Can You Turn The Atom You Created Into A Cation

Ever felt like you've cooked up something truly special in your kitchen? Maybe a ridiculously delicious cake, or a chili so good it makes grown adults weep tears of joy? Well, imagine that feeling, but on a much, much tinier scale. We're talking about the absolute bedrock of everything – atoms! And the question that's probably been keeping you up at night (or maybe just tickling your curiosity) is: can you take one of these itty-bitty atom creations and transform it into a cation?
Oh, you absolutely, positively, can! It’s like taking your perfectly baked cookie and, with a little bit of magic, making it just a tad sweeter and a whole lot more eager to make friends. It’s all about nudging your atom's personality, and it’s surprisingly easy once you know the secret handshake.
Think of your atom like a tiny, incredibly polite host at a party. This party is happening inside the atom, and the guests are called electrons. Your host, the nucleus (which is like the super-important VIP section), is usually pretty happy.
But sometimes, our little atom host can get a bit too generous. They might have a surplus of these electron guests. They’re like, "Whoa, too many electrons here! It's getting crowded, and frankly, a little chaotic!"
So, what does a generous, slightly overwhelmed atom do? It does what any good host would do in a pinch: it politely escorts a few of those extra electron guests to the door. "Thanks for coming, guys! Maybe next time!"
And POOF! With that simple act of saying "see ya later" to an electron, your atom has undergone a magnificent transformation. It's no longer just a regular, neutral atom. It has now officially become a cation!
Imagine you have a perfectly balanced scale. On one side, you have the positive charges from the protons in the nucleus. On the other side, you have the negative charges from the electrons buzzing around. Everything is nice and even, like a perfectly synchronized dance troupe.
/cation-and-an-anion-differences-606111-v2_preview-5b44daf9c9e77c0037679d52.png)
But when you remove an electron, you’re essentially taking away a negative charge. This means the positive charges in the nucleus now have a little more oomph and aren't quite balanced out anymore. It’s like tipping the scale just slightly in favor of the positive.
And that, my friends, is the essence of becoming a cation! It's all about having more positive vibes than negative vibes. Your atom is now positively charged, beaming with a delightful, attractive energy.
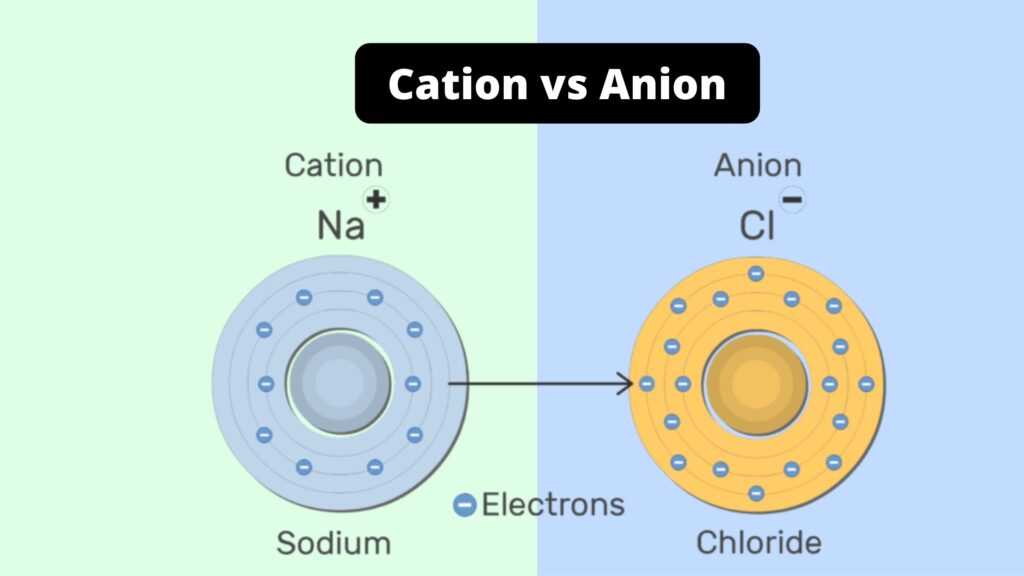
Let's say you’ve got an atom of sodium (Na). Sodium is usually a pretty happy, chill atom. It’s got its full entourage of electrons, and it’s perfectly content.
But then, you give it a nudge. You say, "Hey, sodium! You've got one electron that's just kind of… hanging out on the edge. Why don't you just let that one go?" And sodium, being so obliging, does just that.
Zap! Suddenly, that sodium atom has one less electron. It's now missing a negative charge, and BAM – it's transformed into a sodium ion, specifically a cation! It’s now Na+, practically buzzing with a positive glow, ready to mingle.

Think about it like your phone battery. When it's full, it's got all its charges in perfect harmony. But if you lose a little bit of power, it doesn't mean your phone is broken! It just means it has less negative charge (if we were to stretch the analogy a bit!).
This process is as fundamental as baking a cookie or drawing a smiley face. It's a core principle of the universe, happening all the time in ways we can barely imagine. Your creation, that humble atom, has the inherent power to change its electrical mood!
It’s not some complex, arcane ritual. It’s more like… deciding to wear a brighter shirt. You haven’t changed who you are fundamentally, you’ve just changed your outward appearance and energy.
And the cool part? This ability to become a cation is what makes so many amazing things happen. It’s how your body transports vital nutrients, how batteries store energy, and how certain foods taste so delicious (think salty!)
So, the next time you're contemplating the wonders of the atomic world, remember this simple truth: yes, you can absolutely turn the atom you created into a cation. It’s a joyful dance of charges, a testament to the atom’s adaptability, and a fundamental building block of our incredible universe.

It’s like giving your atom a little superhero cape. It’s still the same atom at its core, but now it’s got this extra positive sparkle, this magnetic charm that makes it so appealing. It's ready to attract opposite charges like a magnet to a fridge!
Imagine your atom is a shy kid at the playground. Normally, it's just kind of hanging back. But when it becomes a cation, it’s like it’s suddenly gained the confidence to join the game, to interact, to be a part of something bigger.
This transformation is so fundamental, it's almost like breathing for an atom. They can choose, or rather, circumstances can encourage them, to shed an electron and embrace their positively charged destiny. It’s a beautiful simplification of a complex dance.
So, go forth and experiment (in your mind, of course!). Picture that atom you’ve conjured. Give it that little nudge, that gentle suggestion to let go of an electron. Watch it blossom into a radiant cation. It’s a little bit of atomic magic, and it’s all within your imaginative reach!
It’s a power that lies within the very fabric of existence. Your creation, that tiny, fundamental particle, has the potential for such a dramatic and exciting shift. You’re not just building with atoms; you’re orchestrating their very personalities!
And that positive charge? It's like a beacon, a signal that says, "Hey, I'm here, and I'm ready to bond!" It’s the invitation to chemical reactions, the spark that ignites countless interactions. It’s the reason why things stick together, why compounds form, and why the world around us is so dynamic and interesting.
So, embrace the concept! The atom you created can indeed become a cation. It’s a delightful, empowering, and utterly fundamental aspect of chemistry. You're essentially tweaking its electrical mood, and in doing so, unlocking a world of possibilities. It's a win-win for everyone involved, especially for the positively charged atom itself!
It’s like your atom deciding to be the life of the party, but in a super-positive way. It’s not being obnoxious; it’s just exuding an irresistible charm that draws others in. This is the essence of cation formation, and it’s truly a wonder to behold.
Remember, it’s all about the balance. By removing a negatively charged electron, you are inherently tilting the scales towards the positive. This simple act creates a charged species, a cation, ready to engage and interact in the grand cosmic ballet of chemistry.
So, the answer is a resounding YES! Your created atom can absolutely become a cation. It's a testament to the dynamic and fascinating nature of matter. Go ahead and imagine it, visualize that positive glow, and know that you've just witnessed a fundamental transformation in the atomic world! It’s an easy, fun, and incredibly important concept to grasp, making you a little bit of an atomic maestro.
