Chemical Equation That Illustrates The Autoionization Of Water
Ever wondered what makes water, well, water? It’s more than just a clear, life-giving liquid. Deep down, even pure water is a hive of tiny, unseen activity, a constant dance of molecules that is absolutely fundamental to so much of what we experience. Think of it like a bustling city where everyone is a bit of a shapeshifter, constantly changing roles. This fascinating process, known as the autoionization of water, is surprisingly simple at its core, yet its implications are colossal. It’s the secret ingredient that allows acids to be acidic and bases to be basic, and understanding it unlocks a whole new appreciation for the chemistry all around us. Plus, who doesn't love a little bit of molecular magic?
At its heart, the autoionization of water is like a tiny, self-contained chemical reaction happening within any sample of water, whether it's a drop from your tap or the vast ocean. It’s a process where water molecules are constantly breaking apart and reforming, but in a very specific way. Imagine two water molecules bumping into each other. It’s not a violent collision, but more of a friendly exchange. One water molecule, acting like a gracious host, generously gives away one of its hydrogen atoms (which, remember, is just a single proton) to its neighbor. This seemingly small act has big consequences for both molecules involved.
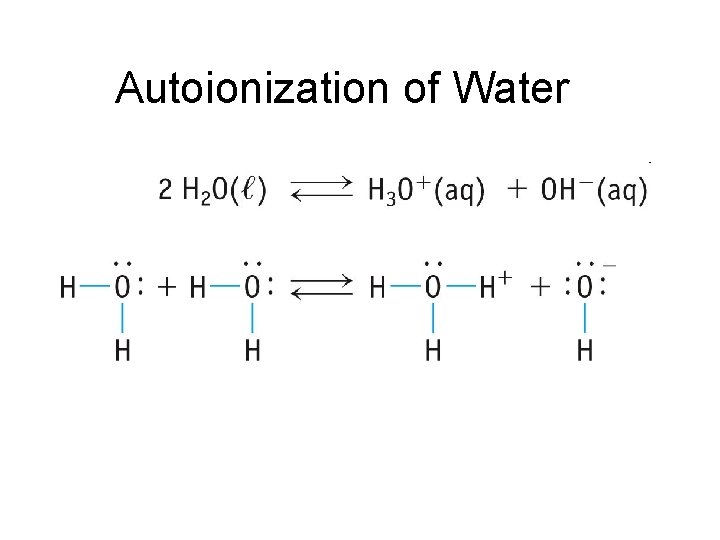
So, what’s the actual chemical equation that captures this molecular ballet? It's elegantly simple:
2H₂O ⇌ H₃O⁺ + OH⁻
Let’s break down this cryptic-looking formula. On the left side, we have 2H₂O. This simply represents two molecules of water. The little ‘2’ means we're talking about two of them. Now, the double arrow (⇌) is super important. It’s not a one-way street; it’s a two-way highway. This signifies that the reaction can go forward (water molecules breaking apart) and backward (the resulting ions reforming into water). This constant back-and-forth is what we mean by a state of equilibrium.
When those two water molecules perform their little dance, one gives up a hydrogen ion (H⁺). This lone hydrogen ion doesn’t like to be by itself for long in water. It’s a bit of a social butterfly and immediately attaches itself to another, intact water molecule. This creates a new species called the hydronium ion, represented by H₃O⁺. Think of it as a water molecule that has enthusiastically adopted an extra hydrogen proton. It's like a single scoop of ice cream getting a cherry on top!

What about the water molecule that lost the hydrogen ion? It's left with one hydrogen atom less, and crucially, an imbalance of charge. It becomes what we call the hydroxide ion, written as OH⁻. It’s basically a water molecule minus a proton. So, in this whole process, a single water molecule has transformed into two charged particles: the positively charged hydronium ion and the negatively charged hydroxide ion.
The beauty and utility of this equation lie in its ability to explain the fundamental properties of all aqueous solutions. Even in the purest water imaginable, there’s always a tiny concentration of both H₃O⁺ and OH⁻ ions. These two ions are the architects of acidity and alkalinity. When you add an acid to water, you are essentially increasing the concentration of H₃O⁺ ions. Conversely, when you add a base, you are increasing the concentration of OH⁻ ions (often by releasing them directly or by reacting with H₃O⁺ to form more water).

The balance between these two ions is what determines the pH of a solution, a scale that tells us how acidic or basic something is. For pure water at room temperature, the concentrations of H₃O⁺ and OH⁻ are equal, and this perfect balance gives it a neutral pH of 7. This constant, albeit small, generation of ions is the reason why even neutral water can conduct electricity, albeit very poorly compared to saltwater.
Understanding the autoionization of water is not just an academic exercise. It’s the bedrock of countless chemical processes. From the digestion of food in our bodies to industrial chemical reactions, the behavior of acids and bases, and even the functioning of batteries, the interplay of H₃O⁺ and OH⁻ ions is crucial. It’s a testament to how a simple, spontaneous molecular event can have profound and far-reaching impacts on the world around us. So, the next time you take a sip of water, remember the tiny, bustling chemical dance happening within, governed by that wonderfully revealing equation: 2H₂O ⇌ H₃O⁺ + OH⁻.
