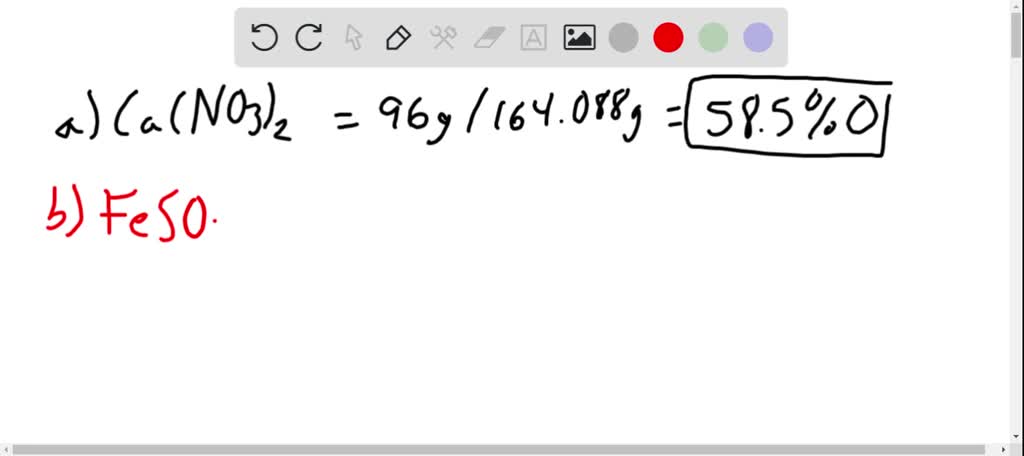
Calculate The Molecular Mass Of Calcium Nitrate

So, you've decided to tackle the mystical world of chemistry, huh? Good for you! It’s a wild ride, and sometimes, it feels like scientists are speaking a whole different language. But fear not, brave explorer! Today, we're going to casually dip our toes into a little calculation. Nothing scary, promise! We're talking about the molecular mass of calcium nitrate. Sounds fancy, right? It's basically just a fancy way of saying "how much a tiny bit of this stuff weighs" if we were to count it out by its building blocks.
Now, before you start picturing a miniature scale in your kitchen, let's simplify. Think of it like a recipe. You need ingredients, and each ingredient has a certain weight. Molecular mass is kind of like the total weight of your finished dish, based on how much of each ingredient you used. And our main star today is calcium nitrate. It’s the stuff that helps plants grow big and strong, so it’s actually pretty cool.
It's like trying to figure out the weight of a LEGO castle! You count the bricks.
To figure out the molecular mass of calcium nitrate, we need to know what it's made of. It's not just one thing, you see. It’s a team effort! The name itself gives us clues. We’ve got calcium. That’s the ‘Ca’ part. And then we have nitrate. That's the tricky part because nitrate is actually made of two different elements: nitrogen (N) and oxygen (O). And there’s more than one nitrate in our calcium nitrate.
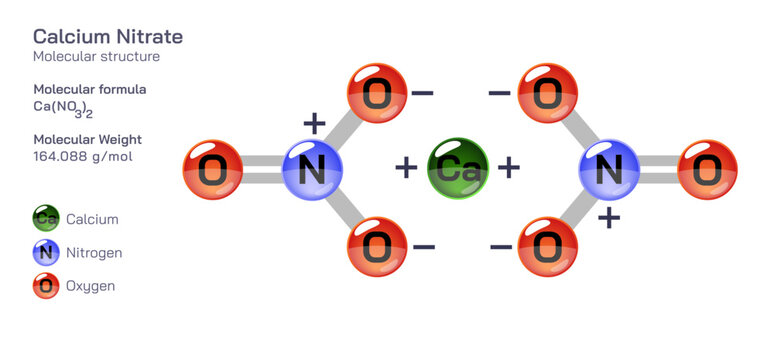
This is where things get a tiny bit like a detective story. For calcium nitrate, the chemical formula is Ca(NO₃)₂. See that little ‘2’ at the end, outside the parentheses? That’s super important! It means we have two of those nitrate groups for every one calcium atom. So, it's not just one nitrogen and one oxygen per nitrate; it's actually one nitrogen and three oxygen atoms inside each nitrate group. And since we have two nitrate groups, we’ve got double the trouble… I mean, atoms!

So, to get our total weight, we need to look up the atomic mass of each element. These are numbers you can find on something called the periodic table. It’s like a big chart of all the elements, and each element has its own little box with its symbol and its atomic mass. Don't worry about memorizing it; nobody actually does that! We just glance it up when we need it. Think of it like checking a cookbook for the amount of flour you need.
For calcium (Ca), its atomic mass is roughly 40.08. Let's just round it to 40 for simplicity in our heads, okay? Chemists do this all the time. They're not always about hyper-precision unless they absolutely have to be. For nitrogen (N), it's about 14.01. We can think of that as 14. And for oxygen (O), it's around 16.00. Easy peasy. So, 40 for calcium, 14 for nitrogen, and 16 for oxygen. Got it?
Now, let's put on our math hats, but keep them stylish, like a fedora. We have one calcium atom. So, that’s 1 * 40 = 40. Then we have our nitrate groups. Remember, there are two of them, and each one has one nitrogen and three oxygen atoms. So, for the nitrogen inside those two nitrates, we have 2 * 14 = 28. And for the oxygen, since each nitrate has three, and we have two nitrates, that's 2 * 3 = 6 oxygen atoms in total. So, 6 * 16 = 96.
Now, we just add up all these bits and pieces. We have our calcium weight (40), our nitrogen weight from both nitrates (28), and our oxygen weight from both nitrates (96). So, 40 + 28 + 96. Let’s do a little dance with the numbers. 40 + 28 is 68. And 68 + 96… well, if you think of 96 as 100 minus 4, it's easier! So, 68 + 100 = 168, and then take away 4. That gives us 164. So, the molecular mass of calcium nitrate is roughly 164!
There you have it! We just calculated the molecular mass of calcium nitrate. It's not some dark magic, is it? It’s just counting and adding. It’s like figuring out how much your grocery bill will be before you get to the checkout. A little bit of anticipation, a little bit of math, and a whole lot of understanding what you’re actually buying.
And honestly, I think it’s kind of a superpower. Being able to look at a chemical formula and know, even roughly, how much it weighs. It’s like having a secret decoder ring. Most people just see letters and numbers, but you? You see the building blocks and their weights. It’s a small victory, but hey, I’ll take it! It’s an unpopular opinion, perhaps, but I think calculating molecular masses is secretly kind of fun. Don't you?
