Carbon Bonding And Functional Groups Mastering Biology Answers
Hey there, science explorers! Ever wondered what makes that delicious cookie taste so good, or why that superhero's suit is so tough? Believe it or not, it all comes down to the tiny, invisible building blocks of everything around us – molecules! And today, we're going to chat about the coolest part of molecular architecture: carbon bonding and these super-important bits called functional groups. Don't worry, we're not heading back to school for a pop quiz; think of this as a friendly chat over coffee, with a sprinkle of awesome science!
So, what's the big deal about carbon? Well, carbon is like the master architect of the molecular world. It's incredibly versatile, meaning it can link up with itself and a bunch of other elements in so many different ways. Imagine Lego bricks – carbon atoms are like the most adaptable ones. They can form long chains, rings, even branched structures. This ability is the foundation for all life on Earth!
Think about it: the proteins that build our muscles, the DNA that carries our genetic information, the sugars that give us energy – they're all built around a backbone of carbon atoms. Without carbon's amazing bonding skills, we wouldn't have pizza, puppies, or even our own amazing bodies. Pretty important stuff, right?
Carbon: The Ultimate Connector
Carbon has this special talent for forming four strong bonds with other atoms. This is like having four hands to hold onto other molecules. It can connect with other carbon atoms, creating those long chains we talked about. This is how you get molecules like fats and carbohydrates, which are basically energy storage units for your body. Think of them as tiny rechargeable batteries!
But carbon isn't selfish; it also loves to bond with other elements like hydrogen (think water, H₂O, but in organic molecules!), oxygen (essential for breathing and also found in sugars!), nitrogen (key for proteins!), and even sulfur and phosphorus. This variety of connections allows for an astonishing diversity of molecules. It's like an artist with a massive palette of colors, able to create an infinite number of unique paintings.
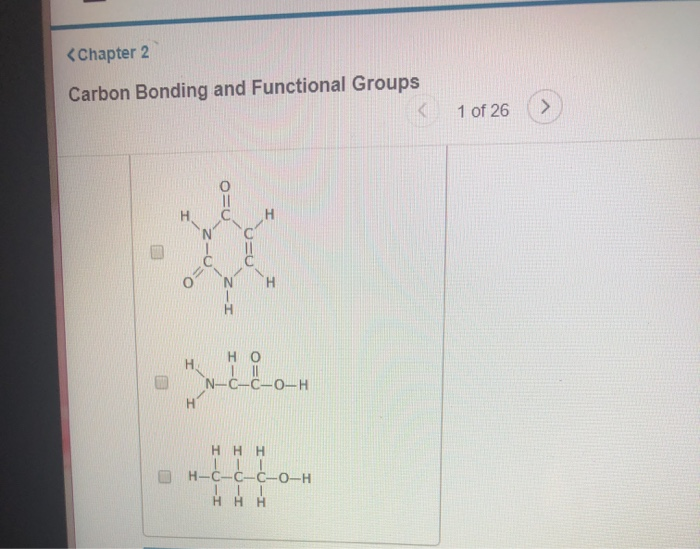
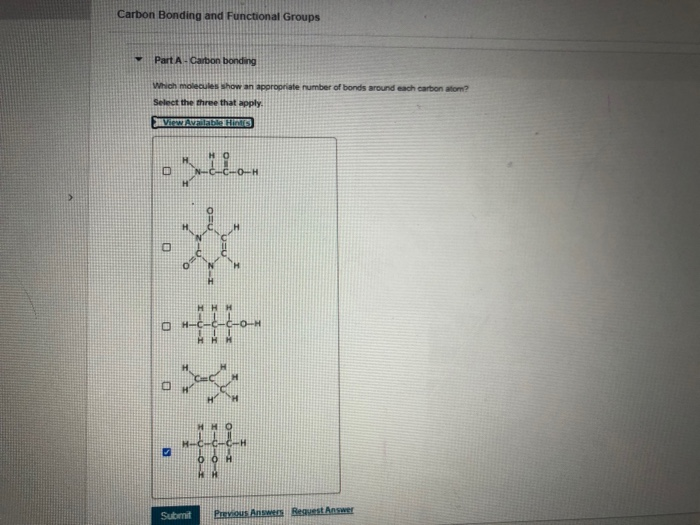
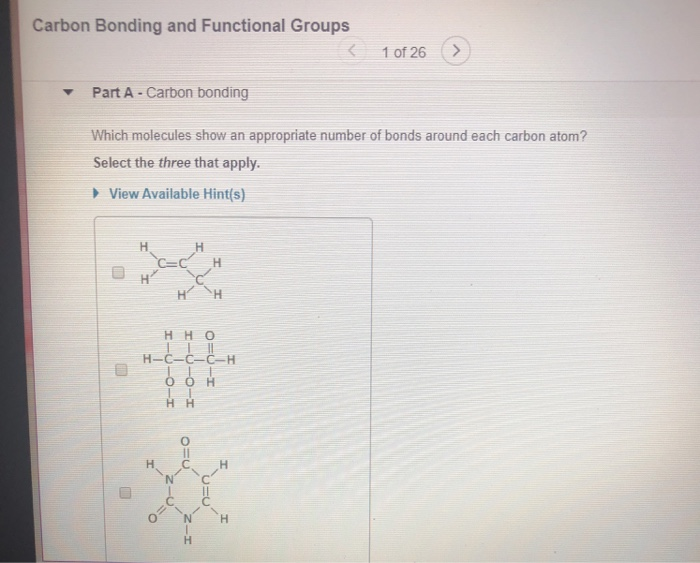
Let’s say you have a simple chain of carbon atoms. It might look like a string of pearls. Now, imagine attaching different things to those pearls. Some might have little hydrogen atoms, making it a basic hydrocarbon. Others might have oxygen atoms, which can change its properties dramatically. This is where functional groups come in!
Functional Groups: The "Special Effects" of Molecules
Think of functional groups as the "party hats" or "superpowers" that attach to the carbon backbone. They are specific arrangements of atoms that give a molecule its unique chemical personality and determine how it will behave. They're like adding a different condiment to your fries – the fries (carbon backbone) are the same, but the ketchup or the aioli (functional groups) completely change the flavor and experience!
So, why should you care about these "party hats"? Because they dictate everything from how food tastes and smells to how medicines work in your body. They're the secret ingredients that make life happen!
Meet Some of the "Party Animals" (Functional Groups)
Let's meet a few of the common functional groups. Don't be intimidated by the names; they're more like nicknames for specific molecular features.

The Hydroxyl Group (-OH): The "Alcohol" Crew
When you see an oxygen atom bonded to a hydrogen atom, attached to a carbon chain, you've found a hydroxyl group. This is what makes alcohols, well, alcohols! Think of the ethanol in your hand sanitizer or that refreshing glass of juice (which contains sugars with hydroxyl groups). These groups make molecules more soluble in water, kind of like adding a little soap to water makes it easier to clean things. So, they help molecules mingle better in our watery bodies.
The Carbonyl Group (C=O): The "Sweet & Savory" Duo
This is where a carbon atom is double-bonded to an oxygen atom. It's like a really strong handshake! If the carbonyl group is at the end of a carbon chain, it's called an aldehyde. If it's somewhere in the middle, it's a ketone. Sugars often have carbonyl groups, which is why they taste sweet! Ketones are also important in our bodies, like in the process of breaking down fats.
The Carboxyl Group (-COOH): The "Acidic Avengers"
This group is like a hybrid: it has both a carbonyl group (C=O) and a hydroxyl group (-OH) attached to the same carbon atom. This combination makes the molecule slightly acidic. Think of the acetic acid in vinegar – that's what gives it its tangy bite! Many amino acids, the building blocks of proteins, have carboxyl groups. They are the "sour patch kids" of the functional group world, adding a distinct flavor.

The Amino Group (-NH₂): The "Basic Builders"
This group has a nitrogen atom bonded to two hydrogen atoms. Amino groups are slightly basic and are absolutely crucial for building proteins. They're like the "glue" that helps hold amino acids together to form those long, complex protein chains. Without them, we wouldn't have the enzymes that digest our food or the antibodies that fight off sickness.
The Phosphate Group (-PO₄³⁻): The "Energy Packets"
This one is a bit more complex, with a phosphorus atom surrounded by oxygen atoms. Phosphate groups are super important for energy transfer in our cells. Think of ATP (adenosine triphosphate) – it's like the universal energy currency of our cells, and it's packed with phosphate groups! They're the tiny power-ups that keep everything running.
Why Should You Care, Really?
Okay, so we've talked about carbon as the master architect and functional groups as the fancy decorations. But why is this relevant to your everyday life beyond knowing your food is made of molecules?
Understanding these basic principles helps us understand ourselves and the world around us. When you eat a banana, you're getting carbohydrates (chains of carbon with hydroxyl groups) for energy. When your body repairs a cut, it's using proteins built from amino acids (with amino and carboxyl groups). When you take medicine, you're often taking a molecule with specific functional groups designed to interact with certain targets in your body.
Think of a doctor diagnosing an illness or a scientist developing a new drug. They're working with these molecular structures. Even if you're not planning on becoming a biochemist, having a basic grasp of how molecules are built and how they interact is like having a secret decoder ring for understanding how life works. It’s the foundation of biology, and it’s happening inside you right now!
These concepts are often the first big hurdle in biology, and sometimes they can feel a bit abstract. But remember, they're the fundamental rules that govern the universe of living things. By understanding carbon bonding and functional groups, you're unlocking a deeper appreciation for the complexity and beauty of life. It’s not just about memorizing terms; it’s about grasping the underlying logic that makes everything tick.
So, the next time you’re enjoying a meal or marveling at the intricate design of a flower, take a moment to appreciate the incredible world of molecular chemistry. It’s the silent, unseen force that makes it all possible, and it's far more fascinating than you might have ever imagined!
