Carbon Monoxide And Chlorine Gas React To Form Phosgene

So, you know how sometimes you hear about chemicals doing their thing, and it sounds super complicated and maybe a little scary? Well, guess what? Today, we're going to chat about something that sounds like it’s straight out of a mad scientist’s lab, but it’s actually pretty fascinating. We're talking about carbon monoxide and chlorine gas deciding to, like, get together. Yep, you heard me. They have a little chemical rendezvous, and guess what they make? Phosgene. I know, right? Phosgene. Sounds like a medieval torture device, doesn't it? But hang in there, it’s not all doom and gloom… well, not entirely. We're just going to have a friendly chat about it, like we’re dissecting a weird science experiment over a cup of our favorite brew. So, grab that mug, settle in, and let’s dive into this rather… interesting chemical pairing.
First off, let's talk about our players. We've got carbon monoxide, right? That sneaky gas that you can't see, smell, or taste. Kind of like that friend who always finishes the last cookie without telling anyone. Invisible and a little bit of a problem if you get too much of it. We usually think of it coming from car exhaust or faulty furnaces. It's basically a carbon atom doing a solo act with an oxygen atom. CO, they call it. Simple, but don't underestimate its power to cause trouble. It's like the quiet kid in class who secretly aced all the tests. You just… wouldn't know.
Then there's chlorine gas. Now, chlorine, that’s a different vibe. You know that smell at swimming pools? That's kind of like chlorine, but real chlorine gas is a lot more intense. It's a greenish-yellow gas, and let me tell you, it's no joke. It’s one of those elements that's super reactive. It's like that friend who's always up for an adventure, maybe a little too up for it. It's diatomic, meaning it likes to hang out with its twin, Cl₂, forming this rather enthusiastic molecule. It's got a real… bite to it, if you catch my drift. It’s a bit of a drama queen in the chemical world, always ready to react with pretty much anything that stands still for too long.
So, you have carbon monoxide, the quiet troublemaker, and chlorine gas, the flamboyant daredevil. What happens when these two decide to team up? It’s like the universe thought, "Let's see what happens when we put these two together." And BAM! They form… phosgene. Yes, that phosgene. It's a chemical compound with the formula COCl₂. And honestly, the name itself just sounds… menacing, doesn't it? Phosgene. It’s got this old-school, slightly sinister ring to it. Like something you'd find in a dusty alchemist's book, whispering secrets of the universe (or, you know, creating chemical weapons). Not exactly what you’d want to stumble upon during a casual picnic, that’s for sure.
Now, the actual how of it. It's not like they just meet and say, "Hey, let's make phosgene!" There’s a bit of… persuasion involved. Usually, you need a little something to get the party started, like a bit of a spark, or heat, or even some other catalysts. Think of it like needing a DJ to get the dance floor going. You can't just put two shy people in a room and expect them to spontaneously break out into a tango, right? You need some music, some… energy. In the case of CO and Cl₂, this energy often comes in the form of UV light or heat. So, if you expose them to sunlight, or heat them up, they’re more likely to say, "Okay, you know what? Let’s do this thing."

The reaction itself is pretty straightforward, chemically speaking. It's an addition reaction. The chlorine molecule, Cl₂, splits into two individual chlorine atoms, which are super eager to bond with something. The carbon monoxide molecule, CO, is also ready for some action. So, one of those free chlorine atoms latches onto the carbon, and the other latches onto the oxygen. It’s like they’re playing chemical musical chairs, but with only two players and a very specific outcome. The result is COCl₂ – phosgene. And it happens pretty efficiently under the right conditions. It’s like a perfectly choreographed chemical dance, except the result is… well, not exactly something you'd frame and put on your mantelpiece.
Why is this particular concoction so… noteworthy, you ask? Because phosgene is what we call a lachrymator. Fancy word, right? It means it makes your eyes water. A lot. Like, cry-your-eyes-out, can’t-see-anything, snot-running-down-your-face level of watering. But it’s not just your eyes that get a rough deal. Oh no. Phosgene also likes to mess with your lungs. It can cause severe irritation and damage to the respiratory system. It’s like it’s got a personal vendetta against your lungs. It’s a highly toxic gas, and even small amounts can be incredibly dangerous. We’re talking about fluid buildup in the lungs, difficulty breathing, and in severe cases, well… it’s not pretty. It’s definitely not the kind of "surprise" you want.
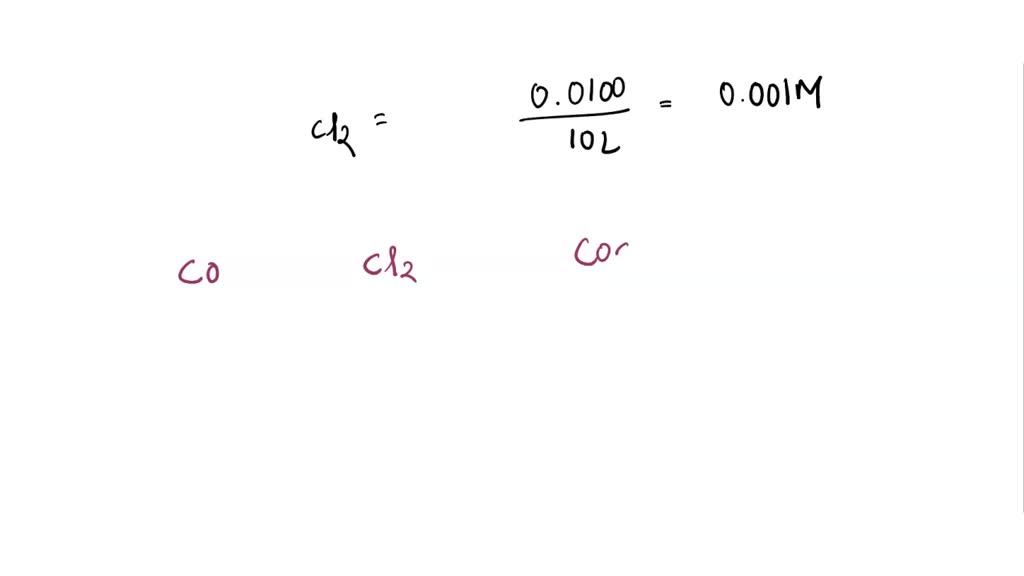
Historically, phosgene has a rather grim reputation. It was, unfortunately, used as a chemical weapon during World War I. Yeah, I know. Talk about a bad rap. They were looking for something to be effective, and unfortunately, phosgene fit the bill. It’s insidious because the effects aren't always immediate. You can be exposed, feel a little bit of irritation, and then hours later, the real problems start to kick in. It’s like a slow-burn disaster. This delayed effect made it particularly terrifying. Imagine thinking you’re in the clear, and then… bam. The realization hits you, and not in a good way. It's like a twisted prank, but with potentially fatal consequences. Definitely not in the "funny when you're old" category of stories.
So, where might you encounter this dynamic duo and their potentially problematic offspring in everyday life? Honestly, in a controlled, everyday sense, it’s pretty rare. The conditions needed for the direct reaction to occur in significant amounts aren't usually found in your average home. You’re not going to accidentally create phosgene by leaving your CO detector next to your pool chlorine tablets, for instance. That's a relief, right? Your everyday CO detectors are designed to detect carbon monoxide, and while they're crucial for safety, they're not going to accidentally trigger a phosgene explosion. Phew. However, it's worth noting that sometimes, in industrial settings or specific chemical processes, these gases might be present, and the risk of phosgene formation could be a concern. Safety protocols are super important in places like that. Think of it like handling raw chicken; you need to be careful, wash your hands, and cook it properly. With chemicals, the stakes are just… higher.
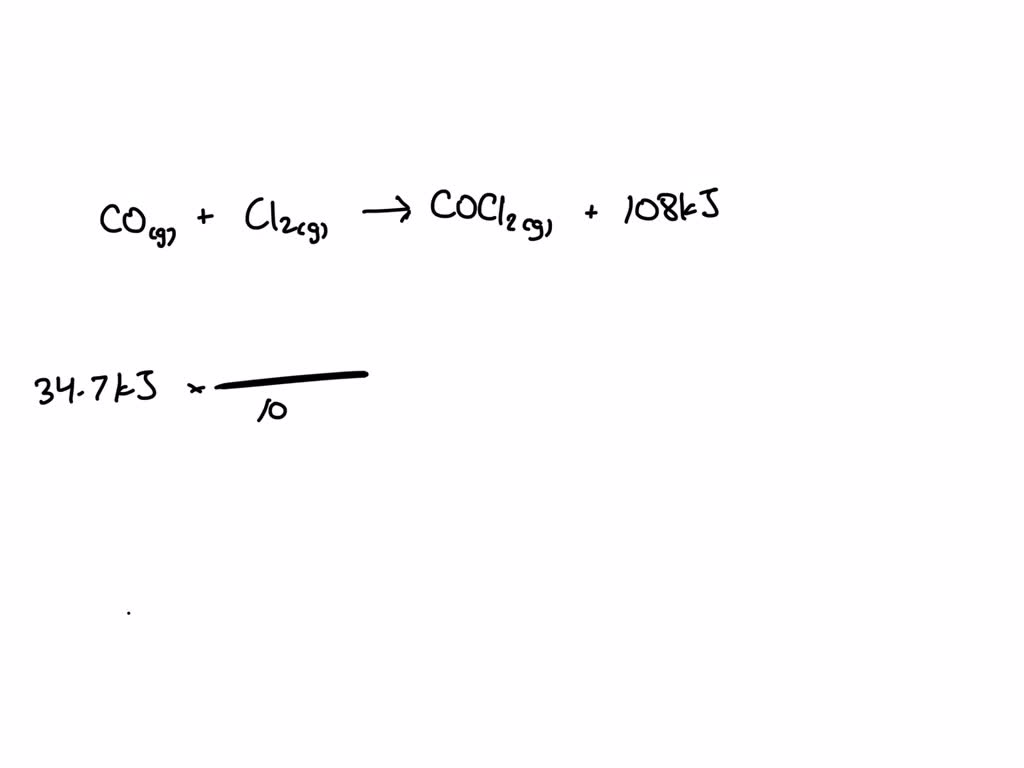
Interestingly, though, there are other scenarios where phosgene can be formed indirectly. For example, if you have materials that contain both carbon and chlorine, and they're exposed to high temperatures or certain types of radiation, you might get some phosgene. Think about certain plastics or even some cleaning products. Accidentally mixing certain cleaning chemicals can also be a recipe for disaster, and while phosgene might not be the primary culprit in most common household mixing mishaps, it's a good reminder that you should never mix cleaning products unless the label explicitly says it's okay. Seriously, read those labels! They’re not just suggestions; they’re often there to prevent you from creating your own little chemical hazard zone. It’s like a choose-your-own-adventure book, but the bad endings are significantly more… unpleasant.
What about the science behind why this reaction happens? Well, it’s all about electron configurations and stability. Carbon monoxide has a triple bond between the carbon and oxygen atoms, which is pretty strong. But it's also a bit polar. Chlorine, on the other hand, loves to grab electrons. When you give them the energy boost – the UV light or heat – it’s like you’re giving them the courage to break apart and form new, more stable bonds. The carbon atom in CO can become quite electron-deficient, and the chlorine atoms are just itching to fill that void. It’s a bit like a chemical matchmaking service gone right, but again, the resulting couple isn't exactly a match made in heaven for us humans. They’re finding stability, which is what most things in nature strive for, even if that stability is detrimental to us.

So, what’s the takeaway from all this? Carbon monoxide and chlorine gas are two common, but potentially dangerous, substances. When they’re nudged in the right way, they can form phosgene, a gas that’s a serious irritant and toxic. While you’re not likely to accidentally create a phosgene bomb in your kitchen, understanding these basic chemical reactions is pretty cool, and it also highlights why safety is so important when dealing with chemicals, even the seemingly mundane ones. It’s a good reminder to be aware of your surroundings and the potential hazards, both obvious and… less obvious. It’s like knowing that leaving the stove on unattended is a bad idea; it’s just common sense applied to the chemical world.
Think of it this way: carbon monoxide is like that quiet friend who's surprisingly strong. Chlorine is like the energetic friend who's always making a scene. Put them together under the right circumstances, and you get phosgene, which is like that third friend who shows up at the party and causes all the drama. It’s a chemical drama, to be precise. And honestly, while the science behind it is pretty neat, the consequences are definitely something we want to avoid. So, we'll appreciate the chemical reaction from a safe distance, thank you very much. It’s a perfect example of how even simple elements can combine to create something with a very significant, and often dangerous, impact. Science is amazing, isn't it? Even when it's a little bit terrifying.
The key takeaway here, really, is just awareness. We're not going to be performing this reaction in our backyards, but knowing that it can happen helps us appreciate the importance of handling chemicals safely. It’s about respecting the power of chemistry. Carbon monoxide alarms are important. Proper ventilation is important. Not mixing random household cleaners is definitely important. It's all about those little steps that keep us safe and sound. So, the next time you hear about chemical reactions, remember this little chat about CO and Cl₂. It’s a reminder that the world around us is constantly buzzing with activity, and sometimes, understanding that activity can be pretty darn useful. And hey, it makes for a pretty interesting conversation topic, wouldn't you agree? Now, who wants more coffee?
