Calculate The Molar Mass Of Ammonium Carbonate
Ever wondered about the building blocks of the world around you? It might sound a bit like a science lab, but understanding things like the molar mass of ammonium carbonate can actually be surprisingly fun and useful! Think of it as a little puzzle that helps you figure out how much "stuff" is packed into a certain amount of a substance. It’s a popular concept because it bridges the gap between the tiny, invisible atoms and the everyday amounts we can see and measure.
So, why should you care about calculating the molar mass of ammonium carbonate? For beginners dipping their toes into chemistry, it’s a fantastic starting point. It’s one of those foundational calculations that, once you get the hang of it, opens the door to understanding many other chemical concepts. For families looking for engaging ways to learn together, it can be a fun kitchen-science experiment. Imagine using it to figure out how much baking soda (which contains sodium bicarbonate, a related compound) you're adding to your cookies for the perfect rise! And for hobbyists, whether you're into gardening, brewing, or even making your own bath bombs, understanding molar mass can help you create more consistent and predictable results. For instance, if you’re trying to create a specific soil amendment, knowing the molar mass helps you add the right proportions of ingredients.
Let's talk about ammonium carbonate itself. It's a compound with the chemical formula (NH₄)₂CO₃. This little formula tells us it's made up of ammonium ions (NH₄) and carbonate ions (CO₃). The "2" next to the ammonium tells us there are two of them for every carbonate ion. So, when we calculate its molar mass, we're essentially adding up the masses of all these atoms. You can also find variations of this calculation for other common compounds. For example, the molar mass of water (H₂O) is a classic example, and understanding how to calculate it is key to many other chemical reactions.
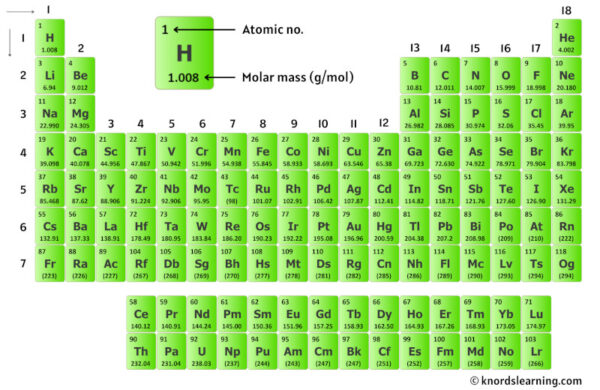
Getting started is easier than you think! All you need is a periodic table. This is your treasure map of elements, and each element has a number associated with it – that's its atomic mass. To calculate the molar mass of ammonium carbonate, you'll need the atomic masses of Nitrogen (N), Hydrogen (H), and Carbon (C), and Oxygen (O). Once you have those numbers, it's a simple process of multiplication and addition. For ammonium carbonate (NH₄)₂CO₃:
![Ammonium carbonate [(NH4)2CO3] Molar Mass (With Calculations)](https://knordslearning.com/wp-content/uploads/2023/10/Featured-on.webp)
- Find the atomic mass of Nitrogen (N).
- Multiply it by 2 (because there are two N atoms in NH₄).
- Find the atomic mass of Hydrogen (H).
- Multiply it by 4 (because there are four H atoms in NH₄).
- Add the results from the Nitrogen and Hydrogen calculations together to get the mass of one ammonium ion (NH₄).
- Multiply that sum by 2 (because there are two NH₄ ions).
- Find the atomic mass of Carbon (C) and add it to your running total.
- Find the atomic mass of Oxygen (O) and multiply it by 3 (because there are three O atoms in CO₃).
- Add the mass of the carbonate part to your total.
The final number, usually expressed in grams per mole (g/mol), is your molar mass! It might seem like a lot of steps, but with a periodic table and a little practice, it becomes second nature. You can even try it with table salt (NaCl) or sugar (C₁₂H₂₂O₁₁)!
So, don't shy away from a little bit of chemistry. Calculating the molar mass of ammonium carbonate, or any compound, is a rewarding skill that can add a new layer of understanding and enjoyment to your everyday life and your hobbies. It’s a small step that leads to a bigger appreciation for the material world around us.
