Calculate The Mass Percent Of Sodium Bicarbonate In The Solution

Hey there, fellow kitchen adventurers and accidental scientists! Ever found yourself staring at a fizzy mess, a baking project gone slightly sideways, or maybe just pondering the magical properties of that white powder lurking in your pantry? Yep, I’m talking about sodium bicarbonate – or as we affectionately call it, baking soda. It’s the unsung hero of so many of our domestic escapades, from de-gunking the oven to soothing a tummy ache that feels like a whole family of angry badgers has moved in.
But have you ever wondered, amidst the whisking and the measuring, just how much of this wonder-stuff is actually in that solution you just whipped up? You know, the one for that extra fluffy pancake recipe that’s supposed to make you a breakfast legend? Or perhaps you’re trying to figure out if that little bit of baking soda you dropped into your murky aquarium water is really doing anything, or if you’re just conducting an expensive science experiment with your goldfish as the unwilling test subjects.
Today, we’re diving headfirst, no goggles required, into the surprisingly simple world of calculating the mass percent of sodium bicarbonate in a solution. Don’t let the fancy name scare you. It’s less about arcane laboratory rituals and more about understanding the oomph, the sheer oomph, that baking soda brings to your mix. Think of it like this: you’re trying to figure out how much of the party is actually the pizza (that’s our baking soda!) and how much is just, well, the table and the slightly-too-loud music (that’s our water, or whatever else you’ve dissolved it in).
We’ve all been there, right? Staring at a recipe that calls for “a pinch of baking soda.” A pinch! What does that even mean? Is it a dainty little pinch like you’re sprinkling fairy dust on a unicorn’s birthday cake? Or a “whoops, I accidentally dropped half the box” kind of pinch? It’s a culinary enigma wrapped in a mystery, seasoned with a dash of uncertainty. And that’s where our little bit of science comes in. It’s like getting the cheat codes for your kitchen experiments.
So, grab your imaginary lab coat (or just your favorite apron, which is way more practical) and let’s get down to it. We’re going to break down how to figure out the mass percent of sodium bicarbonate. It’s not rocket science, folks. It’s more like… well, it’s baking science. And who doesn’t love baking? Unless you’re like my Uncle Barry, who once tried to bake a cake using salt instead of sugar. Let’s just say the birds enjoyed his garden more than his family did that year.
First off, what is this mysterious “mass percent”? Imagine you’ve got a big bowl of fruit salad. The mass percent of strawberries in that salad is simply the weight of all the strawberries divided by the total weight of the entire salad, then multiplied by 100. Easy peasy, right? You’re basically asking, “How much of this deliciousness is just the strawberries?”
In our case, the “deliciousness” is our solution, and the star of the show, the ruby red strawberry of our solution, is our beloved sodium bicarbonate. The other stuff, the juicy melon and the wobbly grapes, is, you guessed it, whatever else is in there – usually water. But it could be anything, really. Maybe you’re making some experimental bath bombs with essential oils, or you’re trying to neutralize a particularly stubborn acidic spill in your garage that smells suspiciously like forgotten gym socks.
The Grand Equation: Unpacking the Mystery
So, how do we actually do this calculation? It’s not like we have a tiny scale inside our solution, diligently weighing the baking soda and the water separately. But we have ways, oh yes, we have ways. The formula itself is actually quite friendly. It looks like this:
Mass Percent of Solute = (Mass of Solute / Mass of Solution) x 100%
Now, let’s translate this into plain English, the kind that doesn’t require a secret handshake with a chemistry professor.

The “Solute” is the thing that gets dissolved. In our baking soda adventure, the solute is, you guessed it, sodium bicarbonate. It’s the ingredient that’s going on a little vacation in the liquid.
The “Solution” is the final concoction – the solute all happily mixed and mingled with the other stuff. It’s the complete fruit salad, not just the strawberries. So, it’s our sodium bicarbonate plus whatever it’s dissolved in.
And “Mass” is just a fancy word for how much “stuff” there is. We usually measure this in grams (g) or kilograms (kg). Think of it as how heavy something is. If you’ve ever tried to lift a toddler who’s had too much sugar, you know exactly what mass is.
So, putting it all together, we need two key pieces of information:
- The weight of the sodium bicarbonate you used.
- The total weight of your final solution.
Once you have those two numbers, you’re basically golden. It’s like having the ingredients list and the nutritional information all rolled into one handy-dandy calculation.
Scenario 1: The "I Just Whipped This Up" Approach
Let’s say you’re making some homemade antacids because you’ve just inhaled an entire pizza and a family-sized bag of chips in one sitting. You know the drill: baking soda and water. You carefully measured out 5 grams of sodium bicarbonate. You then added 50 grams of water to make your solution. Easy, right?
First, what’s our solute? Sodium bicarbonate! How much? 5 grams.

Next, what’s our solution? It’s the sodium bicarbonate plus the water. So, the total mass of the solution is 5 grams (baking soda) + 50 grams (water) = 55 grams.
Now, we plug these numbers into our friendly formula:
Mass Percent = (5 grams / 55 grams) x 100%
Do the math (or whip out your phone calculator, no judgment here – we’re all about efficiency!), and you get approximately 9.09%.
So, in your homemade antacid, about 9.09% of the mixture is the mighty baking soda. That’s like saying that for every 100 grams of your antacid solution, just over 9 grams are the pure, unadulterated baking soda, ready to tackle that heartburn. Not bad for a couple of kitchen staples!
This is super useful if you’re experimenting with cleaning solutions too. Maybe you’re trying to create the ultimate all-purpose cleaner that can tackle grease, grime, and those mysterious sticky spots that seem to breed in the dark corners of your kitchen. Knowing the concentration of baking soda helps you understand its cleaning power. Too little, and it’s just sad water. Too much, and you might end up with a powdery residue that looks like a miniature snowstorm has hit your countertops.
Scenario 2: The "What Did I Actually Put In Here?" Dilemma
Okay, sometimes life isn’t that neat and tidy. You’re making that legendary pancake batter, and you’ve already added eggs, flour, milk, and then you remember the baking soda. You scoop some in, you whisk, you pour. Now, you want to know the mass percent, but you’re not entirely sure how much you actually added. This is where things can get a little… shall we say, creative.
Here’s where a bit of detective work comes in. If you have the initial weight of the baking soda container and the final weight after you’ve scooped, you can subtract to find out how much you used. Or, if you’re feeling particularly ambitious, you can carefully scoop out a portion of your finished solution, evaporate the water (gently! no explosions!), and weigh the remaining baking soda. It’s a bit like excavating an archaeological site, but with more flour dust.
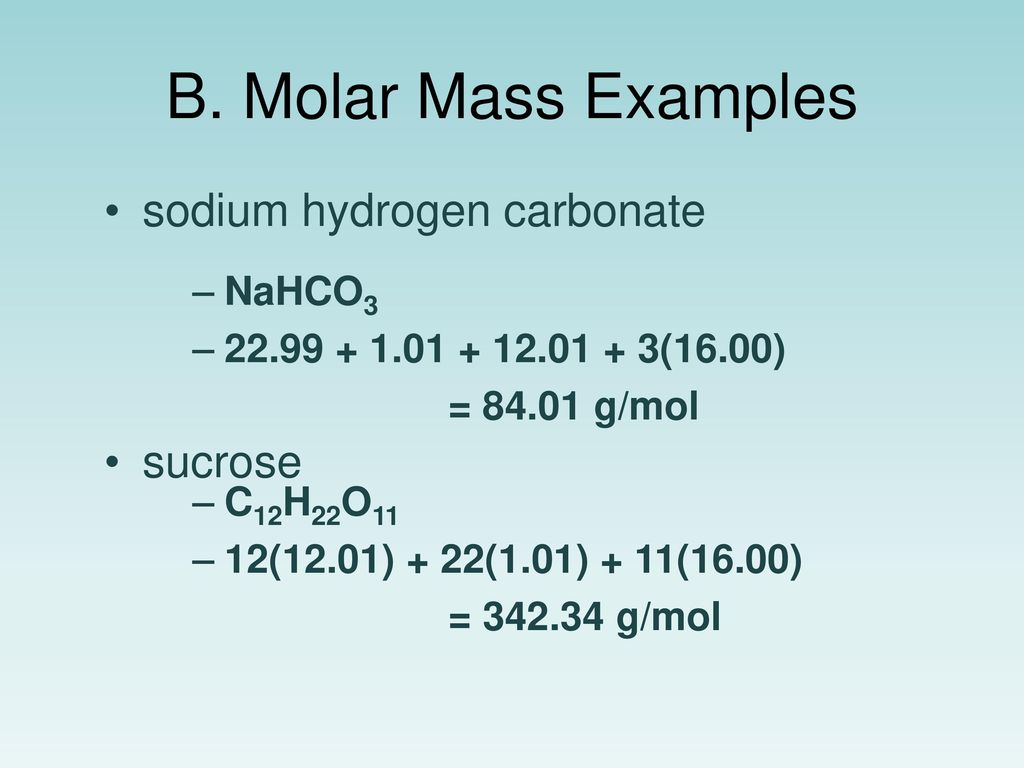
Let’s imagine a slightly different scenario. You’ve bought a pre-made baking soda solution for some reason (maybe you’re feeling fancy, or you’ve lost all your spoons). The label says it contains 100 grams of sodium bicarbonate dissolved in 400 grams of water.
Our solute is again, sodium bicarbonate, weighing in at 100 grams.
Our solution is the sodium bicarbonate plus the water: 100 grams + 400 grams = 500 grams.
Plugging into our trusty formula:
Mass Percent = (100 grams / 500 grams) x 100%
Which gives us a nice, round 20%.
So, this particular solution is 20% baking soda. That’s a pretty concentrated mix! This might be something you’d use for a more industrial-strength cleaning task, like tackling that mysterious, hard-to-remove grime on the grout lines that’s been haunting your bathroom for weeks. Or perhaps for a science fair project involving volcanic eruptions that could rival Mount Vesuvius.

The Importance of Accurate Measurement (Even When You're Not Trying to Blow Anything Up)
The key takeaway here, my friends, is that accuracy matters. Even in casual kitchen experiments, a little bit of precision can go a long way. If you’re aiming for that perfect, fluffy cake, you don’t want to accidentally add too much baking soda and end up with a bitter, soapy taste. Trust me, I’ve been there. My “chocolate lava cake” once tasted suspiciously like a cleaning product, and the only thing that melted was my pride.
Measuring the mass of your ingredients is generally more reliable than measuring by volume (like using cups or spoons). Why? Because different substances pack differently. A cup of flour can vary in weight depending on how loosely or tightly you pack it. But a gram is a gram, no matter what. It’s like comparing a loosely packed suitcase full of socks to a neatly folded pile of shirts – the number of socks might be the same as the number of shirts, but the actual weight can be wildly different.
So, for those times when you’re serious about your results, a kitchen scale is your best friend. It’s like having a tiny, very honest accountant for your food. It won’t judge your questionable late-night snack choices, but it will tell you exactly how much that bag of chips weighs.
Beyond the Kitchen: Where Else Does This Pop Up?
It’s not just about making your home sparkle or your baked goods rise. Understanding mass percent is a fundamental concept in chemistry and is used in countless everyday applications. Think about:
- Medicines: When you take a pain reliever, the label tells you the percentage of the active ingredient. That’s a mass percent calculation!
- Fertilizers: Farmers need to know the exact percentage of nutrients in their fertilizers to ensure healthy plant growth.
- Food Science: Ever wondered about the sugar content of your favorite drink? Mass percent plays a role in that too.
- Environmental Science: Measuring pollutants in water or air often involves calculating their mass percentages.
So, while you might be using baking soda to scrub your sink today, the principles you’re applying are the same ones used in laboratories and industries around the globe. You’re basically a citizen scientist, armed with a whisk and a calculator!
The Final Fizz: Bringing it All Together
Calculating the mass percent of sodium bicarbonate in a solution is a straightforward process once you understand the components. You need the weight of the baking soda (your solute) and the total weight of the mixture (your solution). Plug those numbers into the formula: (Mass of Solute / Mass of Solution) x 100%.
It’s a way to quantify the power and presence of that humble white powder in your life. Whether you’re aiming for perfectly risen cookies, a sparkling clean bathroom, or just trying to understand what’s going on in that fizzy drink you’re making, this calculation gives you the inside scoop.
So next time you reach for that box of baking soda, remember that you’re not just adding an ingredient; you’re adding potential. And with a little bit of simple math, you can measure that potential. It’s a small skill that can lead to bigger, tastier, and cleaner results. Happy calculating, and may your solutions always be perfectly balanced (and never taste like cleaning products)! Now, if you’ll excuse me, I think I hear my oven calling my name. It’s time to put this knowledge to the test, with a little less Uncle Barry-level experimentation this time.
