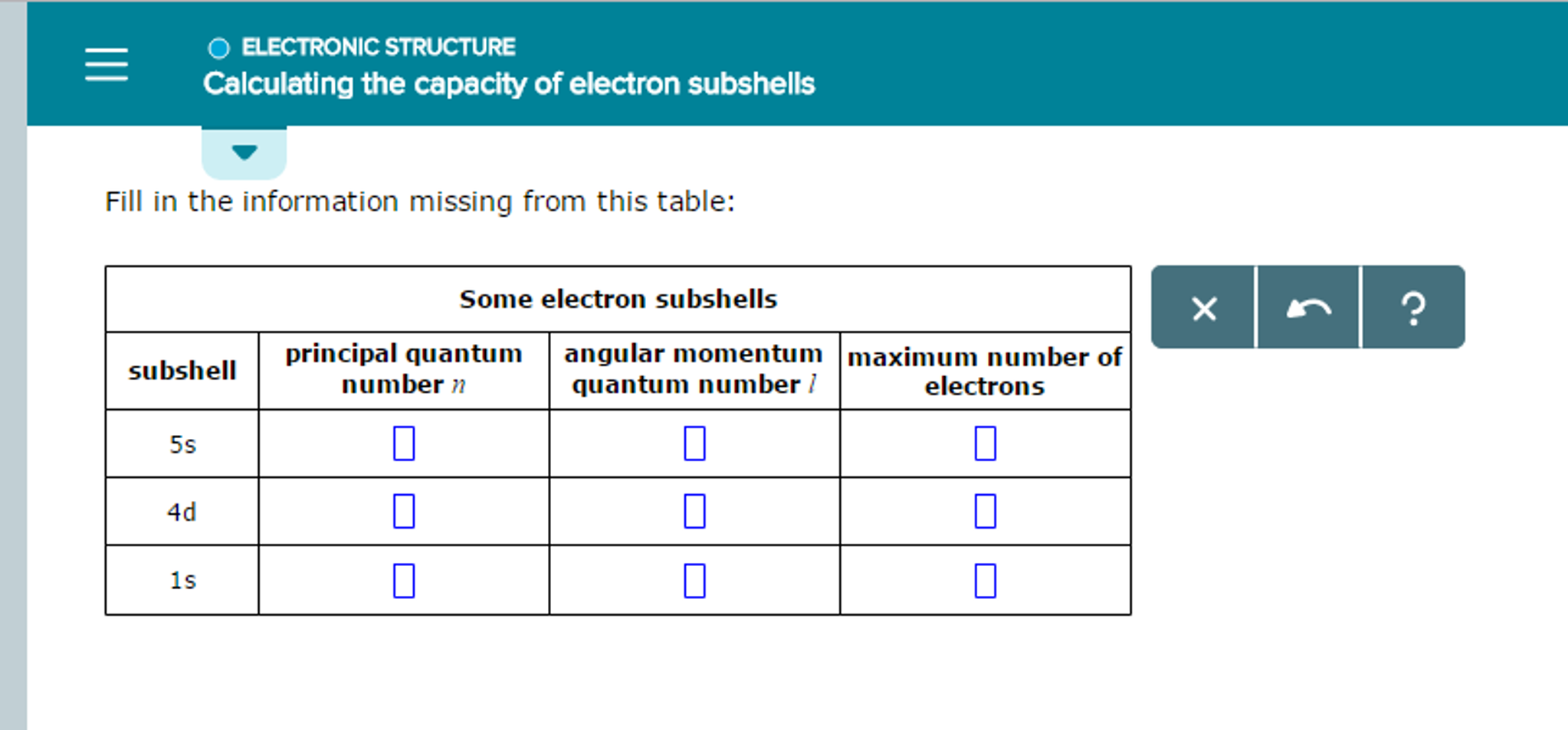
Calculating The Capacity Of Electron Subshells

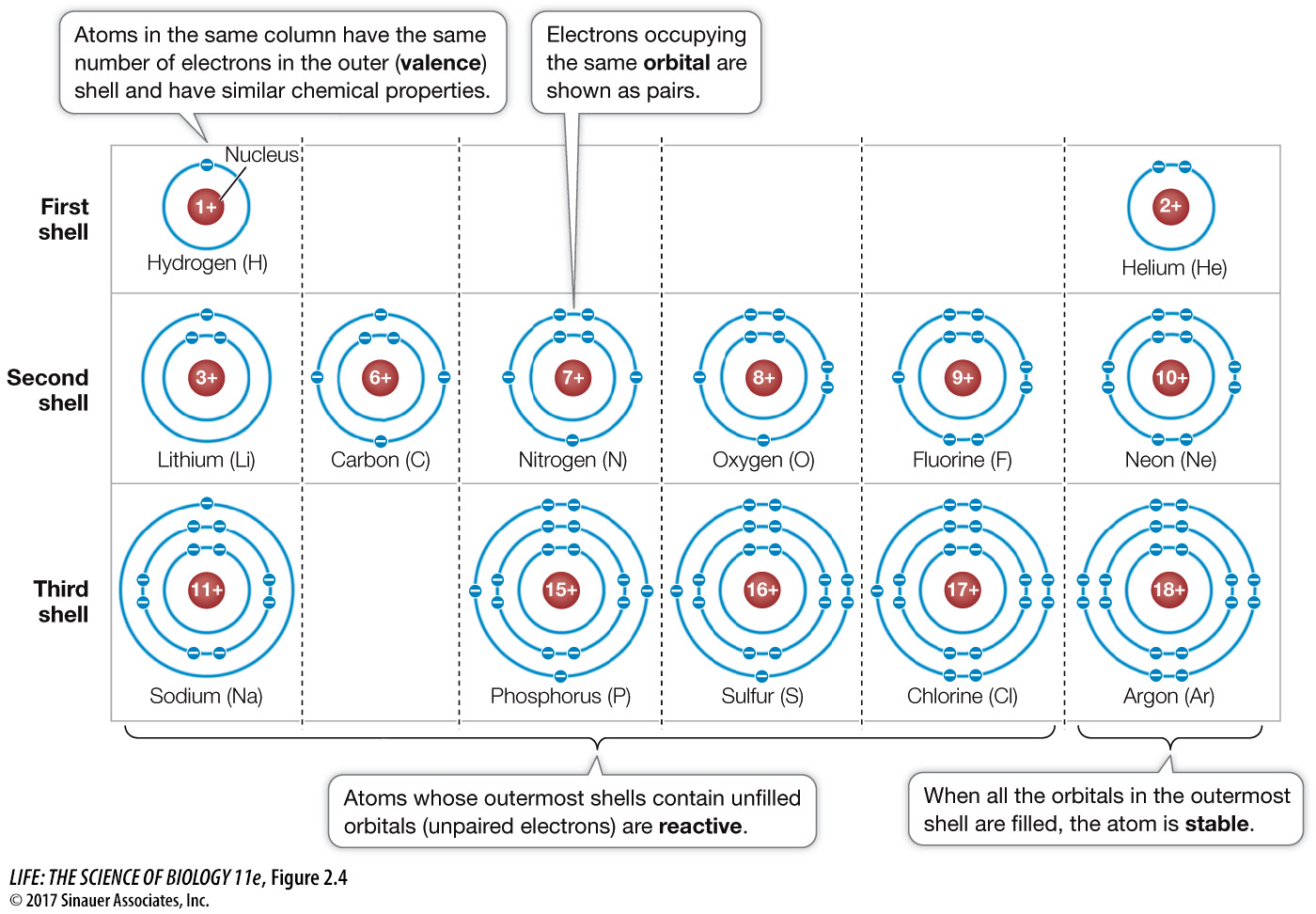
Hey there, curious minds! Ever wondered what makes up the teeny-tiny world inside an atom? We all know about electrons, right? Those speedy little guys whizzing around the nucleus. But have you ever thought about where they hang out? Well, they don't just randomly float around; they have their own special neighborhoods called electron subshells. And guess what? These subshells have a limited capacity, like a favorite cafe that can only fit so many people. Today, we’re going to dive into how we figure out just how many electrons can squeeze into these subshells, and why it's actually pretty darn cool!
Think of an atom like a miniature apartment building. The nucleus is the super central, really important lobby. The main energy levels, which we usually learn about first (like n=1, n=2, etc.), are like the different floors of this building. Each floor has a certain number of apartments. But within those apartments, there are even smaller, more specific rooms where the electrons really settle in. These are our electron subshells.
Now, these subshells aren't all the same. They have different shapes and therefore different capacities. It's like some rooms are spacious living rooms, while others are cozy little bedrooms. The main types of subshells we talk about are named after letters: s, p, d, and f. You might have heard of them before!
So, how do we know how many electrons can fit in each type of subshell?
It’s not just a random guess, thankfully! There are some fundamental rules in quantum mechanics that dictate this. The simplest way to think about it is based on the shape and the orientation of these subshells. Each subshell can be thought of as containing a certain number of "orbitals." An orbital is like a specific parking spot for electrons. And here's the kicker: each orbital can hold a maximum of two electrons.
But wait, why two? This is where the concept of electron "spin" comes in. Electrons are like tiny spinning tops. They can spin in one of two directions – either "up" or "down." To occupy the same orbital, two electrons have to have opposite spins. This is a fundamental principle called the Pauli Exclusion Principle. It’s like saying you can have two people in a single bed, but they have to be sleeping head-to-toe, not facing each other. This principle is super important for keeping everything stable in the atomic world.
Let's break it down by subshell type:
The 's' Subshell: The Simple Studio Apartment
The 's' subshell is the most basic of the bunch. It's spherical in shape, kind of like a perfectly round balloon. Because of its simple shape, it only has one orbital. Remember our rule? One orbital can hold two electrons. So, the 's' subshell can hold a maximum of 2 electrons. Easy peasy, right?
Imagine a single parking garage with just one spot. Only two cars, with opposite directions of approach (or spin!), can fit in there. That's your 's' subshell. It’s neat, tidy, and straightforward.
The 'p' Subshell: The Slightly More Complex Pad
Now, the 'p' subshell gets a little more interesting. Its shape is a bit like a dumbbell, with two lobes. And crucially, there are three different orientations for these dumbbell shapes in space. Think of them as being aligned along the x, y, and z axes of a room. So, you have three 'p' orbitals. If each orbital can hold two electrons, then what’s the capacity of the 'p' subshell?
Yep, you guessed it! 3 orbitals x 2 electrons per orbital = a maximum of 6 electrons. The 'p' subshell can house up to six electrons. This is where atoms start to get a bit more crowded and interesting.
It's like a small apartment building with three separate, identical studio apartments, each capable of holding two tenants. Total capacity: six residents!
The 'd' Subshell: The Deluxe Duplex
The 'd' subshell is where things get a bit fancier. These subshells have more complex shapes, often described as four-leaf clover-like, with one exception that looks like a dumbbell with a donut around it. Mathematically, it turns out there are five different orbitals within a 'd' subshell. Five orbitals!
So, following our trusty rule: 5 orbitals x 2 electrons per orbital = a maximum of 10 electrons. The 'd' subshell is quite spacious, capable of holding up to ten electrons. These are often found in the transition metals, which have some pretty cool and diverse chemical properties because of these extra electron slots.
Think of it as a set of five individual rooms in a house, each capable of housing a pair of roommates. That brings the total number of residents to ten.

The 'f' Subshell: The Really Roomy Residence
And finally, we have the 'f' subshell. These are the most complex in shape and the most spacious. The 'f' subshell has seven orbitals. Seven!
So, the math is getting bigger: 7 orbitals x 2 electrons per orbital = a maximum of 14 electrons. The 'f' subshell can hold a whopping 14 electrons. These are typically found in the lanthanides and actinides, which are elements at the bottom of the periodic table and are known for their unique behaviors.
Imagine a large mansion with seven guest suites, and each suite can accommodate two visitors. That’s 14 guests who can stay there!

Putting It All Together: Why Does This Matter?
So, we’ve got 's' with 2, 'p' with 6, 'd' with 10, and 'f' with 14. These numbers are fundamental to understanding the periodic table. The way elements are arranged, their properties, and how they interact with other elements all stem from how their electrons fill these subshells.
It's like filling up those apartment building floors (energy levels) with different types of rooms (subshells). Each floor can only hold so many people, and the types of rooms available on each floor dictate the building’s overall capacity and character. The 's' subshell is usually on the first floor, the 's' and 'p' on the second, 's', 'p', and 'd' on the third, and so on.
Understanding these capacities helps chemists and physicists predict how atoms will behave. Why does sodium have one electron in its outermost 's' subshell and is highly reactive? Because it's very eager to get rid of that one electron to achieve a more stable configuration, much like someone quickly leaving a crowded elevator to get more personal space. Why are noble gases so unreactive? Because their outermost electron shells and subshells are completely full, making them very stable and content, like people who have found their perfect, comfortable spot and don't want to move.
It’s a beautiful, intricate dance of electrons filling these quantized spaces. So, the next time you see the periodic table, remember that beneath those element names and symbols lies this fascinating world of subshells, each with its own distinct capacity, all governed by the elegant rules of quantum mechanics. Pretty cool, right?
