Chapter 8 Covalent Bonding Chapter Test B Answer Key

Alright, imagine you've just survived the thrilling, sometimes perplexing, adventure that is Chapter 8: Covalent Bonding. You’ve grappled with sharing electrons, wrestled with molecular shapes, and maybe even dreamt in Lewis structures. Now, you’ve bravely tackled the Chapter Test B, and the ultimate moment of truth has arrived: the answer key! It’s like the treasure map to figuring out if your valiant efforts paid off.
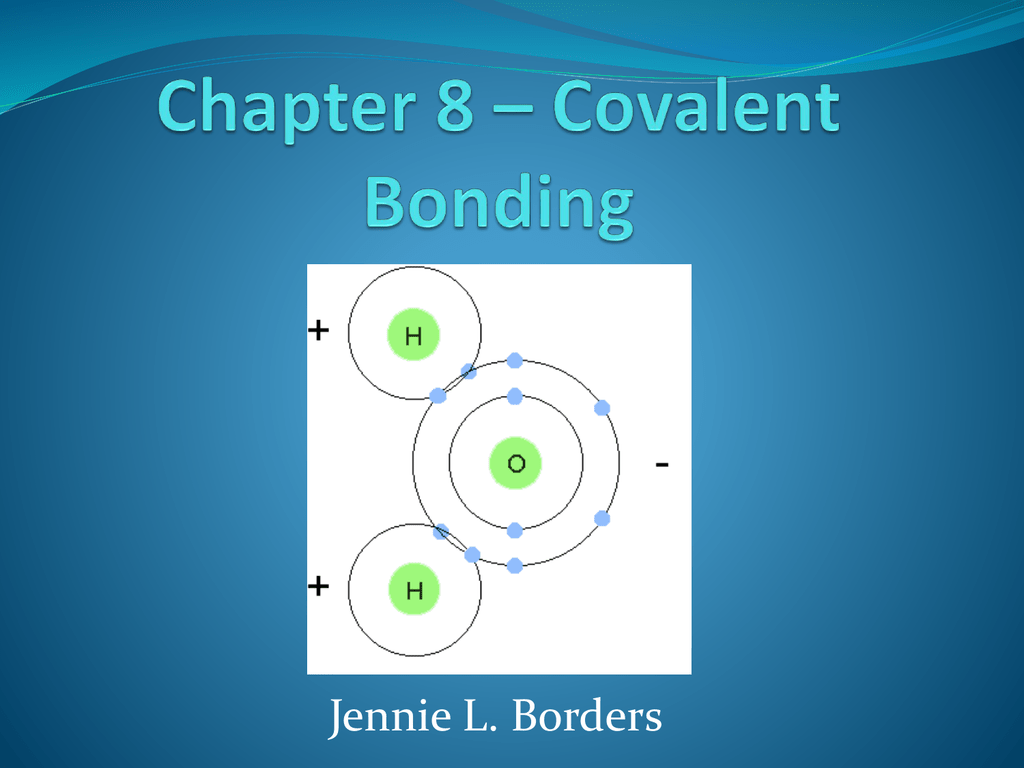
Think of this answer key not as a stern judge, but as your trusty sidekick, whispering the secrets of the universe of covalent bonds. Did you nail that question about water (H₂O) and its wacky, bent shape? That's right, it’s not a perfectly straight line, and the key will confirm your brilliant deduction! It's a little like finding out your favorite cartoon character has a hidden talent – utterly delightful and makes perfect sense once you see it.
Remember that tricky section on electronegativity? That’s just science’s way of talking about how much atoms fancy holding onto those shared electrons. Some atoms are like greedy toddlers with a cookie, pulling it closer. Others are more generous sharers. The answer key will tell you if you correctly identified the electron-hogging champions and the more laid-back sharers. It’s a subtle dance, and understanding it is like finally cracking the code to why some people are naturally better at sharing their snacks.
And what about those polyatomic ions? They’re like little chemical families, a group of atoms sticking together and carrying a charge. The answer key will guide you through them, confirming if you recognized that the sulfate ion (SO₄²⁻) is a strong, stable unit. It’s heartwarming, really, seeing how these tiny entities form bonds and stick together, much like our own families.
Let’s talk about some of the more visual aspects. The answer key will probably give you a nod if you correctly sketched out the VSEPR theory. This is where things get a bit like playing with molecular LEGOs. You’re figuring out the 3D shape of molecules based on how many electron groups are around the central atom. Did you correctly predict that a molecule with four electron groups would aim for a tetrahedral shape, like a perfectly balanced pyramid? High five! The answer key is your digital high-five, confirming your mastery of molecular architecture.
Sometimes, these tests throw in a curveball, asking about exceptions or special cases. The answer key is there to gently point out those nuances. It’s like when you’re learning a new language and discover a silly idiom that makes no literal sense but is used all the time. The answer key helps you understand those "idioms" of covalent bonding.
Consider the humble molecule of methane (CH₄). It’s the basis of natural gas, and it has a beautiful, symmetrical tetrahedral shape. If you got that one right, the answer key is basically saying, "You’ve got this chemistry thing down pat!" It's a simple molecule, but its stability is crucial to so many things we take for granted.

And then there are the cases where bonds are polar. This is where the electron sharing is a little uneven. Think of it like a tug-of-war where one side is slightly stronger. The answer key will reveal if you correctly identified which atoms are pulling harder, creating a slight electrical imbalance within the molecule. It’s these subtle differences that lead to some of the most fascinating chemical behaviors. For instance, that uneven pull in water molecules is what allows them to stick together so well, creating surface tension and enabling life as we know it. The answer key is validating your understanding of these fundamental forces.
So, as you pore over that Chapter 8 Covalent Bonding Chapter Test B Answer Key, don’t just see it as a list of right and wrong answers. See it as a narrative. It’s the story of how atoms, these tiny building blocks, come together, share, and create the vast diversity of substances that make up our world. It’s about understanding the invisible forces that hold everything together, from the air you breathe to the food you eat.
Each correct answer is a little victory, a confirmation that you’ve successfully navigated the intricate world of electron sharing. And if there are a few misses? That’s okay! The answer key is also a guide for learning. It’s not about perfection; it’s about understanding. It’s about appreciating the elegance and sometimes surprising simplicity of how chemistry works. So, embrace the answer key, learn from it, and remember the amazing power of covalent bonds that you've begun to unravel.
Think of the answer key as your chemistry superhero decoder ring!
It helps you translate the complex language of atoms into understandable wins. You’ve done the work, you’ve faced the challenge, and now, with the answer key, you get to see the fruits of your intellectual labor. Whether you aced it or learned where to focus next, the journey through covalent bonding is a fascinating one, and the answer key is your trusty compass.
