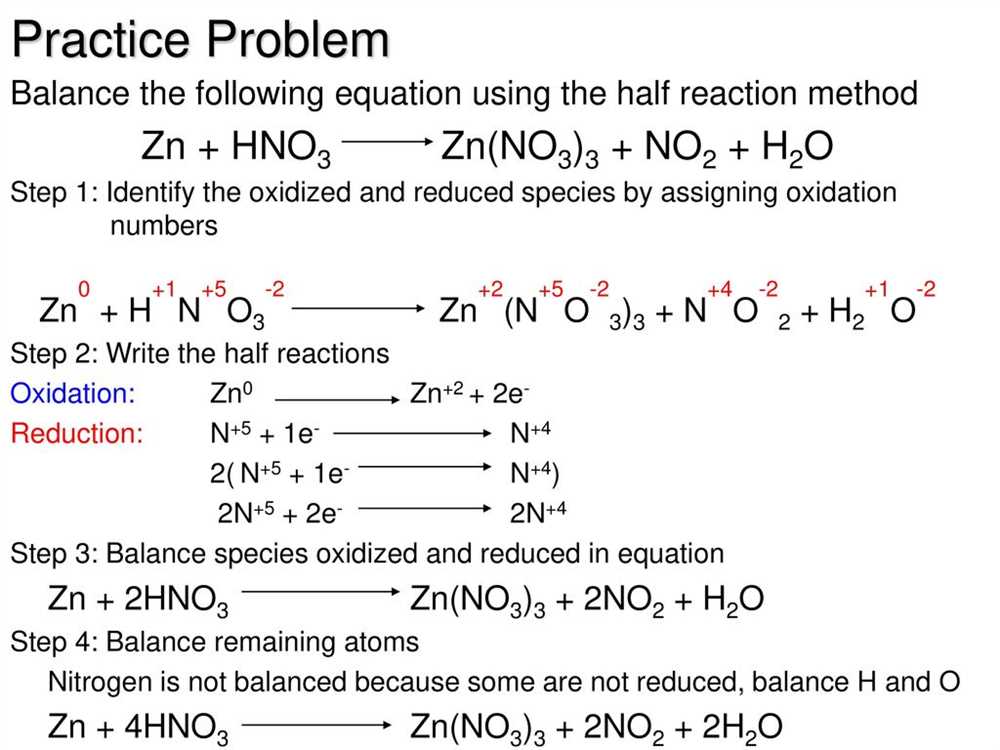
Chapter 20 Oxidation Reduction Reactions Answer Key
Ever find yourself wondering about the magic behind a sizzling steak, a glowing battery, or even how your body uses food for energy? Well, get ready for a little peek behind the curtain, because we're diving into the fascinating world of oxidation-reduction reactions, often shortened to redox reactions. Think of it as a cosmic dance of electrons, a fundamental process that underpins so much of what we see and experience every single day. Learning about redox isn't just about memorizing chemical formulas; it's about understanding the very essence of change and energy transfer in the universe.
The purpose of studying redox reactions is to unravel how substances gain or lose electrons, leading to transformations. It's like observing a tug-of-war where electrons are the prize. When one substance loses electrons, it's called oxidation, and when another substance gains them, it's called reduction. These two processes always happen together – you can't have one without the other. The benefits are immense. Understanding redox is crucial for fields like chemistry, biology, environmental science, and even engineering. It helps us develop new technologies, understand biological processes, and tackle environmental challenges.
Let's sprinkle in some real-world magic. Think about the rusting of iron. That orange coating isn't just pretty (or annoying, depending on your perspective!); it's a classic example of oxidation. The iron atoms lose electrons to oxygen. On the flip side, consider the power behind your smartphone or electric car: batteries. These devices work because of carefully controlled redox reactions, where chemical energy is converted into electrical energy. Even something as simple as photosynthesis, the process by which plants convert sunlight into energy, involves intricate redox steps. And don't forget about the combustion of fuels that power our vehicles and heat our homes – that's a rapid redox reaction happening right before our eyes!
So, how can you explore this electrifying topic without feeling overwhelmed? Start with the basics. Look for everyday examples around you. Observe how food browns when exposed to air (oxidation!) or how a metal might tarnish. You can even try simple home experiments, like observing the reaction between a copper penny and some vinegar, which involves a mild redox process. Many educational websites and videos offer fantastic visual explanations of redox reactions. They often use analogies to make the electron transfer concept more intuitive. Don't be afraid to ask questions and explore the "why" behind these chemical transformations. The more you look, the more you'll see the silent, constant dance of electrons shaping our world, making the answer key to Chapter 20 not just a set of solutions, but a gateway to deeper understanding.
