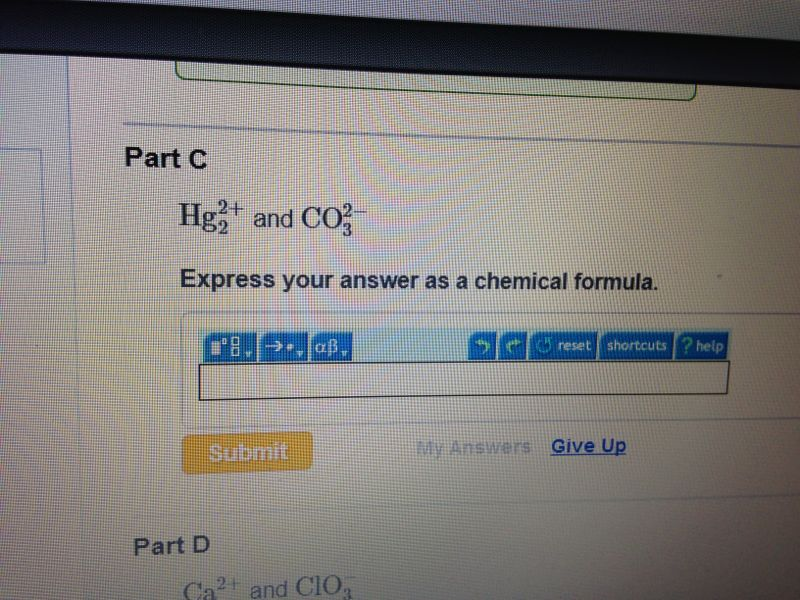
Carbonate Express Your Answer As A Chemical Formula
Okay, picture this: I was at my cousin Brenda's wedding last weekend. Lovely affair, really. Lots of flowers, questionable karaoke choices (Uncle Gary, we love you, but…), and, as always, a slightly-too-long reception. During the appetizer portion, I ended up chatting with Brenda's new father-in-law, a distinguished gentleman named Arthur. Arthur, bless his heart, is a bit of a character. He’s the type who’ll launch into a detailed explanation of the merits of different pruning shears while you’re just trying to snag a mini quiche.
Anyway, Arthur gets on the topic of… wait for it… rocks. Specifically, rocks from his garden. Apparently, he has this fascinating collection of… wait for it again… limestone. He started waxing poetic about its “ancient origins” and “geological significance.” My brain, fueled by champagne and a desperate need to avoid discussing Brenda’s aunt’s cat’s latest grooming mishap, started to glaze over. But then, he uttered something that, surprisingly, snagged my attention. He said, with a twinkle in his eye, that these rocks were basically holding onto a hugely important chemical secret.
And that, my friends, is how I found myself thinking about… well, about something Arthur probably thinks is deeply profound, but which, in reality, is everywhere. You know, those tiny, fizzy bubbles that sometimes surprise you in your drink? Or that chalky feeling when you’re trying to scrape off some stubborn gunk? Yep, we’re talking about the Carbonate Express, and its answer, as promised, is a chemical formula: CO₃²⁻.
Yeah, I know. Not exactly the cliffhanger you were expecting. But stick with me. This little ion, this CO₃²⁻, is kind of a rockstar. And by "rockstar," I mean it's absolutely everywhere, doing its thing in ways you probably don't even realize.
Think about it. Arthur was talking about limestone. What is limestone, chemically speaking? It's mostly calcium carbonate. And guess what calcium carbonate is? It's essentially a calcium ion (Ca²⁺) holding hands with our friend, the carbonate ion (CO₃²⁻). So, the formula for calcium carbonate is CaCO₃. See it? That little powerhouse, CO₃²⁻, is right there in the middle of a rock that’s been forming for… well, geological ages. Mind-blowing, right?
But it's not just about fancy rocks in a garden. This CO₃²⁻ is involved in some pretty epic stuff. Let’s take a little detour, shall we? Ever been to the beach and seen those shells? Or that sand? A ton of that is also made of carbonates. Mussels, oysters, clams, the tiny shells that make up coral reefs – they all use calcium carbonate to build their homes. So, when you're building a sandcastle, you're literally playing with the skeletal remains of countless tiny marine creatures, all held together by our friend CO₃²⁻. It's kind of beautiful, and also a little bit macabre if you think about it too hard. Don't think about it too hard.
And then there’s the whole… breathing thing. Yes, breathing. Our blood needs to maintain a very specific pH level to keep us alive and kicking. And guess what plays a huge role in that delicate balance? Yep. The carbonate system. When you exhale carbon dioxide (CO₂), it dissolves in your blood and, through a series of chemical reactions, eventually forms carbonic acid (H₂CO₃). Now, carbonic acid is a bit unstable, and it breaks down into… you guessed it… a hydrogen ion (H⁺) and our trusty bicarbonate ion (HCO₃⁻). And guess what the bicarbonate ion is? It's basically a carbonate ion that's picked up a hydrogen. It’s like the slightly more introverted cousin of CO₃²⁻.
This whole bicarbonate buffer system is incredibly important. It helps to soak up excess hydrogen ions, preventing your blood from becoming too acidic. It's like having a tiny chemical sponge in your bloodstream, working overtime to keep you in the land of the living. So, the next time you take a deep breath, thank CO₃²⁻. It’s helping you do that. Pretty wild, huh?
But wait, there’s more! The oceans. Oh, the oceans. They are full of dissolved carbon dioxide. And when CO₂ dissolves in water, it forms carbonic acid, which then dissociates into hydrogen ions and bicarbonate ions, and also, you guessed it, carbonate ions. The ocean is like a giant, salty bathtub for our friend CO₃²⁻. It's estimated that about 90% of the world’s dissolved carbon is stored in the oceans, a massive chunk of which is in the form of bicarbonate and carbonate ions.
This is super important for regulating the Earth’s climate. The oceans act as a massive carbon sink, absorbing CO₂ from the atmosphere. Without this process, the greenhouse effect would be a whole lot worse. However, as we pump more and more CO₂ into the atmosphere, the oceans absorb more of it, leading to ocean acidification. This is where our friend CO₃²⁻ gets a little stressed. More CO₂ means more carbonic acid, which means more hydrogen ions, which can then react with carbonate ions, reducing the amount of free carbonate available for marine organisms to build their shells and skeletons. It’s a bit of a domino effect, and not a good one. So, while CO₃²⁻ is a hero in many ways, it’s also on the front lines of some pretty serious environmental challenges.
Let’s rewind a bit and think about those fizzy drinks I mentioned. Ever wondered why soda fizzes? That’s carbonation. And carbonation is basically dissolved carbon dioxide under pressure. When you open the can or bottle, the pressure is released, and the dissolved CO₂ starts to escape, forming those delightful little bubbles. These bubbles are a transient state, a fleeting glimpse of carbon dioxide interacting with water. But the chemistry behind it is still tied to our carbonate family. The CO₂ dissolves and forms carbonic acid, which then releases hydrogen ions and bicarbonate ions. The carbon dioxide itself is the star, but the presence of water and the subsequent reactions involve these carbonate species.
And what about baking? You know, when you’re whipping up some cookies or a cake? Many baking ingredients rely on the magic of carbonates to make things rise. Baking soda, for instance, is sodium bicarbonate (NaHCO₃). When you add it to a recipe with an acidic ingredient (like buttermilk or lemon juice) and heat, it decomposes to produce carbon dioxide gas. This gas gets trapped in the batter, causing it to expand and become fluffy. So, that light and airy texture in your favorite baked goods? Yep, you can thank a close relative of CO₃²⁻ for that. It’s like a miniature chemical explosion happening in your oven, all thanks to a few simple ingredients and a very helpful ion.

I’ve always been fascinated by how these seemingly simple chemical formulas represent such complex and vital processes. CO₃²⁻ isn’t just a string of symbols; it’s a building block of life, a regulator of our planet, and a key player in everything from the deepest oceans to the air we breathe. It's in the chalk on a blackboard, the marble of a statue, the shells of ancient sea creatures, and the very blood coursing through our veins.
Arthur, with his passion for limestone, was actually onto something pretty big. That seemingly inert rock was a testament to the enduring power and prevalence of the carbonate ion. It’s a reminder that even the most ordinary things around us are often underpinned by incredible chemistry. It’s like finding a secret code embedded in the fabric of our world.
So, the next time you see a chalky residue, hear the fizz of a drink, or admire a shell on the beach, take a moment to appreciate the unsung hero: the carbonate ion. It’s the silent workhorse, the ubiquitous presence, the essential component. And its answer, when you ask what it is? CO₃²⁻. Simple, yet profound. And now you know. You’re welcome. (And maybe don't mention Uncle Gary's karaoke to Arthur, unless you want to hear about the geological strata of the dance floor).
