Calculate Zeff For A Valence Electron In An Oxygen Atom
Hey there, science curious folks! Ever stop and wonder what makes the world tick? It’s all about these tiny building blocks called atoms, and inside those atoms, there are even tinier bits doing all the heavy lifting. Today, we’re going to chat about something super cool that happens inside an oxygen atom, specifically for its outermost electron. Think of it like the shy kid on the edge of the playground, or the last cookie in the jar – that’s our valence electron!
Now, the fancy term we’re going to tackle is Zeff, which is short for effective nuclear charge. Don't let the big words scare you! Imagine your nucleus, the super-dense center of the atom, is like a magnet. It’s got a positive charge, and it loves to pull on those negatively charged electrons orbiting around it. Zeff is basically how much of that nuclear magnet's pull our specific valence electron actually feels.
Why should you even care about this, you ask? Well, it’s like understanding why your favorite cookie crumbles just right or why your cat purrs. It explains a whole lot of the stuff around us! This Zeff is a big reason why oxygen is so good at, well, being oxygen – it's a chatty, reactive element, always looking to make friends (or rather, grab some electrons!). It dictates how atoms bond, how they behave, and ultimately, how the universe is pieced together. Pretty neat, right?
Let's Meet Our Oxygen Atom
So, picture an oxygen atom. It’s a relatively small guy, like a tiny, energetic hummingbird. Its nucleus is buzzing with positive energy. Around this nucleus, electrons are zipping around in different energy shells, like little planets in orbit. Now, oxygen has a total of 8 electrons. The first two are chilling in the innermost shell, practically glued to the nucleus. They’re like the super-organized, always-on-time folks who get to the party early and never leave the front row.
The remaining 6 electrons are in the outer shell. These are our valence electrons. They’re the ones that get to interact with the outside world, the ones that decide if oxygen is going to be friends with a hydrogen atom (hello, water!) or a carbon atom (hi, carbon dioxide!). These valence electrons are the rockstars of chemical reactions. They’re a bit more laid-back, a bit more exposed to the elements, and they feel the nuclear pull, but not as strongly as their inner siblings.
The "Pull" and the "Screening"
Here’s where Zeff comes into play. The nucleus has a strong positive charge, let’s say it’s like a super-duper strong magnet. If there were only our one valence electron and no other electrons, it would feel the full might of that magnet. But, we’ve got those other electrons hanging around, don’t we? They’re like a bunch of little shields, or maybe a flock of noisy pigeons, getting between the nucleus and our valence electron.
These inner electrons, those first two in the cozy inner shell, are particularly good at shielding or screening the valence electron. They're closer to the nucleus and absorb some of that positive pull, like a thick coat blocking the sun’s rays. So, our valence electron doesn’t feel the full nuclear charge. It feels a reduced, or effective, charge. That’s our Zeff!
A Little Story About the "Shields"
Imagine you’re trying to hear your best friend whispering a secret across a crowded room. Your friend is the nucleus, and the secret is the pull on the valence electron. If there were no one else around, you’d hear them perfectly. But, if there are lots of people talking, laughing, and bumping into each other between you and your friend – those are the inner electrons – it’s much harder to hear the whisper. You still hear something, but it’s muffled, less intense. That muffled sound is like the Zeff. The inner electrons are acting like a noisy crowd, making the direct communication (the full nuclear pull) a bit weaker.
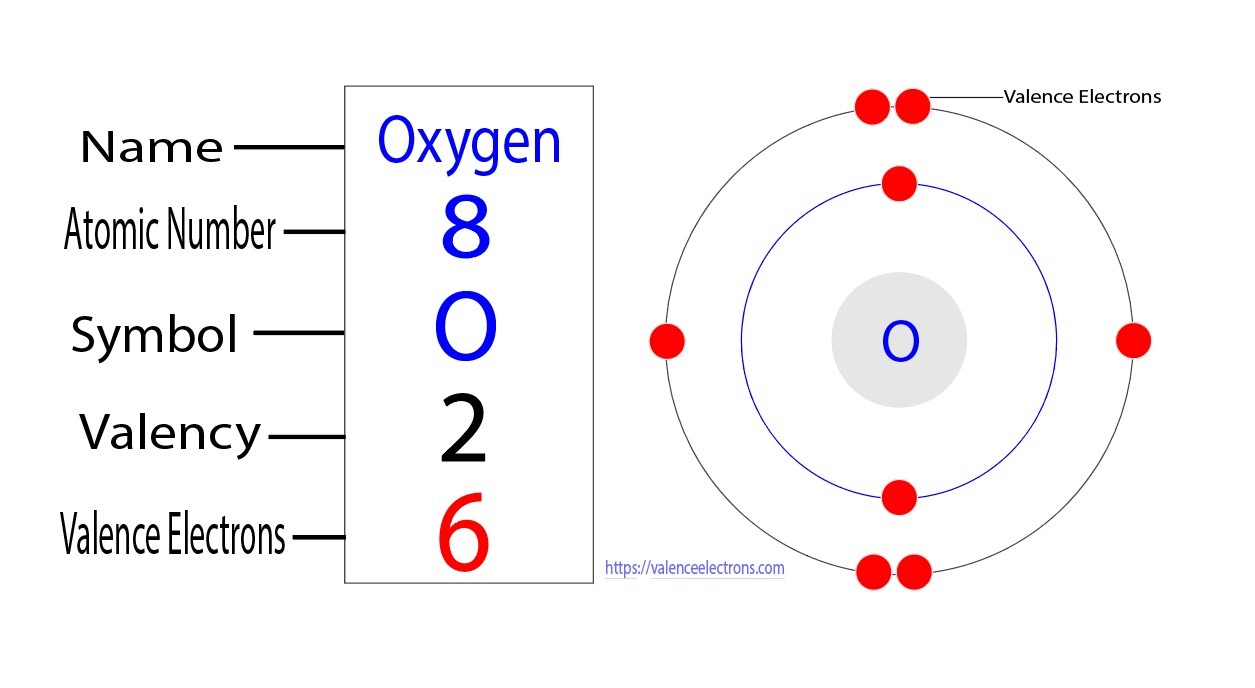
Calculating the "Rough" Zeff (No need for calculators!)
We don’t need to get bogged down in super-complex math here, but it’s helpful to understand the basic idea. The total positive charge of the nucleus comes from the number of protons it has. For oxygen, that’s 8 protons. So, you might think the valence electron feels a charge of +8. But remember those inner electrons acting like shields?
We kind of subtract the "screening effect" of the inner electrons. Each inner electron reduces the pull on the outer ones. So, for oxygen, the nucleus has +8 charge. The two inner electrons are really good at shielding, so they "cancel out" some of that pull. It’s like saying, "Okay nucleus, you have 8 positive friends, but 2 of them are really good at hugging and keeping others away from the main attraction."

So, a simplified way to think about Zeff is the nuclear charge minus the number of inner electrons that effectively shield the valence electrons. For oxygen, with its 8 protons and 2 inner electrons, the simplified Zeff for a valence electron is roughly 8 - 2 = 6. This means our valence electron feels an effective pull equivalent to a nucleus with a +6 charge, not the full +8.
Why Zeff Matters to You (Yes, YOU!)
This Zeff concept is the hidden architect of many everyday phenomena! It’s why water (H2O) is such a fantastic solvent, allowing us to dissolve sugar in our tea or salt in our cooking. Oxygen’s relatively high Zeff makes it electronegative – meaning it has a strong desire to snatch electrons from other atoms. This drive is what makes water molecules have a slight positive and negative end, making them great at dissolving other polar substances.
Think about rusting! Iron atoms have their own Zeff values, and when they interact with oxygen, oxygen’s strong electron-grabbing ability (thanks to its Zeff) plays a huge role in that flaky, reddish transformation. It’s the difference between a shiny new bike and a well-loved, slightly weathered one.
Even breathing! The oxygen you inhale is readily accepted by your body's molecules because of oxygen's chemical reactivity, which is directly linked to its Zeff. It’s like oxygen is saying, "Here I am, ready to form bonds and help you do all sorts of amazing biological things!"
The Bottom Line
So, the next time you’re marveling at the taste of your favorite drink, the strength of a piece of metal, or just the simple act of breathing, you can nod to yourself and think about Zeff. It’s a fundamental concept that explains why atoms behave the way they do, and why the world around us is so wonderfully diverse and functional.
It’s not just some abstract number in a textbook; it’s the silent conductor of the chemical symphony that makes everything happen. It’s the reason oxygen is the life-giving element it is, always ready to engage and transform. So, a little bit of math and a lot of chemistry, all wrapped up in the everyday magic of atoms!
