Chemistry Worksheet Lewis Dot Structures Answer Key

Ever feel like you’re just… going through the motions? Like your days are a bit too predictable, a tad too… well, basic? We get it. Life can sometimes feel like a perfectly balanced chemical equation, where everything has its place and nothing truly surprises us. But what if we told you there’s a way to inject a little more spark, a little more oomph into your understanding of the world, even the seemingly dry bits like chemistry worksheets?
Today, we’re diving headfirst into the fascinating world of Lewis Dot Structures, and more importantly, the often-elusive answer key. Think of it as your secret decoder ring to unlocking the molecular mysteries that surround us, from the fizz in your soda to the air you breathe. And don’t worry, we’re not going to drown you in complex jargon. We’re talking easy-going, lifestyle-friendly chemistry here. So, grab your favorite beverage – perhaps a sparkling water, a perfect example of molecular interactions! – kick back, and let’s explore.
The Magic of the Dots: What Exactly ARE Lewis Dot Structures?
Alright, let's break it down. Imagine you have a LEGO set. Lewis Dot Structures are kind of like the instruction manual for building molecules, but instead of plastic bricks, we’re dealing with atoms, and instead of complex diagrams, we have… dots! Each dot represents a valence electron – those are the outer shell electrons that are ready to mingle and form chemical bonds.
So, for instance, if you look at a simple molecule like water (H₂O), a Lewis Dot Structure shows you how the two hydrogen atoms and the one oxygen atom are connected, and how all those little valence electron dots are arranged around them. It’s a visual representation of how atoms share or transfer electrons to achieve stability, kind of like how we might share our Netflix password to make everyone happy (okay, maybe not exactly like that, but you get the idea!).
Why is this important? Because understanding these structures helps us predict how molecules will behave. Will they be reactive? Will they dissolve easily? Will they smell like that questionable thing you found at the back of the fridge? All of this can be hinted at by their Lewis Dot Structure. It's like having a superpower that lets you peek into the hidden world of atoms!
Unlocking the Secrets: The Elusive Answer Key
Now, let’s talk about the elephant in the room: the answer key. We’ve all been there, staring at a worksheet, drawing our best guess at those electron dots, and then… panic. Did we get it right? Is that a valid arrangement, or did we just accidentally create a molecule that will spontaneously combust (spoiler: probably not, but in our heads, the stakes are high!).
The answer key is your trusty sidekick, your guiding star. It’s not just about checking your work; it’s about learning. Think of it as a curated gallery of perfectly drawn Lewis Dot Structures. By comparing your attempts to the correct ones, you start to pick up on the patterns, the rules of the game. It’s like seeing a perfectly styled outfit and then trying to replicate that chic vibe in your own wardrobe.
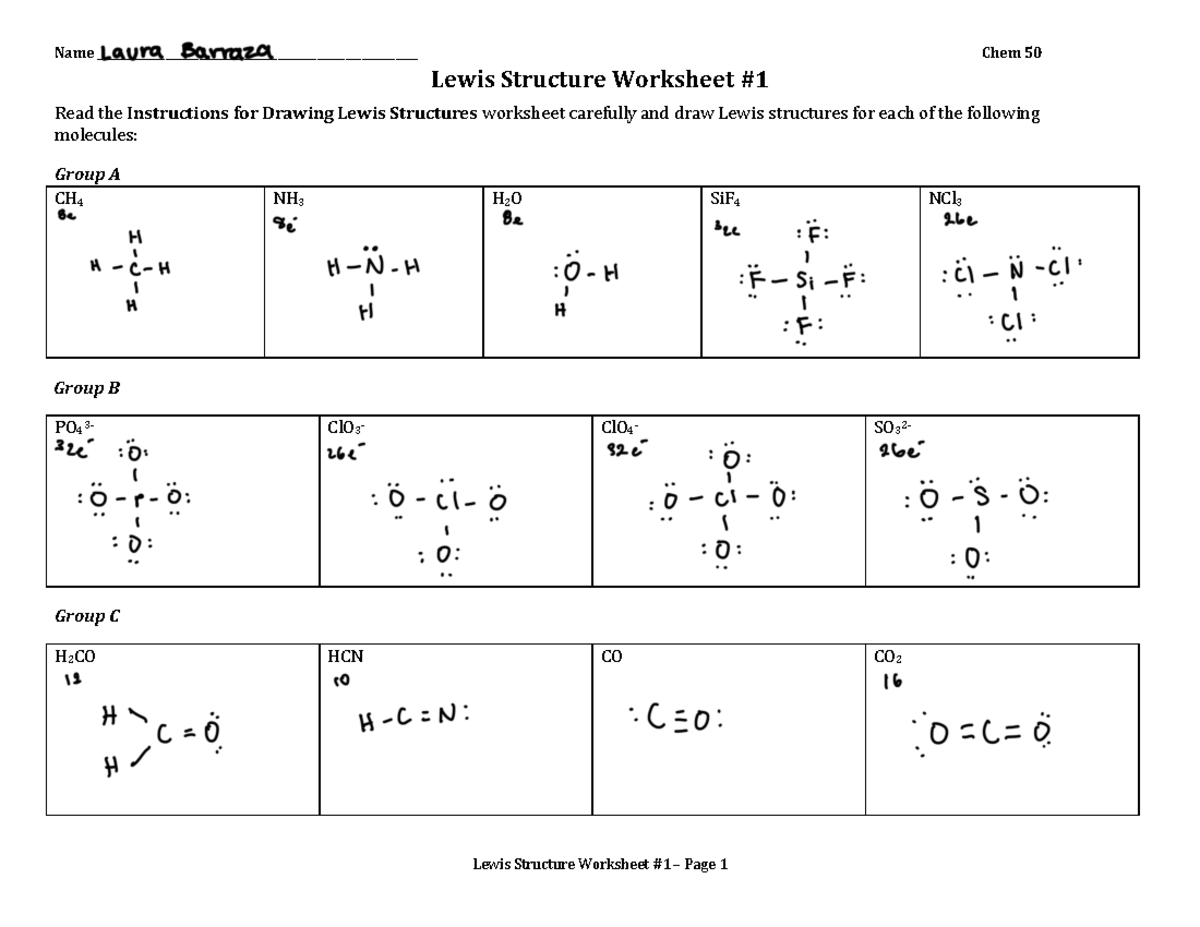
Finding a reliable Lewis Dot Structures answer key is crucial. You want one that’s clear, accurate, and covers a good range of common molecules. Many textbooks come with them, and online resources abound. Just remember, the goal isn’t to blindly copy. It’s to use it as a tool for understanding. If your structure looks nothing like the answer key’s, don’t despair! It’s an opportunity to go back, re-evaluate, and ask yourself, "Where did I go wrong?"
Practical Tips for Mastering Lewis Dots (Without the Stress!)
Let’s be honest, staring at a blank worksheet can feel a bit like staring into the void. But we can make this process smoother, more enjoyable, and dare we say, even fun. Here are a few tips:
1. Start with the Basics (The Molecular Equivalent of a Simple Black Dress)
Don’t jump straight into complex organic molecules. Begin with the fundamentals: diatomic molecules (like O₂, N₂, Cl₂) and simple polyatomic ions. These are the building blocks. Once you’ve got a handle on them, everything else will feel less daunting. It’s like mastering the basic knots before you try to tie a sailor’s bowline.
2. Count Those Electrons! (Your Molecular Inventory)
This is the golden rule. Before you even start drawing, count the total number of valence electrons for all the atoms in the molecule or ion. This is your electron budget. You can’t spend more than you have! For example, for water (H₂O), hydrogen has 1 valence electron, and oxygen has 6. So, 2 hydrogens * 1 electron/hydrogen + 1 oxygen * 6 electrons/oxygen = 8 valence electrons total. Keep this number handy!

3. Sketch and Connect (The Molecular Blueprint)
Draw the atoms, put the least electronegative atom (usually carbon or the central atom) in the middle, and connect them with single bonds (each bond uses 2 electrons). Then, distribute the remaining electrons as lone pairs around the outer atoms to satisfy the octet rule (most atoms want 8 electrons around them, like a cozy little electron cuddle). Any leftover electrons go on the central atom.
4. Double and Triple Bonds: The Molecular Remix
If your central atom doesn’t have an octet, try forming double or triple bonds by moving lone pairs from outer atoms to create more bonds. This is where the structure really starts to come alive! Think of it as rearranging your furniture to make the room feel more spacious and functional.
5. Formal Charge: The Molecular Scorecard
This is a slightly more advanced but super useful tool. Formal charge helps you determine the most likely Lewis structure when you have multiple possibilities. It's calculated as (valence electrons) - (non-bonding electrons) - (1/2 bonding electrons). The structure with the formal charges closest to zero is usually the most stable. It’s like giving each atom a little score to see who’s playing nicely.
6. Use Online Tools (Your Digital Muse)
There are some fantastic online Lewis Dot Structure drawing tools and calculators out there. They can help you visualize the process and check your work. It’s like having a digital sketchpad that can instantly show you if your proportions are right. Just don’t rely on them entirely; the learning happens when you do the drawing yourself!
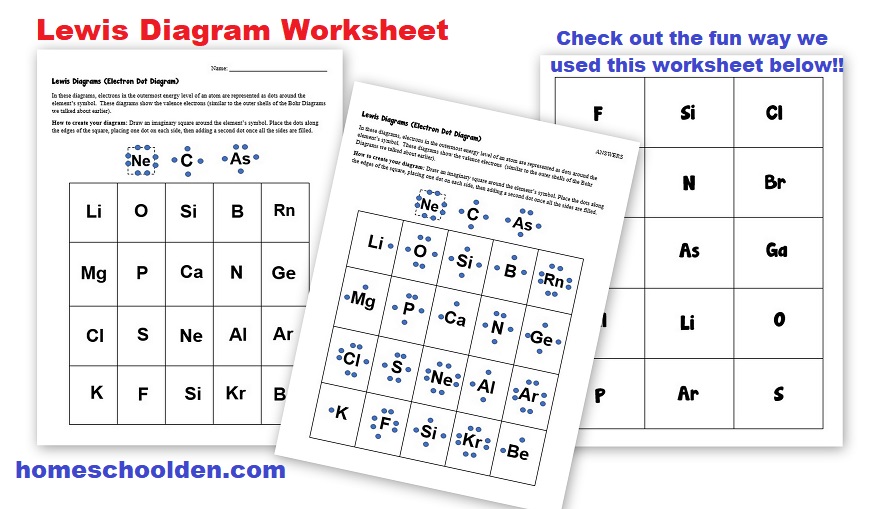
Beyond the Worksheet: Culture, Fun Facts, and Your Daily Dose of Chemistry
The beauty of chemistry, even something as seemingly technical as Lewis Dot Structures, is that it’s woven into the fabric of our lives. You see these molecular arrangements everywhere!
The Pop Culture Connection: Hollywood's Take on Molecules
Ever watched a science fiction movie where they’re manipulating molecules? While the visuals are often dramatized, the underlying concept of atoms bonding and interacting is rooted in these principles. Think about the vibrant colors of fireworks – those are due to the electrons in metal atoms absorbing and emitting energy, a direct consequence of their electron configurations and bonding. It’s like the ultimate light show, powered by chemistry!
Fun Fact Fiesta: The Ubiquitous Water Molecule
Let’s revisit water (H₂O). Its bent shape, dictated by its Lewis Dot Structure and VSEPR theory (which we’ll save for another day!), is what gives it its unique properties, like its ability to dissolve so many substances. It’s the universal solvent, the lifeblood of our planet, and it all starts with those simple hydrogen and oxygen atoms and their electron sharing. It's responsible for everything from your morning coffee to the vast oceans!
The Smell of Science: Odor Molecules
Ever wondered why a rose smells like a rose and a skunk smells… well, like a skunk? It all comes down to the shape and electron distribution of the molecules involved. The specific arrangement of atoms and their electrons determines how these molecules interact with the scent receptors in our noses. So, that lovely perfume? A complex ballet of Lewis Dot Structures!

Your Kitchen Chemistry: Baking Soda and Vinegar
That classic baking soda and vinegar volcano experiment? It’s a fantastic demonstration of acid-base reactions, all explained by Lewis Dot Structures. Baking soda (sodium bicarbonate) and vinegar (acetic acid) react to produce carbon dioxide gas, water, and sodium acetate. The release of gas is a direct result of the breaking and forming of chemical bonds, which we can understand by looking at the structures of the reactants.
A Moment of Reflection: The Art of Connection
As you work through your Lewis Dot Structures and consult those all-important answer keys, take a moment to appreciate the bigger picture. These tiny, invisible arrangements of electrons are the architects of our physical world. They dictate everything from the sturdiness of a bridge to the sweetness of a fruit.
Life, much like chemistry, is about connection. It’s about how different parts come together to form something new, something greater than the sum of its parts. Our relationships, our communities, even the way we organize our thoughts – these are all about forming bonds, sharing energy, and striving for a kind of stability. So, the next time you’re wrestling with a particularly tricky Lewis Dot Structure, remember that you’re not just doing a worksheet. You’re exploring the fundamental principles that hold the universe together, one electron dot at a time. And that, my friends, is pretty darn cool.
So, go forth, conquer those worksheets, and embrace the delightful, intricate dance of molecules. Your understanding of the world, and perhaps even your own life, might just become a little bit more beautifully structured.
