Chapter 4 Arrangement Of Electrons In Atoms Answer Key
Ever felt like your sock drawer is a chaotic mess, or your bookshelf is a jumble of stories waiting to be discovered? Well, imagine the universe's tiniest residents, the electrons, trying to find their perfect spot. That's essentially what Chapter 4, "Arrangement of Electrons in Atoms," is all about. It's like a cosmic game of musical chairs, but with incredibly important rules!
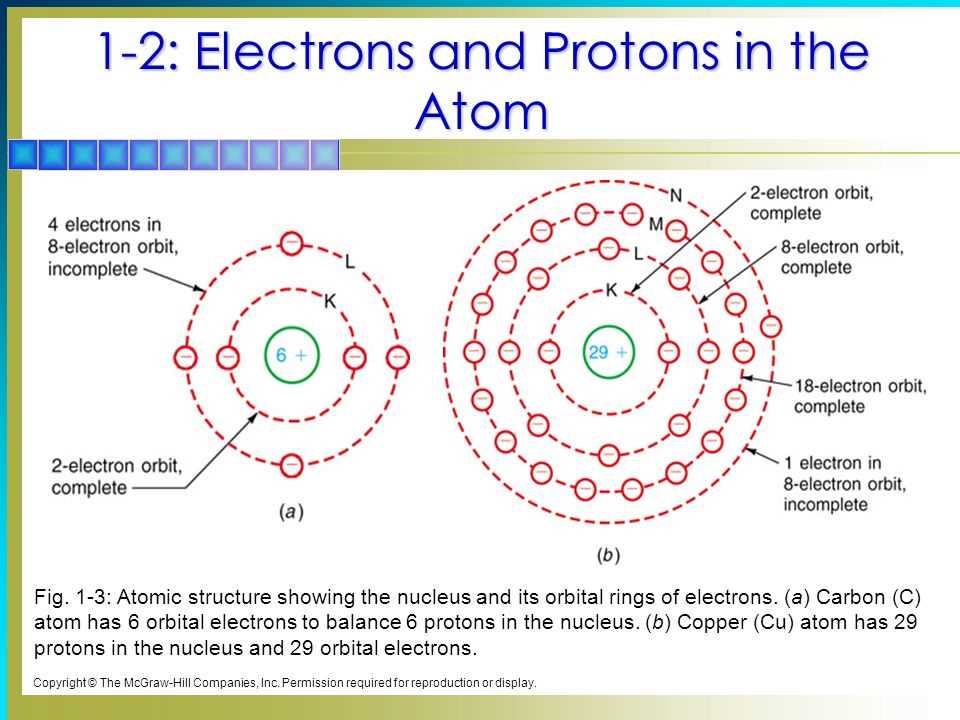
Think of an atom as a miniature solar system, with a grumpy nucleus (that's the core, where the protons and neutrons hang out) at the center. And then you have the electrons, these zippy little particles, whizzing around. But they don't just zoom anywhere! Oh no, these electrons are quite particular. They have preferred neighborhoods, like different floors in a high-rise apartment building, and specific rooms within those floors.
These "floors" are called energy levels. The closer you are to the nucleus, the less energy you need to hang out there. It's like being on the ground floor of a building – less effort to get to the lobby! As you go up to higher energy levels, the electrons need more "oomph" to stay there. They're like teenagers needing extra snacks and caffeine to stay awake!
Now, within each energy level, there are different types of "apartments," called orbitals. These are like cozy little spaces where electrons love to chill. Each orbital can only hold a maximum of two electrons, and they're a bit like bickering siblings – if they're in the same orbital, they have to have opposite "spins." Imagine two identical twins trying to share a bunk bed; they need to figure out their own personal space to avoid constant squabbles!
This whole arrangement isn't random. There are clever rules that dictate where every single electron gets to park itself. One of the most famous is the Aufbau principle, which, in plain English, means electrons fill up the lowest energy orbitals first. They're like bargain hunters, always looking for the best deal on a spot!

Then there's Hund's rule. Imagine you're in a communal living space, and there are three empty beanbag chairs in a room. Most people would rather have their own chair than share one, right? Hund's rule says that electrons prefer to occupy individual orbitals within a subshell before pairing up. So, they'll spread out first, like friends picking out their favorite seats before anyone gets cozy on the same couch.
And finally, the Pauli exclusion principle. This one is the strict roommate rule: no two electrons in the same atom can have the exact same quantum state. Basically, if two electrons are in the same orbital, they must have opposite spins. It's like a password that only one person can use, or a specific parking spot reserved for only one type of car. This little rule is crucial for keeping everything orderly.

Why is all this important? Because this electron arrangement is the secret sauce behind everything we see and experience! It determines how atoms interact with each other, how they form bonds, and ultimately, how they create molecules. It's the reason why water is wet, why fire is hot, and why your favorite ice cream tastes so delicious. It’s the tiny dance of electrons that allows for the grand symphony of the universe.
Think about it: the vibrant colors of a rainbow? The ability of a plant to photosynthesize? Even the way your brain sends signals? All of this relies on the precise dance of electrons. It’s a humbling thought that something so incredibly small dictates so much of our reality. It's like the subtle stitching on a masterpiece painting that you might not notice at first, but without it, the whole picture would fall apart.

The "answer key" to Chapter 4 isn't just a list of numbers; it's the blueprint for the entire chemical world. It's a story of order emerging from what could be pure chaos, a testament to the elegant simplicity that underpins complexity.
Sometimes, understanding these concepts can feel like deciphering a secret code. But once you get the hang of it, you start to see the world with new eyes. You'll look at a simple salt crystal and imagine the orderly rows of ions, each with its perfectly placed electrons. You'll see a vibrant flower and marvel at the intricate electron configurations that allow it to capture sunlight. It's like learning a new language, and suddenly, you can understand the whispers of the atoms around you.
So, the next time you encounter "Chapter 4 Arrangement of Electrons in Atoms," don't groan. Instead, smile. You're not just looking at a textbook; you're peering into the fundamental architecture of existence. You're learning about the tiny, energetic dancers that make the universe spin, and that, my friends, is pretty darn amazing.
