Calculate The Ph Of A Solution Prepared By Dissolving
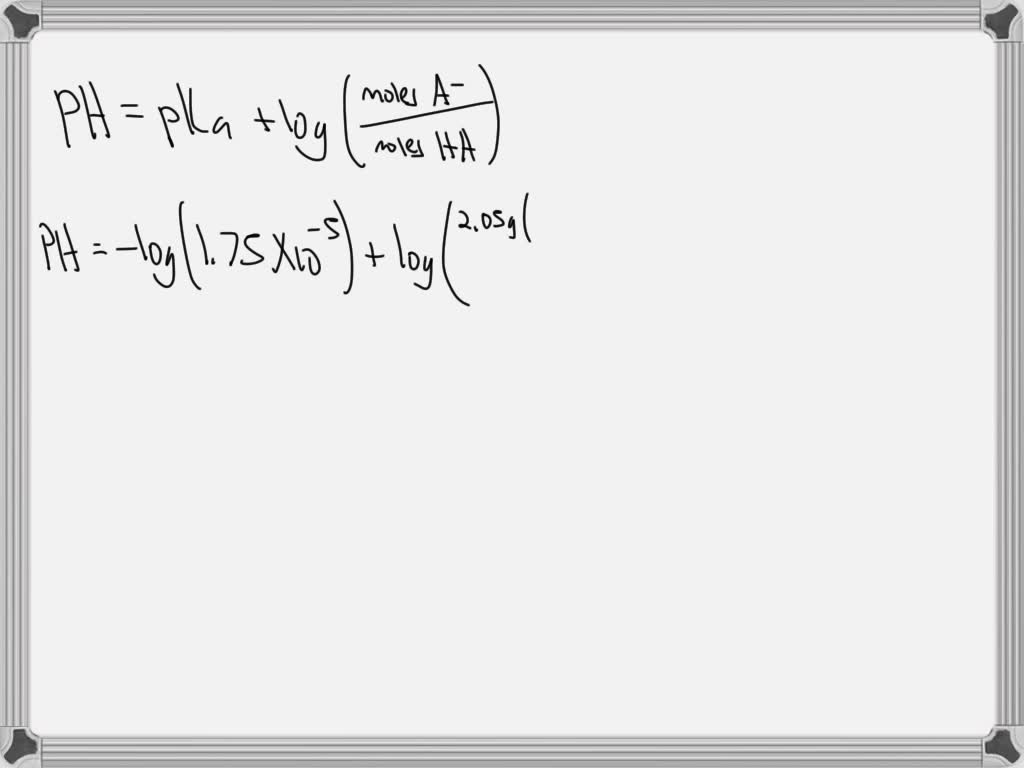
Hey there, science enthusiasts and curious minds! Ever wondered what happens when you take a sprinkle of something solid and plop it into water? It's like magic, right? Suddenly, you've got a whole new liquid with its own personality. And that personality, in the world of chemistry, is often described by something super cool called pH.
Think of pH like a secret code for how "sour" or "slippery" a solution is. It's not just about taste, though! pH is everywhere. It’s in your morning juice, the soap you use, even the soil in your garden. And figuring out the pH of a solution you've just whipped up? That's where things get really fun and, dare I say, a little bit like detective work!
Imagine you're a culinary wizard in your kitchen. You're not just mixing ingredients; you're creating something new. When you dissolve, say, a dash of baking soda in water, you're not just making fizzy water. You're creating a whole new environment. And the pH tells you all about that environment. Is it more on the acidic side, like a zesty lemon? Or is it more on the basic side, feeling a bit more slippery, like some soaps?
The exciting part is when you start to calculate this pH. It sounds super technical, and yes, there are some numbers involved. But don't let that scare you off! It's like learning the secret handshake to a cool club. You're not just memorizing formulas; you're unlocking the why behind the fizz, the reason for the tingle.
Let's talk about what makes this whole process of dissolving and calculating pH so special. It’s the transformation! You start with two simple things: a solid and a liquid. Then, poof! You have a solution. And this solution has a specific character, a specific pH value that tells you about its chemical nature. It's like observing a caterpillar turn into a butterfly, but on a microscopic, molecular level. How cool is that?

When you're calculating the pH, you're essentially measuring the concentration of hydrogen ions (H+) in your solution. These tiny little guys are the real MVPs of pH. More H+ means a lower pH (more acidic), and fewer H+ means a higher pH (more basic). It's a delicate balance, and figuring it out is like solving a puzzle.
Think about it: you've got a beaker, a spoonful of something, and some water. You mix them. Then, with a bit of knowledge and a few calculations, you can pinpoint exactly where your solution sits on the pH scale, which famously runs from 0 to 14. Numbers 0-6 are acidic, 7 is neutral (like pure water!), and 8-14 are basic or alkaline.

And the beauty of it? You can do this with so many different things! Dissolve some table salt? What's its pH? How about some vinegar? You already know that's acidic, but calculating the exact pH is still satisfying. What about something you've never even thought about dissolving before? That’s where the adventure truly begins.
The process of calculating the pH of a dissolved solution isn't just about getting a number. It's about understanding the invisible world happening in your container. It's about appreciating the chemical dance that occurs when substances meet. It’s like reading a secret message that the solution is sending you.
Why is it so engaging? Because it’s tangible! You can see the solid disappear. You can potentially feel the change if it’s a strong acid or base (though always be safe!). And then, with a bit of math, you can quantify that change. You can assign a number to the "personality" of your new creation. It's a bridge between the visible and the invisible, the qualitative and the quantitative.
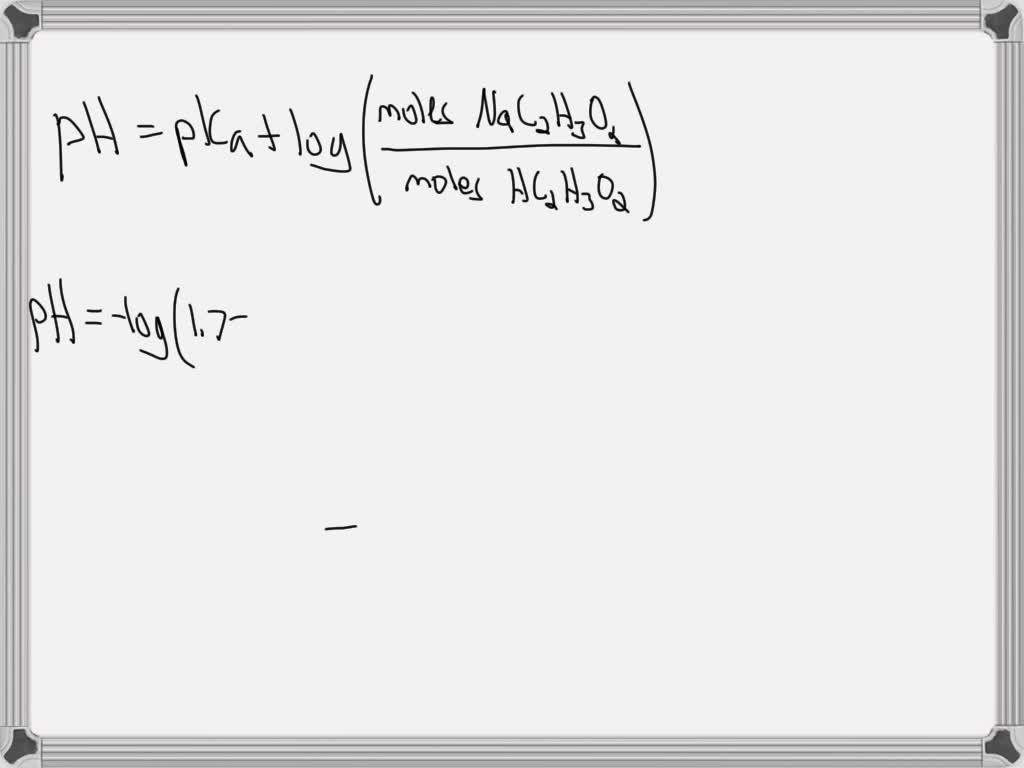
And the tools you might use? Sometimes it's as simple as a pH meter, a little gadget that gives you a direct reading. Other times, it involves using the concentration of your dissolved substance and a handy little equation. This equation often involves a mathematical operation called a logarithm. Don't let that word intimidate you! Think of it as a special way to compress a huge range of numbers into a more manageable scale – exactly what pH is all about!
Imagine your solution is like a story. The pH is the plot twist that reveals its true nature!
SOLVED: Calculate the pH of a solution prepared by dissolving 1.15 g of
What makes it special is the immediate sense of accomplishment. You’ve taken a bit of chemical knowledge, applied it to something you physically created, and arrived at a meaningful answer. It’s empowering! You’re not just passively observing; you’re actively participating in understanding the chemical world around you.
So, next time you're curious about what happens when you dissolve something, consider digging a little deeper. Explore the concept of pH. Learn how to calculate it for your own simple solutions. You might find yourself utterly captivated by the hidden properties of everyday substances. It's a journey into the heart of chemistry, one dissolved particle at a time, and it’s more fun than you might think!
It’s about demystifying the science that’s all around us. It’s about taking a seemingly complex topic and making it accessible, even enjoyable. So, grab some water, pick a (safe!) substance, and get ready to uncover the secret pH life of your very own creation!

