Chemical Equation For Self Ionization Of Water

Ever wondered why plain old water, the stuff you chug on a hot day or use to brew your morning coffee, isn't quite as simple as it seems? It turns out, water's got a secret life, a tiny dance it does with itself that's super important for, well, pretty much everything around us. We're talking about the self-ionization of water, and don't let the fancy name scare you! It's actually a pretty cool and surprisingly relevant phenomenon.
Think of it like this: imagine you're at a huge party. Most people are just mingling, enjoying themselves. But every now and then, two people will break away from their groups, have a quick, intense chat, and then rejoin the crowd, maybe a little changed. That's kind of what water molecules do. They're usually just hanging out, H₂O, you know, two hydrogens and an oxygen. But sometimes, poof! one water molecule will lend a hydrogen atom to its neighbor.
So, what's the chemical equation for this little water-party trick? It's usually written like this:
H₂O + H₂O ⇌ H₃O⁺ + OH⁻
Let's break that down without needing a chemistry degree. The H₂O on the left is just a regular water molecule. When two of them meet, like two pals at that party, something interesting happens. One water molecule (let's call it Water A) is feeling generous and hands over one of its hydrogen atoms (which is basically just a proton, a positively charged particle) to the other water molecule (Water B).
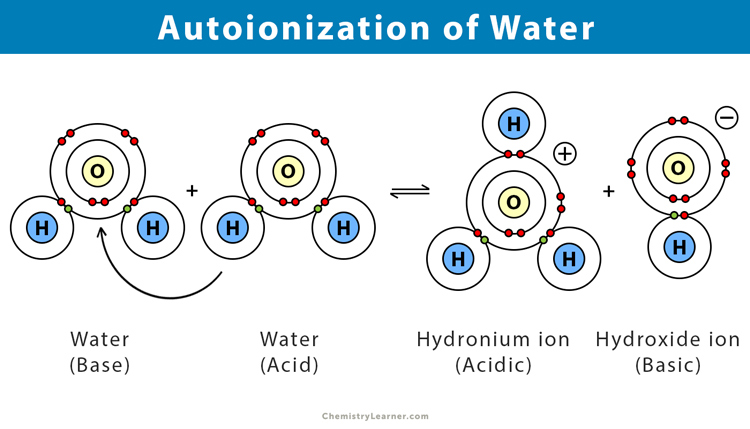
Now, Water A, having lost a hydrogen, becomes something called a hydroxide ion, which has a negative charge. We write this as OH⁻. Think of it as the water molecule that got a little lighter and a bit grumpy. On the other hand, Water B, who happily accepted that extra hydrogen, becomes a hydronium ion. It now has an extra proton, making it positively charged, and we write this as H₃O⁺. It's like the water molecule that got a bit heavier and is now feeling a little energized.
The little arrow pointing both ways (⇌) is super important. It means this isn't a one-way street. The hydronium and hydroxide ions can get back together to form water again. It’s a constant back-and-forth, a delicate balance. It's like those two partygoers can hug it out and become regular water molecules again.
Why Should You Care About This Tiny Water Dance?
Okay, so you're thinking, "This is neat, but how does it affect my life?" Well, prepare to be amazed! This little ionization dance is the foundation for something we talk about all the time: pH. You see that on everything from shampoo bottles to swimming pool testers. pH tells us if something is acidic, neutral, or basic (alkaline).
Remember those H₃O⁺ (hydronium) and OH⁻ (hydroxide) ions? The balance between these two is what determines the pH. In pure water, the self-ionization happens in a way that creates an equal number of hydronium and hydroxide ions. This perfect balance gives us a neutral pH of 7. It's like the ultimate, well-behaved party where everyone is equally happy.
But what happens when you add something to water? That's where things get interesting. If you add something acidic, like lemon juice or vinegar, it encourages the formation of more hydronium ions. Think of it as inviting a few extra energetic dancers to the party. This makes the water more acidic. The pH drops!
Conversely, if you add something basic, like baking soda or soap, it tends to soak up those extra hydrogen protons, meaning more hydroxide ions are left hanging around. It's like some guests are taking a break, leaving others to enjoy the spotlight. This makes the water more basic, and the pH rises!
So, that slightly tingly feeling you get from washing your hands with soap? That's the basic nature of the soap, influenced by its interaction with water's ionization. That sour taste of a pickle? That's the acidity, also related to how added substances affect water's ionization.
A Tiny Balance with Big Consequences
This seemingly small reaction has massive implications. Think about the oceans. They have a delicate pH balance that supports all marine life. If the self-ionization of water, and the subsequent addition of things like CO₂ from the atmosphere (which makes water slightly acidic), were to get out of whack, entire ecosystems could suffer. It’s like a finely tuned orchestra – if one section plays too loud or too soft, the whole performance is off.

Even our own bodies rely on this! Your blood, for instance, has a very tightly controlled pH. If it swings too far in either direction, it can be really dangerous. This stability is maintained, in part, by chemical buffers that work by interacting with those very hydronium and hydroxide ions that pop into existence from water's self-ionization.
Think of it like trying to keep your home at a comfortable temperature. You have a thermostat that kicks in the heater or air conditioner to keep things just right. Water's self-ionization is like a tiny, constant internal thermostat for everything that involves water. It's always there, always doing its thing, ensuring a baseline that allows other things to happen.
So, Next Time You Sip Your Water…
Next time you take a sip of that clear, life-giving liquid, remember the incredible, dynamic dance happening within it. Those H₂O molecules aren't just passive bystanders; they're actively, albeit subtly, participating in a chemical ballet that makes our world work. It’s a reminder that even the simplest things can hold incredible complexity and importance. It’s this tiny, hidden reaction that makes water such a versatile and essential player in pretty much every chemical process we encounter, from the mild tang of your orange juice to the very lifeblood flowing through your veins. Pretty neat, right?
