Chapter 4 Review Arrangement Of Electrons In Atoms

Hey there, science adventurer! Ready to dive back into the wonderfully weird world of atoms? We've been on quite a journey, haven't we? From the basic building blocks of everything to understanding how they're put together, it's been a blast. Today, we're going to have a super chill, super fun review of Chapter 4: The Arrangement of Electrons in Atoms. Think of it as a victory lap around the electron cloud, no lab coats required!
Remember when we first learned about atoms? Like, they're tiny, they make up everything, and they have this nucleus in the middle? Well, Chapter 4 decided to really zoom in on what's happening outside that nucleus. It's all about those zippy little guys called electrons. They're like the hyperactive toddlers of the atom world, always on the move!
So, what's the big deal with electron arrangement? Why should we even care where these little troublemakers hang out? Well, my friend, it's actually a huge deal! The way electrons are arranged is basically the secret sauce that determines how an atom behaves. It dictates how it bonds with other atoms, what kind of properties it has (like being a shiny metal or a squishy gas), and pretty much everything that makes one element different from another. It's like knowing the seating chart at a party – it tells you who's going to mingle with whom!
The Early Days: Bohr's Little Circles
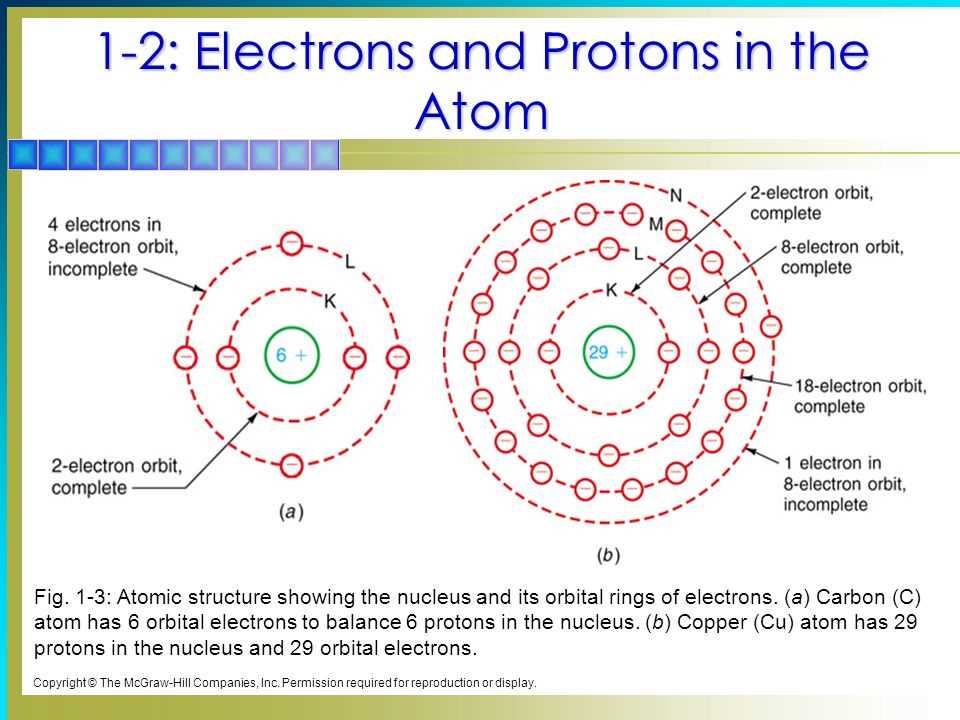
Let's rewind a bit to the good ol' days of atomic theory. We had Niels Bohr, a super smart dude, who proposed a pretty neat model. He thought electrons zipped around the nucleus in nice, orderly, circular paths. Like little planets orbiting the sun. Remember those diagrams? They were so clean and symmetrical. You could practically color them in with crayons!
Bohr's model introduced the idea of energy levels. He said electrons could only hang out in specific "shells" or orbits, and each shell had a different energy. Electrons in shells closer to the nucleus had lower energy, and those further out had higher energy. It was like a ladder – you can stand on a rung, but you can't float in between. This was a huge leap forward because it explained a lot about why atoms emit specific colors of light when they get excited. Think of it like a happy atom showing off its favorite colors!
But, as science often does, we kept learning more. Bohr's model was great for simpler atoms, like hydrogen. But when we started looking at more complex atoms, things got a little… messy. It turned out electrons aren't always so predictable. They don't just follow perfect circles. Sometimes they're here, sometimes they're there. It's like trying to catch a squirrel in your backyard – you know it's somewhere, but pinning down its exact path is a whole other story!
Enter the Quantum Mechanical Model: It's Not Just Circles Anymore!
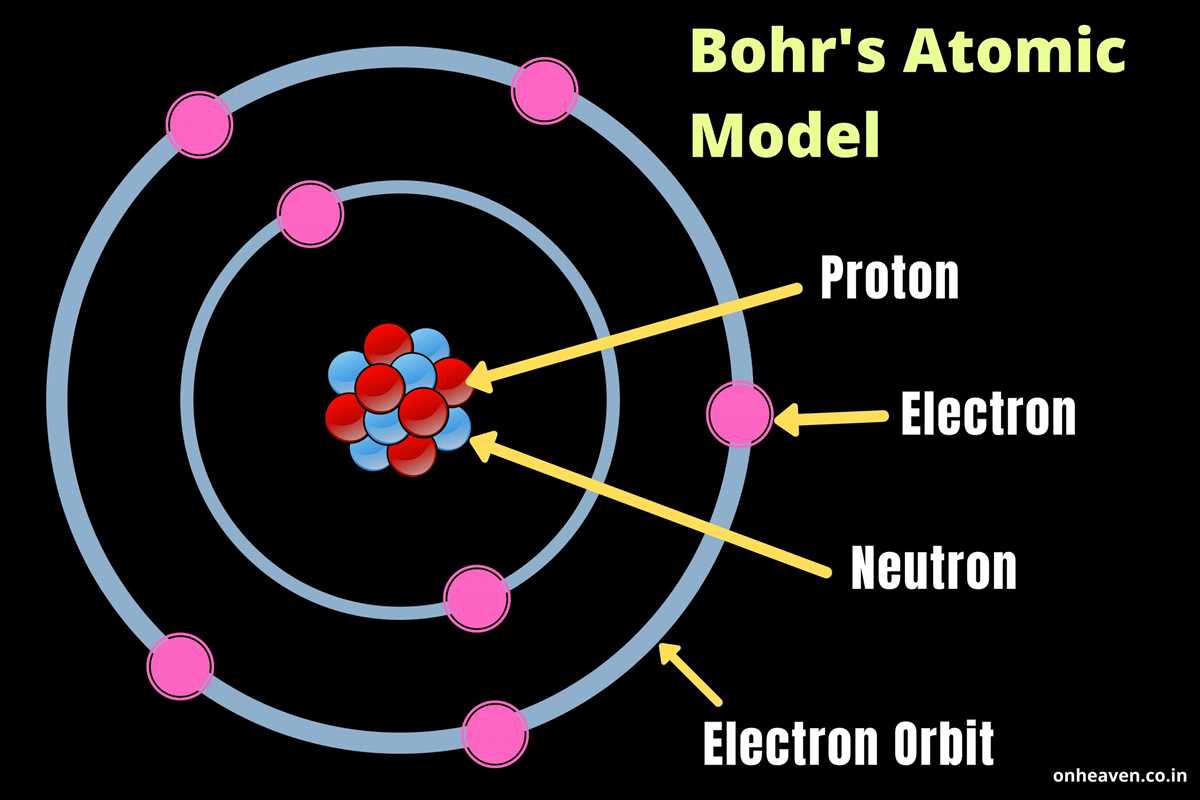
This is where things get really interesting and, I'll admit, a little mind-bending. We moved from Bohr's neat orbits to the quantum mechanical model. This bad boy, developed by a bunch of brilliant minds like Schrödinger and Heisenberg, says electrons don't have fixed paths. Instead, they exist in regions of space called orbitals. Think of an orbital not as a racetrack, but more like a cloud of probability. It’s where we're most likely to find an electron.
So, instead of saying "the electron is here," we say "there's a high probability of finding the electron in this region." It's like saying, "I'm probably going to be on the couch watching TV tonight," rather than "I will be at precisely coordinates X, Y, and Z on the couch." It’s a bit more fuzzy, a bit more… quantum-y.
Orbitals: The Electron Neighborhoods
Now, these orbitals aren't all the same shape. They come in different flavors, and each flavor has a specific name and shape. We've got:
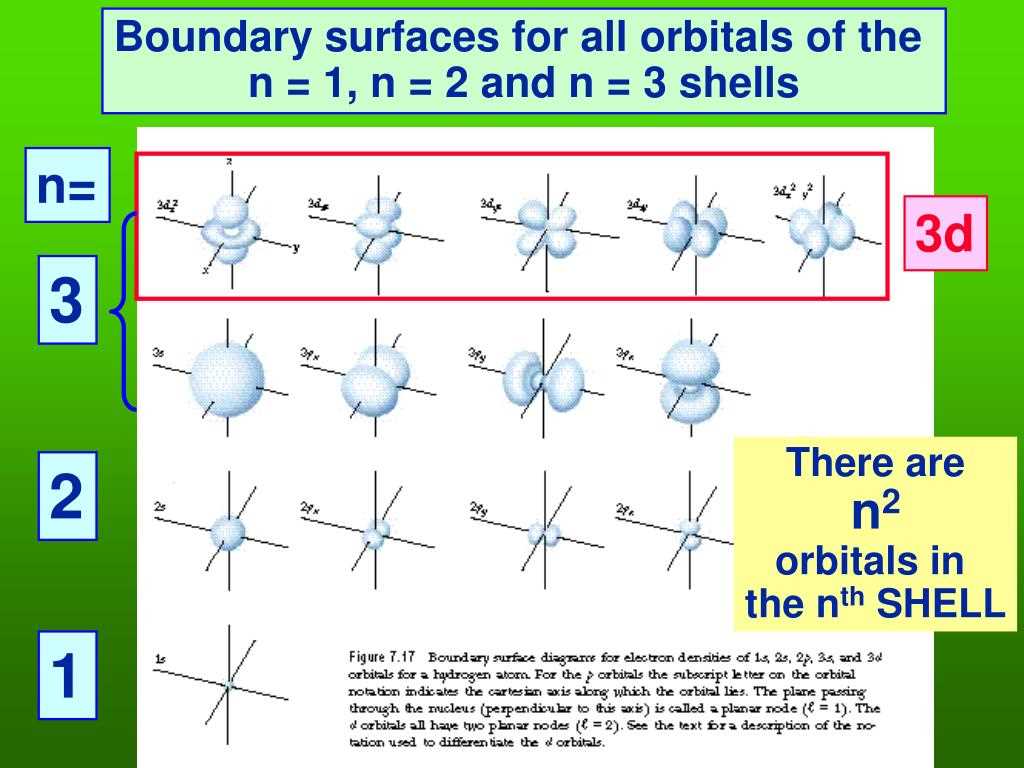
- s orbitals: These are the simplest. They're spherical, like a perfectly round ball. Imagine a little bubble around the nucleus. There's one s orbital for each energy level.
- p orbitals: These are shaped like dumbbells, with two lobes on either side of the nucleus. They're like two balloons tied together at the center. And get this – there are three p orbitals for each energy level, oriented along the x, y, and z axes. So, they're pointing in different directions, ready to interact with other atoms in all sorts of ways.
- d orbitals: These get a bit more complicated, with shapes that look like cloverleafs (mostly!). There are five d orbitals for each energy level, starting from the third energy level. They're like the fancy neighborhoods with unique architecture.
- f orbitals: And then we have the f orbitals, which are even more complex and usually found in the very heavy elements. There are seven f orbitals for each energy level, starting from the fourth energy level. These are the exclusive penthouses of the electron world!
It's like different types of apartments in a building. The s orbitals are like studio apartments, the p orbitals are like one-bedroom apartments, and so on. Each has its own vibe and capacity!
The Rules of the House: How Electrons Fill Up
So, we have these orbitals, and we have electrons that need a place to crash. But how do they decide who gets which orbital? Well, nature has some pretty specific rules, and understanding them is key to understanding electron arrangement. Think of it like filling up a hotel with guests – there are certain policies in place!
The Aufbau Principle: Building Up from the Bottom
First up is the Aufbau principle. This is like the hotel manager saying, "Okay folks, we fill up the lowest energy levels and orbitals first." Electrons will always try to occupy the lowest energy orbital that is available. It's more energy-efficient, just like it's easier to walk downhill than uphill. So, they start at the bottom and work their way up, filling up the s orbitals before moving to p, then d, and so on.
This is why the periodic table is arranged the way it is! The order of filling orbitals follows a specific pattern, and understanding this pattern is like having a cheat code for predicting electron configurations.
The Pauli Exclusion Principle: No Roommates Allowed (Unless You're Different!)
Next, we have the Pauli exclusion principle. This one is super important. It basically says that within a single orbital, you can only have a maximum of two electrons. And here's the kicker: if you have two electrons in the same orbital, they must have opposite spins. Think of it like a cozy hotel room – only two people allowed, and they have to agree to disagree on something, like which way the pillow goes.
Electrons have a property called "spin," which is kind of like them spinning on an axis, either clockwise or counterclockwise. We represent these spins with arrows, usually pointing up or down. So, if you have two electrons in the same orbital, one arrow goes up, and the other goes down. They're in the same space, but they're doing their own thing in opposite directions. It's like a polite disagreement that keeps the peace!
Hund's Rule: Spread Out First!
Finally, we have Hund's rule. This rule comes into play when we have multiple orbitals of the same energy level available, like the three p orbitals or the five d orbitals. Hund's rule says that electrons will first occupy each of these orbitals singly before they start pairing up. It's like people at a new hotel wanting their own room before they have to share.
So, if you have three electrons and three p orbitals, each electron will go into its own p orbital. Only when you have a fourth electron will it start pairing up with one of the first three. This minimizes electron-electron repulsion and keeps things a bit more comfortable for the electrons. They're not exactly fans of sharing tiny spaces unless they absolutely have to!
Electron Configurations: The Atom's Address Book
Putting all these rules together allows us to write an electron configuration. This is like an atom's official address, telling us exactly how many electrons are in each energy level and in each type of orbital. It's a shorthand way of describing the electron arrangement.
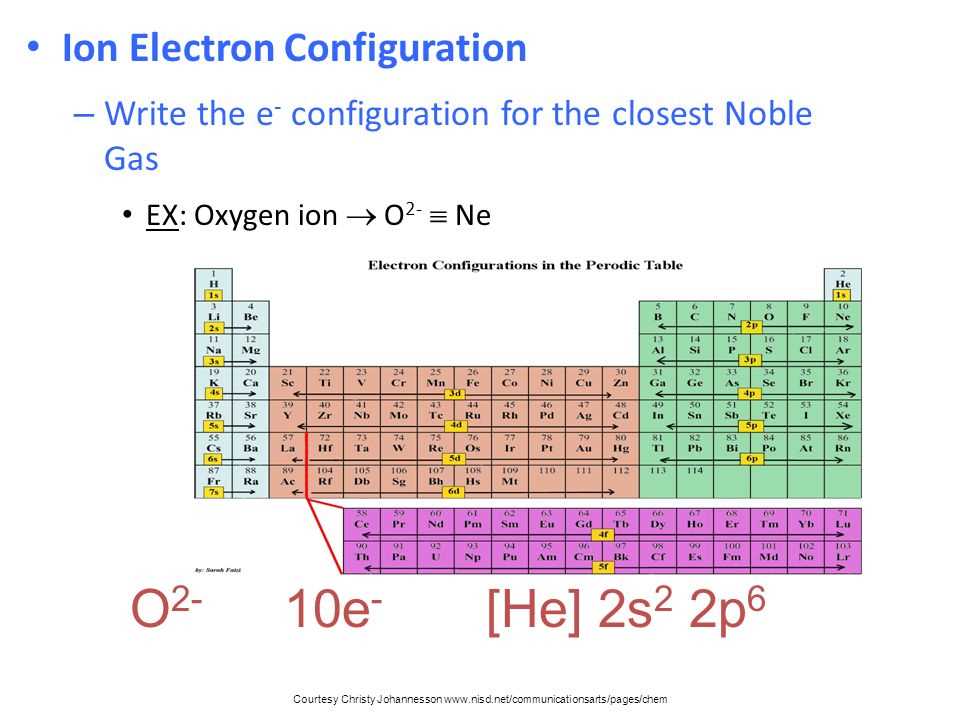
For example, oxygen (atomic number 8) has 8 electrons. Following the rules, its electron configuration would be 1s²2s²2p⁴. What does this mean?
- The 1s² tells us that the first energy level (n=1) has one s orbital (s), and it's filled with two electrons (²).
- The 2s² tells us that the second energy level (n=2) has one s orbital (s), filled with two electrons (²).
- The 2p⁴ tells us that the second energy level (n=2) also has p orbitals (p), and there are four electrons in total occupying these p orbitals (⁴). Remember, the p subshell can hold up to 6 electrons (2 in each of the 3 p orbitals).
It might seem a bit like deciphering a secret code at first, but once you get the hang of it, it’s super rewarding! It’s like learning a new language, and the language of atoms is pretty darn cool.
Valence Electrons: The Party Animals!
Now, out of all the electrons an atom has, there's a special group that gets all the attention when it comes to bonding: the valence electrons. These are the electrons in the outermost energy level. They're the ones that are furthest from the nucleus and, therefore, the easiest to interact with other atoms.
Think of them as the outgoing, social butterflies of the atom. They're the ones that initiate friendships (chemical bonds!) with other atoms. The number of valence electrons an atom has is a huge predictor of its chemical behavior. Elements with similar numbers of valence electrons tend to have similar chemical properties, which is why they're grouped together in the periodic table. It’s like finding your clique at school!
For oxygen, with the configuration 1s²2s²2p⁴, the outermost energy level is the second energy level (n=2). So, it has 2 electrons in the 2s orbital and 4 electrons in the 2p orbitals, for a total of 6 valence electrons. That's a lot of potential for bonding!
Why All This Matters: The Big Picture
So, why did we just spend all this time talking about electron clouds and quantum rules? Because understanding how electrons are arranged is the absolute foundation for understanding chemistry. It explains:
- How elements form bonds: Why does sodium (a metal) love to give away an electron to chlorine (a nonmetal) to form salt (NaCl)? It's all about achieving a stable electron configuration!
- The properties of matter: Why is diamond hard and graphite soft? It comes down to how their atoms' electrons are arranged and how they interact.
- Chemical reactions: Every chemical reaction, from baking a cake to photosynthesis, involves the rearrangement of electrons.
It’s like understanding the blueprint of a house. You can’t really understand how the plumbing works, or why the electricity flows, without knowing where all the wires and pipes are supposed to be.
And here's the really exciting part: every time you look at the periodic table, you're looking at a direct reflection of these electron arrangements. The columns (groups) represent elements with the same number of valence electrons, and the rows (periods) represent the highest energy level occupied by electrons. It's a beautifully organized system, all thanks to the quirky behavior of electrons!
So, don't let the fancy terms like "quantum mechanical model" or "orbitals" scare you off. At its heart, it's all about understanding where those little electron dynamos are and how they like to mingle. It’s a fascinating puzzle, and you’ve just learned how to put a big piece of it together!
You've navigated the world of energy levels, orbital shapes, and electron-filling rules. You’ve got this! The universe is made of these tiny, energetic dancers, and now you know their basic choreography. Keep exploring, keep questioning, and remember that every bit of knowledge you gain about the atom is like discovering a new, tiny galaxy. And that, my friend, is pretty darn awesome. Go forth and be scientifically brilliant!
