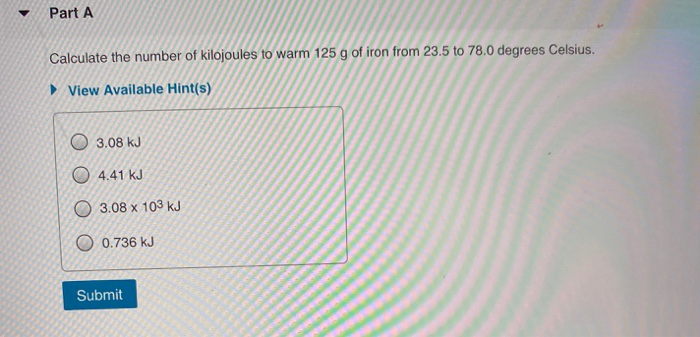
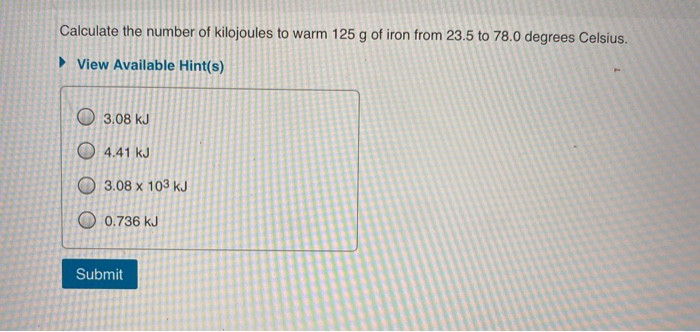
Calculate The Number Of Kilojoules To Warm 125g Of Iron

Ever wondered what makes your morning coffee stay warm for so long, or why a metal spoon left in the sun feels like it's about to melt your hand? It all boils down to heat, and how different materials soak it up and release it. Today, we’re diving into a super cool (and surprisingly fun!) calculation that unlocks a little secret of the universe: figuring out exactly how much energy it takes to warm up a specific chunk of iron. It might sound a bit technical, but trust us, it’s more like a neat party trick for your brain than a grueling math exam. Plus, understanding this helps us appreciate everything from the design of cooking pans to the incredible temperatures found in industrial processes.
The Cozy Comfort of Kilojoules
So, why should you care about calculating the kilojoules needed to warm up some iron? Well, think of kilojoules as tiny little packets of energy. When we talk about warming something up, we’re essentially talking about adding these energy packets to it. It’s like feeding a hungry object until it reaches its desired temperature. This concept is incredibly useful in so many areas of our lives. For example, when engineers design engines, they need to know how much heat will be generated and how it will be absorbed or dissipated. Chefs rely on the principles of heat transfer to get that perfect sear on a steak or to ensure delicate pastries bake evenly. Even when you’re bundling up in winter clothes, you’re essentially creating an insulating barrier to *retain the heat your body generates, measured in those same kilojoules!
The benefits of understanding these calculations are vast. On a practical level, it helps us make informed decisions. If you're choosing cookware, knowing how quickly different metals heat up can make a big difference in your cooking experience. For those interested in science and technology, this is a fundamental concept that underpins many advanced fields, from material science to thermodynamics. It’s the science behind why a cast-iron skillet holds its heat so well, making it a favorite for many home cooks, while a thinner aluminum pan might heat up faster but also cool down quicker. This knowledge empowers us to better understand and interact with the physical world around us.
Unlocking the Iron's Warmth: A Step-by-Step Adventure
Let's get to the exciting part: calculating the kilojoules to warm up a specific amount of iron. Imagine we have 125 grams of this robust metal. Our goal is to figure out precisely how many kilojoules of energy we need to pour into it to nudge its temperature up by a certain amount. This isn’t just a random number; it’s a calculated value based on the material's properties. For iron, a key player in this calculation is its specific heat capacity. Think of this as iron's personal "energy absorption rate." Every substance has its own unique specific heat capacity, and for iron, it’s approximately 0.449 J/g°C. This means it takes 0.449 joules of energy to raise the temperature of 1 gram of iron by 1 degree Celsius.
Now, we need to introduce the temperature change. Let's say we want to warm our 125g of iron by a nice, cozy 50°C. To make our lives easier, we’ll convert our joules to kilojoules later, as kilojoules are often used for larger energy amounts. The formula we’ll use is elegantly simple, a cornerstone of thermal physics:

Q = mcΔT
Where:
- Q represents the amount of heat energy (in joules).
- m is the mass of the substance (in grams).
- c is the specific heat capacity of the substance (in J/g°C).
- ΔT (delta T) is the change in temperature (in °C).
So, let's plug in our numbers! We have:
- Mass (m) = 125 g
- Specific heat capacity of iron (c) = 0.449 J/g°C
- Change in temperature (ΔT) = 50°C
Substituting these values into our formula:
+Heat+or+energy+can+be+in+joules%2C+calories%2C+kilocalories%2C+or+kilojoules.+The+SI+unit+is+the+_________..jpg)
Q = (125 g) * (0.449 J/g°C) * (50°C)
Let's break down the multiplication. First, multiplying the mass by the specific heat capacity:
125 g * 0.449 J/g°C = 56.125 J/°C
This intermediate result tells us how many joules are needed to raise the temperature of our entire 125g sample by a single degree Celsius. Now, we multiply this by our desired temperature change:
56.125 J/°C * 50°C = 2806.25 J
And there we have it! The total amount of energy in joules required to warm 125g of iron by 50°C is 2806.25 joules. But remember, we often talk about these larger energy amounts in kilojoules. To convert joules to kilojoules, we simply divide by 1000 (since "kilo" means 1000).
2806.25 J / 1000 = 2.80625 kJ
So, it takes approximately 2.81 kilojoules to give our 125g of iron a nice warm boost of 50°C. Isn’t that neat? It’s a tangible measure of energy, all thanks to a simple formula and the inherent properties of the material itself. Whether you’re marveling at a blacksmith’s forge or understanding why your car engine gets hot, this fundamental calculation is at play, making the world of thermodynamics accessible and, dare we say, rather exciting!
