! We’re gonna break it down. Think of it as a treasure hunt, and the answers are your map.
What’s the Big Deal with Redox?
Okay, so what IS oxidation-reduction? In super simple terms, it’s all about electron swapping. Imagine atoms as tiny little dancers. Sometimes they’re feeling generous and give away an electron. Other times, they’re feeling a bit needy and snatch one up. That’s the core of it. Seriously, just electrons being passed around like a hot potato.
The “oxidation” part usually means an atom is losing electrons. Think of it as getting a bit “oxidized,” like a rusty nail. It’s gone through a change, and electrons are involved. The “reduction” part is the opposite – an atom is gaining electrons. It’s like it’s getting “reduced” in some way, but in a good way, because it’s gaining something valuable (those electrons!).
These two things always happen together. You can’t have one without the other. It’s like a cosmic dance, a chemical tango. One dancer loses, the other gains. Always. That’s why we call them redox reactions. It’s a mashup of oxidation and reduction. Catchy, right?
Why Should You Even Care About Electron Swapping?
You might be thinking, “Okay, electrons are moving. So what?” Well, this electron swapping is happening everywhere. It’s the basis of so many things we use and experience every single day. Your phone? Powered by redox reactions. Your car? Runs on them. Even the air you breathe involves redox. Pretty wild, huh?
Think about batteries. They are essentially controlled redox reactions. Little electron highways sending power where you need it. Or digestion! Your body is a master chemist, breaking down food through a series of redox steps. So, those worksheet answers? They’re not just abstract chemistry stuff. They’re about understanding the fundamental forces that power our world.

Decoding Your Worksheet Answers
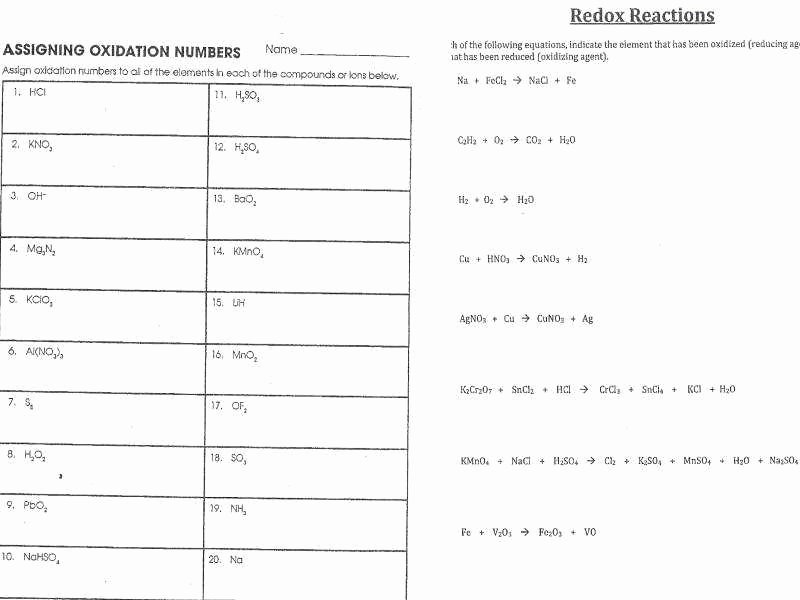
Alright, let’s get down to the nitty-gritty of those answers. You probably saw a bunch of oxidation states. These are like little labels that tell you how many electrons an atom seems to have gained or lost. They’re not always the actual charge, but they’re super useful for tracking those sneaky electrons.
The rules for assigning oxidation states can seem a bit tricky at first. For example, elements in their pure form (like a chunk of pure iron, Fe) have an oxidation state of 0. Makes sense, right? No electrons gained or lost yet. Oxygen usually likes to be -2, except when it’s paired with fluorine (because fluorine is a bit of a bully and steals electrons from everyone).
So, when you look at your worksheet answers, and you see an atom’s oxidation state change from, say, +2 to 0, you know it gained electrons. That’s reduction! If it goes from -1 to +1, it lost electrons. That’s oxidation!
The key is to identify what’s getting oxidized and what’s getting reduced. Your answers will help you see these changes clearly. You might be circling atoms, drawing little arrows, or writing “Ox” and “Red” next to them. Whatever method your worksheet suggests, follow it!
The Fun Bits: Finding the Culprits
One of the fun parts of redox is identifying the oxidizing agent and the reducing agent. These sound like superhero names, don’t they? The oxidizing agent is the substance that causes something else to be oxidized (meaning it itself gets reduced – it takes the electrons). The reducing agent is the substance that causes something else to be reduced (meaning it itself gets oxidized – it gives away the electrons).
Imagine a game of tag. The person who is “it” is the reducing agent. They’re the one doing the chasing, giving energy. The person who gets tagged is the oxidized one. The person who is “it” is actively causing a change in the other person.
Sometimes, worksheets will ask you to identify these agents. Look at your answers. If you’ve identified that substance X got reduced (gained electrons), then substance X is the oxidizing agent. If you’ve identified that substance Y got oxidized (lost electrons), then substance Y is the reducing agent. It’s a bit of a word puzzle, but once you get it, it clicks!
Common Quirks and Funny Details
Did you know that rust is basically a redox reaction? Iron reacts with oxygen and water. The iron gets oxidized, and the oxygen gets reduced. So, that beautiful patina on an old statue? It’s chemistry in action! And honestly, a little bit of controlled rust (like in some cool iron sculptures) is pretty neat.

Or how about the tarnish on your silverware? That’s another redox reaction, usually involving sulfur. It’s the silver reacting with sulfur compounds in the air. A little polish, and you’re reversing that reaction! It’s like a mini-chemical spa day for your cutlery.
Sometimes, the sheer speed of redox reactions is mind-blowing. Think of an explosion. That’s a super-fast redox reaction. Lots of energy released very quickly. On the flip side, some redox reactions are incredibly slow, like the weathering of rocks over millions of years. Nature is the ultimate chemist, playing with reaction speeds.
When Things Get a Little Tricky (But Still Fun!)
There are a few special cases that can trip people up. For example, disproportionation reactions. These are where a single element is both oxidized and reduced in the same reaction. It’s like an atom doing a double-flip. It might lose an electron to one neighbor and gain an electron from another. Pretty chaotic, but perfectly valid chemistry!
Another tricky one is balancing redox reactions. Sometimes, it’s not just about making sure the atoms are equal on both sides. You also have to make sure the total charge is balanced. That’s where those oxidation states really come in handy. Your worksheet answers will be your guide to getting this just right.

Don’t be afraid if some of these take a moment to sink in. Chemistry is like learning a new language. The more you practice, the more fluent you become. And honestly, understanding redox reactions is like getting a peek behind the curtain of how the universe works. It’s pretty darn cool.
Your Answers Are Your Superpower!
So, take a deep breath. Look at those answers on your worksheet. They are your confirmation, your guiding light. They show you where the electrons have been, who’s been a generous electron donor, and who’s been a greedy electron receiver. They are the keys to understanding the energetic dance of atoms.
Don’t just look at them and move on. Think about them. Why did this atom change its oxidation state? What does that tell you about electron transfer? The more you engage with the answers, the more you’ll understand the underlying principles.
And remember, the world of chemistry is full of fascinating, sometimes quirky, and always important phenomena. Redox reactions are a huge part of that. So, next time you see those numbers and symbols, embrace the electron-swapping adventure. It’s way more exciting than it looks!
