Chemical Equation For Acetic Acid And Sodium Hydroxide

Ever wondered what happens when you mix a bit of that zingy stuff in your salad dressing with the stuff that cleans your oven? Well, buckle up, buttercups, because we're about to dive into a chemical party that’s way more exciting than your average Tuesday! Today, we're talking about the dynamic duo: acetic acid and sodium hydroxide.
Think of acetic acid as your super-smart, slightly sour friend. You know, the one who’s always telling you useful facts, like how vinegar (which is basically dilute acetic acid) can make your fries taste amazing or how it can tackle those stubborn water spots on your shower door. It’s got this energetic vibe, always ready to react and make things happen. It’s the kind of molecule that’s not afraid to get its hands dirty – or in this case, its hydrogens!
And then there’s sodium hydroxide. Oh, sodium hydroxide! This one’s the powerful, no-nonsense powerhouse of the chemical world. Think of it as the ultimate organizer. It’s that friend who swoops in and tidies everything up, smoothing out wrinkles and neutralizing any chaos. In the kitchen, it’s a key ingredient in making pretzels wonderfully chewy and golden brown. Out in the world, it’s a superhero for cleaning and manufacturing, but it’s also a bit… intense. It’s super alkaline, meaning it’s the opposite of acidic, and it can be quite a handful if you’re not careful. It’s the friend who’s got a heart of gold but needs a good, firm handshake to get things done.
So, what happens when these two seemingly different characters meet? It’s like a cosmic matchmaking made in a laboratory! They bump into each other, and poof! A beautiful transformation occurs. It’s not a dramatic explosion or anything that’ll send your cat running for cover. It’s more like a gentle, yet profound, symphony of neutralization.
Here’s the magic in its simplest form. We can represent acetic acid with a little chemical shorthand: CH₃COOH. See that H at the end? That’s the "acidic" bit, the part that’s ready to be shared. And our friend sodium hydroxide? Its shorthand is NaOH. That little OH group is like its business end, ready to mingle.

When these two get together, the CH₃COOH says, "Hey, NaOH, you look like you could use a bit of this hydrogen I’ve got here!" And the NaOH, being the gracious (and powerful) substance it is, says, "Why, thank you, CH₃COOH! I’ll take it!" So, the H from the acetic acid hops over to the OH from the sodium hydroxide. What do you get when you put an H and an OH together? You guessed it! A happy little molecule of water (H₂O). How’s that for a friendly exchange?
But wait, there’s more! When the acetic acid lets go of its hydrogen, it becomes something called the acetate ion (CH₃COO⁻). It’s still got that familiar tang, but now it’s looking for a new friend to balance it out. And guess who’s more than happy to step in? The sodium ion from the sodium hydroxide! So, the sodium ion (Na⁺) and the acetate ion (CH₃COO⁻) decide to become best buddies, forming a brand new compound called sodium acetate (CH₃COONa).
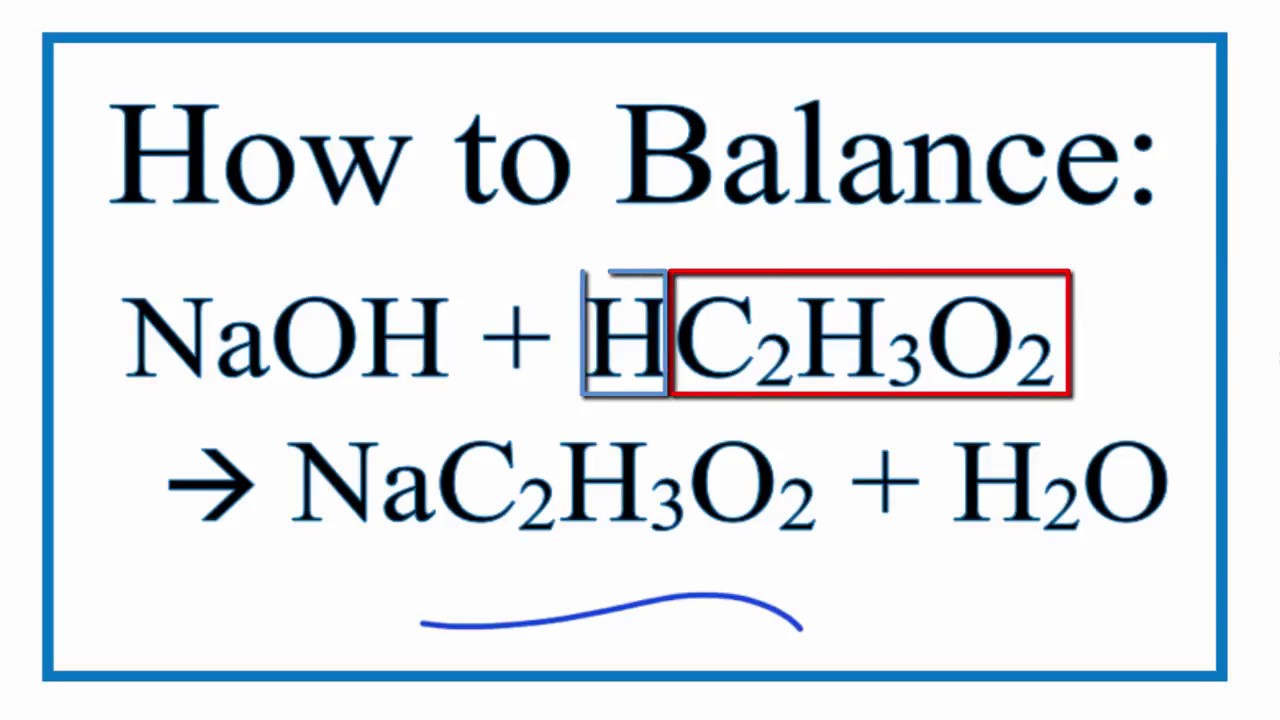
So, the grand chemical equation, the recipe for this amazing transformation, looks like this:
CH₃COOH + NaOH → CH₃COONa + H₂O
Isn’t that just the neatest thing? It’s like a perfect little chemical handshake. You start with something sour and something strongly alkaline, and you end up with something pleasant and, well, neutral, plus a splash of good old water. It’s a reaction that’s not just scientifically fascinating, but also incredibly useful. It’s the reason why sometimes when you have a bit of an upset stomach from too much spicy food, a doctor might suggest a liquid antacid that contains this very reaction. They’re literally neutralizing that excess stomach acid with something like this!

Think about it: a touch of vinegar (acetic acid) and a bit of baking soda (which when mixed with water can create a bit of sodium hydroxide effect, though it's more complex) can create that fizzy eruption you might have seen in science fairs. That fizz is a sign of the gases being released as these compounds react and find their happy, neutral state. It’s like the chemicals themselves are cheering about their successful partnership!
This reaction is a fundamental concept in chemistry, but it’s also a reminder of how interconnected and adaptable the world around us is. Even the seemingly simple act of mixing two substances can lead to a beautiful, balanced outcome. So, the next time you’re enjoying a salad with a vinaigrette, or perhaps watching a science experiment that involves fizzing, remember the unsung heroes: acetic acid and sodium hydroxide, throwing a little chemical party and making the world a bit more balanced, one molecule at a time!
