Calculate The Molar Mass Of Sodium Bicarbonate

Hey there, science curious friends! Ever feel like those big scientific words are just… a little too intimidating? Like they belong in a dusty old textbook and have zero to do with your actual, fabulous life? Well, buckle up, buttercups, because today we're diving into something super cool, and guess what? It’s all about baking soda! Yep, that humble white powder you probably have lurking in your pantry right now.
We're going to calculate the molar mass of sodium bicarbonate. Sounds fancy, right? But trust me, it's more like a fun little puzzle that unlocks a secret superpower for understanding how the world around us works. And who doesn't want a little more superpower in their lives? It's not about memorizing a bunch of numbers; it's about appreciating the tiny, invisible building blocks that make everything possible.
Unlocking the Secrets of Baking Soda
So, what exactly is sodium bicarbonate? It’s that magical stuff that makes your cookies rise and your sinks sparkle. But in science-speak, it's a compound with a specific chemical formula: NaHCO₃. See? Not so scary! It’s just a shorthand way of saying it’s made of sodium (Na), hydrogen (H), carbon (C), and three oxygen atoms (O₃).
Now, why would we want to know its molar mass? Think of it like this: if you were going to bake a cake, you wouldn't just toss in random amounts of flour and sugar, would you? You need to know how much of each ingredient to use to get that perfect texture and taste. Molar mass is just the science version of that – it tells us the "weight" of one mole of a substance. A mole, by the way, is just a really big number of particles, like a dozen eggs, but on a cosmic scale!
Your Mission, Should You Choose to Accept It: The Periodic Table!
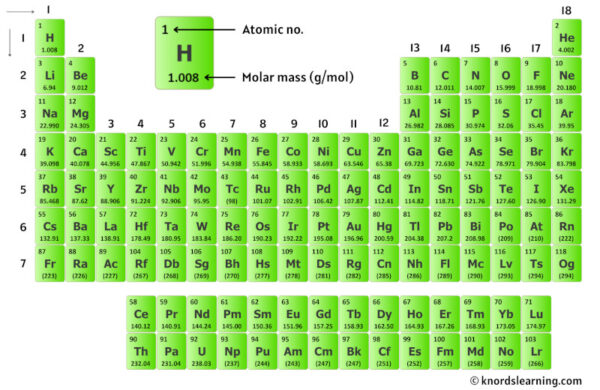
To calculate this "weight," we need a secret weapon: the Periodic Table. This isn't just a wall decoration in your old chemistry classroom; it's a treasure map to the atomic weights of all the elements. Each element on the table has a number associated with it – that’s its atomic mass, which is essentially the average weight of its atoms.

Let's break down NaHCO₃:
- Sodium (Na): We find Sodium on the Periodic Table. Its atomic mass is approximately 22.99.
- Hydrogen (H): Hydrogen is usually the first element you see. Its atomic mass is about 1.01.
- Carbon (C): Ah, Carbon, the backbone of so much life! Its atomic mass is roughly 12.01.
- Oxygen (O): And finally, Oxygen. We have three oxygen atoms in our formula (that little '3' after the O tells us this). Each oxygen atom has an atomic mass of about 16.00.
See? We’re just picking out numbers! It’s like gathering ingredients for our recipe.
Putting the Pieces Together
Now for the fun part! We’re going to add up the atomic masses of each element, making sure to account for how many of each atom we have in our formula. It’s a little bit of simple addition, and before you know it, you’ll have the molar mass!

So, let’s do the math:
- Sodium (Na): 1 atom * 22.99 g/mol = 22.99 g/mol
- Hydrogen (H): 1 atom * 1.01 g/mol = 1.01 g/mol
- Carbon (C): 1 atom * 12.01 g/mol = 12.01 g/mol
- Oxygen (O): 3 atoms * 16.00 g/mol = 48.00 g/mol
Now, we just sum these up. Ready for the grand total? 22.99 + 1.01 + 12.01 + 48.00 = 84.01 g/mol.
Ta-da! You’ve just calculated the molar mass of sodium bicarbonate! 84.01 grams per mole. Isn't that something? You’ve essentially figured out the "standard weight" for a massive collection of baking soda molecules. It’s like knowing exactly how many ounces of flour you need for a dozen cookies, but for the microscopic world!

Why This is Cooler Than You Think
Okay, okay, I hear you. "But how does knowing this make my life more fun?" Well, let me tell you! This little calculation is the gateway to understanding so many things.
Did you know that understanding molar mass helps scientists figure out how much of one ingredient they need to react with another to create new substances? This is how new medicines are developed, how better fuels are engineered, and even how those amazing sparkly glitter glues are made! It’s the foundation of pretty much all chemistry.
Think about baking itself. The recipe calls for a specific amount of baking soda because its molar mass dictates how it interacts with other ingredients to create that perfect, fluffy texture. If you mess with the amounts, you mess with the chemistry, and suddenly your cake is flat as a pancake. Molar mass is the silent hero of your kitchen!
And it's not just about cooking. Understanding these fundamental concepts allows you to appreciate the world around you on a deeper level. When you see a chemical reaction happening, whether it's a volcano science experiment or the fizzing of an antacid tablet, you’ll have a little peek behind the curtain, knowing the numbers that are making it all happen.
The Joy of Discovery Awaits
Calculating the molar mass of sodium bicarbonate might seem like a small step, but it’s a huge leap in your journey of curiosity. It’s a reminder that even the most complex scientific ideas are built from simple, understandable parts. And once you start understanding those parts, the whole universe starts to make a little more sense, and a whole lot more fun!
So, next time you reach for that box of baking soda, give a little nod to its molar mass. You've unlocked a tiny piece of scientific magic! And the best part? This is just the beginning. The Periodic Table is brimming with more elements, more compounds, and more fascinating calculations waiting for your brilliant mind to discover. Go forth, my friends, and keep exploring. The world of science is calling, and it’s ready to wow you!
