Calculate The Molarities Of Hc2h3o2 And Nac2h3o2
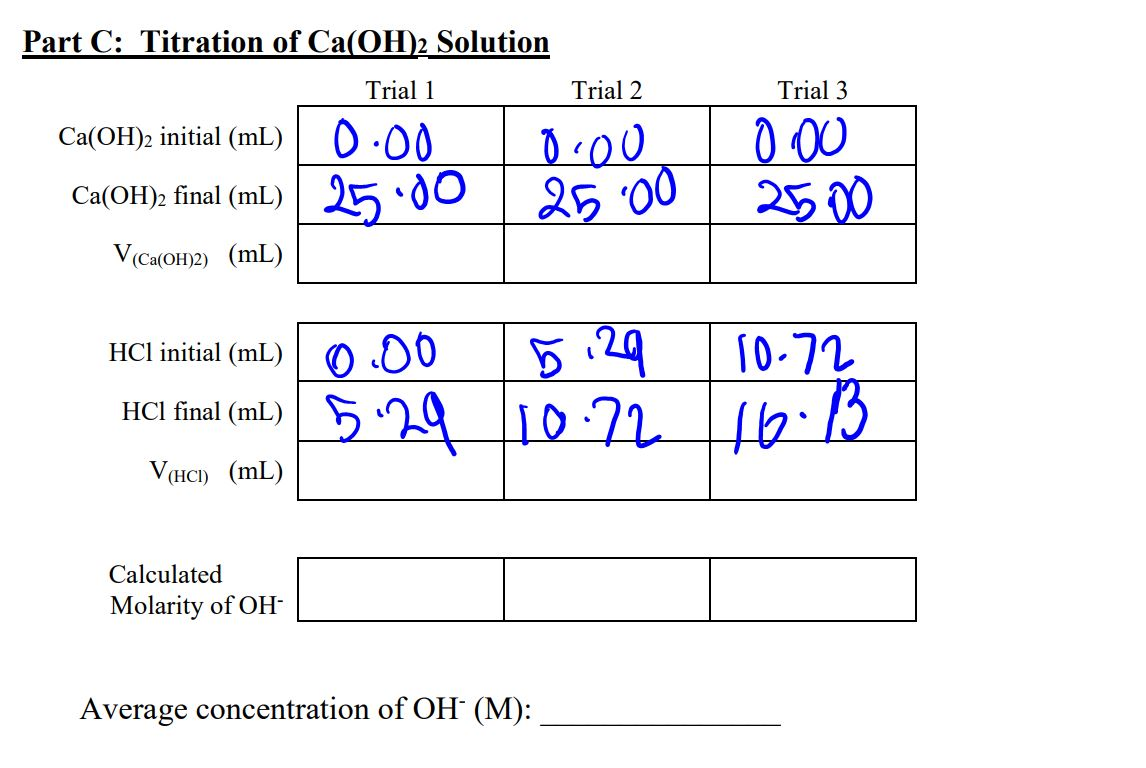
Okay, so picture this: I'm chilling, you know, doing my usual thing, which sometimes involves staring blankly at my chemistry textbook and questioning all my life choices. Suddenly, a thought pops into my head, a brilliant thought, if I do say so myself. It's about these things called acids acids and their buddies, the salts salts. Specifically, I was thinking about acetic acid, the stuff that makes vinegar smell… well, like vinegar. And its friend, sodium acetate. My brain, in its infinite wisdom, decided it was the perfect time to ponder, "Hey, how do you figure out how much of this stuff is actually in a solution?"
And that, my friends, is how we stumble upon the glorious, sometimes terrifying, world of molarity. Don't let the fancy word scare you. It's just a way of saying "how concentrated something is." Think of it like figuring out how many sprinkles are on a cupcake. You could say "a lot of sprinkles" or you could be all scientific and say "X number of sprinkles per square inch of frosting." Molarity is the science-y version for chemicals.
So, my initial existential crisis about life choices morphed into a very specific, albeit slightly less dramatic, question: How do we calculate the molarities of HC2H3O2 and NaC2H3O2? And why should you even care? Well, buckle up, buttercup, because it's more interesting than it sounds, I promise. Or at least, I'll try to make it interesting. 😉
The Players: Acetic Acid and Sodium Acetate
First, let's meet our stars. We've got HC2H3O2, which is acetic acid. You know, vinegar? That tangy stuff that makes your eyes water and your taste buds do a little dance? Yep, that's it. It's a weak acid. What does "weak" mean in chemistry terms? It means it doesn't completely let go of its hydrogen ion (H+) when you dissolve it in water. It's a bit… reluctant. Like me before my first cup of coffee.
Then we have NaC2H3O2, sodium acetate. This is the salt of acetic acid. Think of it as the more stable, less angsty offspring of acetic acid and a strong base (like sodium hydroxide). When you dissolve sodium acetate in water, it pretty much breaks apart completely into its ions: Na+ and C2H3O2-. The acetate ion (C2H3O2-) is the conjugate base of acetic acid. They're like family, but in a chemical sense.
So, you've got your weak acid (HC2H3O2) and its conjugate base (C2H3O2-, from NaC2H3O2). This dynamic duo is super important in chemistry, especially when you start talking about buffers. Buffers are like the steady hand in a chaotic chemical reaction, helping to maintain a stable pH. Ever wondered how your blood can stay at a pretty consistent pH even when you eat a lemon? Buffers, my friend!
Molarity: The "How Much?" Question
Now, back to molarity. In the most basic sense, molarity (often symbolized by a capital 'M') is defined as the number of moles of solute divided by the liters of solution.
Mathematically, it looks like this:
Molarity (M) = Moles of Solute / Liters of Solution
This is the golden rule. This is what you'll be coming back to, time and time again. So, what are these "moles" and why do we need them?
Moles are basically a chemist's way of counting. Instead of saying "one gazillion molecules of water," we say "one mole of water." It's a convenient unit because it relates the microscopic world of atoms and molecules to the macroscopic world we can actually measure (like grams and liters). One mole of any substance contains approximately 6.022 x 10^23 particles (atoms, molecules, ions, you name it). This is Avogadro's number, and it’s pretty darn important. Think of it as a super-duper-sized dozen.
So, to calculate molarity, you need two key pieces of information:

- How much of your substance you have, expressed in moles.
- The total volume of the solution it's dissolved in, expressed in liters.
If you have one of those but not the other, don't despair! There are usually ways to convert.
Calculating Moles: The Gateway to Molarity
Let's say you have a certain amount of acetic acid or sodium acetate, but it's in grams. How do you get to moles? This is where the molar mass comes in. The molar mass of a compound is the mass of one mole of that compound, expressed in grams per mole (g/mol).
To find the molar mass, you just look at the periodic table and add up the atomic masses of all the atoms in the chemical formula. For our friends:
Acetic Acid (HC2H3O2):
- Carbon (C): 2 atoms x 12.01 g/mol = 24.02 g/mol
- Hydrogen (H): 4 atoms x 1.01 g/mol = 4.04 g/mol
- Oxygen (O): 2 atoms x 16.00 g/mol = 32.00 g/mol
- Total Molar Mass of HC2H3O2 ≈ 60.06 g/mol
So, if you have 60.06 grams of acetic acid, you have approximately 1 mole of it. Easy peasy, right?
Sodium Acetate (NaC2H3O2):
- Sodium (Na): 1 atom x 22.99 g/mol = 22.99 g/mol
- Carbon (C): 2 atoms x 12.01 g/mol = 24.02 g/mol
- Hydrogen (H): 3 atoms x 1.01 g/mol = 3.03 g/mol
- Oxygen (O): 2 atoms x 16.00 g/mol = 32.00 g/mol
- Total Molar Mass of NaC2H3O2 ≈ 82.04 g/mol
Similarly, 82.04 grams of sodium acetate is about 1 mole. See? You're basically a chemist now. I knew you had it in you.
Once you have the molar mass, converting grams to moles is a simple division:
Moles = Mass (in grams) / Molar Mass (in g/mol)
Putting It All Together: Calculating Molarities
Okay, so let's get to the main event! How do we actually calculate the molarities? This usually happens in one of two scenarios:
Scenario 1: You're Making the Solution
This is the fun part where you're the chemist, measuring out ingredients and creating your own solutions. Let's say you want to make a 0.5 M solution of acetic acid. You decide you need to make 250 mL (which is 0.250 L, remember to convert mL to L by dividing by 1000!) of it.
Here's what you do:
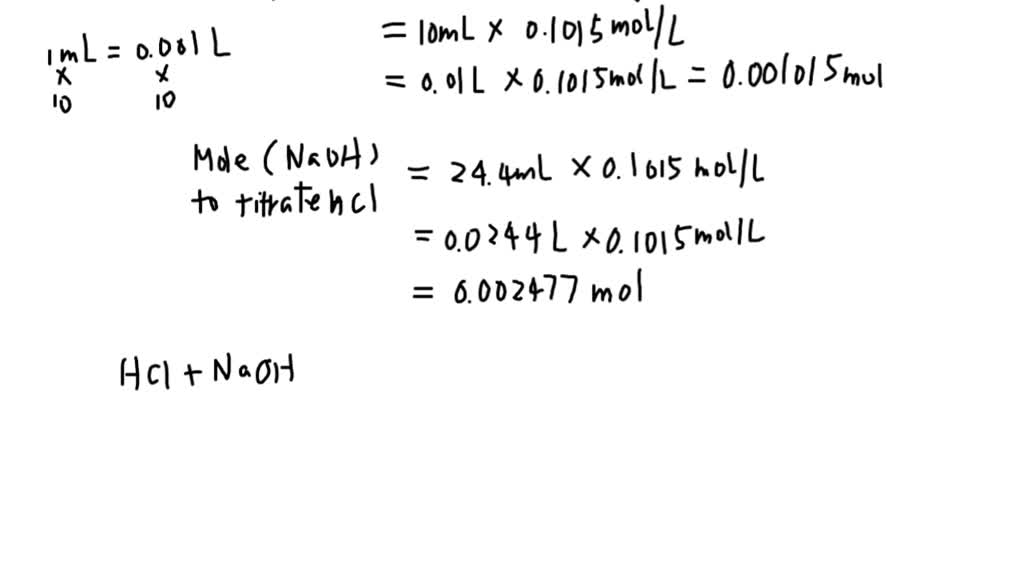
- Figure out the desired moles:
- Convert moles to grams:
- The procedure:
You know Molarity = Moles / Liters. So, Moles = Molarity x Liters.
Moles of HC2H3O2 = 0.5 mol/L * 0.250 L = 0.125 moles
You'll need to weigh out your acetic acid, so you need grams. Grams = Moles x Molar Mass.
Grams of HC2H3O2 = 0.125 moles * 60.06 g/mol ≈ 7.51 grams
So, you'd carefully weigh out approximately 7.51 grams of acetic acid and dissolve it in enough water to make a final volume of 250 mL. Voilà! You've made a 0.5 M acetic acid solution. High five!
You can do the exact same thing for sodium acetate. If you wanted to make a 0.2 M solution of sodium acetate in 500 mL (0.500 L) of water:
- Desired moles:
- Convert moles to grams:
- The procedure:
Moles of NaC2H3O2 = 0.2 mol/L * 0.500 L = 0.100 moles
Grams of NaC2H3O2 = 0.100 moles * 82.04 g/mol ≈ 8.20 grams
Weigh out 8.20 grams of sodium acetate and dissolve it in enough water to reach a final volume of 500 mL. Bam! You've got your sodium acetate solution.
See? Making solutions with a specific molarity is all about working backward from your desired concentration and volume.
Scenario 2: You Have a Solution and Want to Know Its Molarity
This is where you might be given the mass of solute dissolved in a specific volume of solution. Or, you might be given the concentration of a stock solution and need to figure out the molarity after dilution. Let's stick to the simpler case for now.
Suppose you have a bottle that contains 25 grams of acetic acid dissolved in a total volume of 1.0 Liter of solution. What is its molarity?
- Convert grams of solute to moles:
- Calculate molarity:
Moles of HC2H3O2 = 25 g / 60.06 g/mol ≈ 0.416 moles
Molarity = Moles of Solute / Liters of Solution
Molarity = 0.416 moles / 1.0 L ≈ 0.416 M
So, that bottle is approximately a 0.416 M acetic acid solution. You're basically a detective now, uncovering hidden chemical truths!
Let's try another one with sodium acetate. You dissolve 15.0 grams of sodium acetate in enough water to make 200 mL (0.200 L) of solution. What's the molarity?
- Convert grams of solute to moles:
- Calculate molarity:
Moles of NaC2H3O2 = 15.0 g / 82.04 g/mol ≈ 0.183 moles
Molarity = Moles of Solute / Liters of Solution
Molarity = 0.183 moles / 0.200 L ≈ 0.915 M

And there you have it! A 0.915 M sodium acetate solution.
What About the "Acid" Part in Acetic Acid?
Now, a quick detour. When we talk about the molarity of acetic acid (HC2H3O2), we're usually referring to the total concentration of the acetic acid molecule. Remember how I said it's a weak acid and doesn't fully dissociate?
HC2H3O2 (aq) ⇌ H+ (aq) + C2H3O2- (aq)
This means that in a solution of acetic acid, you'll have HC2H3O2 molecules, as well as some H+ ions and C2H3O2- ions. If you're asked for the molarity of HC2H3O2, you're typically looking for the concentration of the undissociated molecules plus the dissociated acetate ions (because they came from the original HC2H3O2). It's the total amount of the "acetic acid stuff" that was put in.
When you calculate the molarity of HC2H3O2 from its mass, you are indeed calculating the molarity of the total acetic species, which includes both the intact molecules and the dissociated ions. It's like counting all the pieces of pizza, whether they're whole slices or smaller crumbs.
For sodium acetate (NaC2H3O2), it's a bit more straightforward. When it dissolves, it completely breaks into Na+ and C2H3O2-. So, if you make a 0.5 M solution of NaC2H3O2, you effectively have 0.5 M of Na+ ions and 0.5 M of C2H3O2- ions. We usually refer to the molarity of the formula unit, which in this case represents the combined entities that make up the salt.
Why Does This Even Matter? (Besides Buffers!)
You might be thinking, "Okay, this is mildly interesting, but why do I need to be able to calculate this?" Well, beyond the world of buffers (which are super cool, by the way!), understanding molarity is fundamental to so many chemical processes:
- Stoichiometry: When you do reactions, knowing the molarities of your reactants is crucial for predicting how much product you'll get. It's all about the mole ratios!
- Titrations: This is a common lab technique where you use a solution of known molarity to determine the concentration of an unknown solution. Guess what you need to know to do that? Molarity!
- Solution Preparation: As we saw, if you need a specific concentration for an experiment, you need to know how to calculate the amounts of stuff to mix.
- Biological Systems: Even in your body, concentrations of various molecules are expressed in molarity. It helps understand how things are working at a cellular level.
So, while it might seem like just a bunch of numbers and formulas now, mastering molarity is like getting the key to unlock a whole lot more chemistry. It's a building block, a foundation. And honestly, once you get the hang of it, it's not that intimidating. It's just a systematic way of thinking about how much of something is in a given space.
Next time you're looking at a vinegar bottle or a box of baking soda (which is sodium bicarbonate, by the way – different salt, same principle!), you'll have a little more appreciation for the chemistry going on. You can even mentally calculate the molarity of the acetic acid in your vinegar if you know its density and percentage by mass! (Okay, that’s a bit advanced, but you get the idea!)
So, keep practicing, don't be afraid to grab your calculator, and remember that even the most complex chemical concepts can be broken down into manageable steps. And who knows, you might even start to enjoy it. Or at least tolerate it. That’s progress, right?
