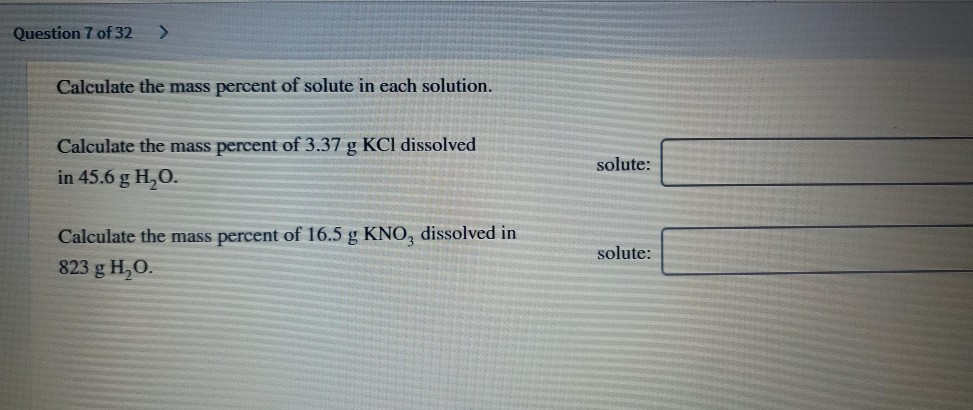
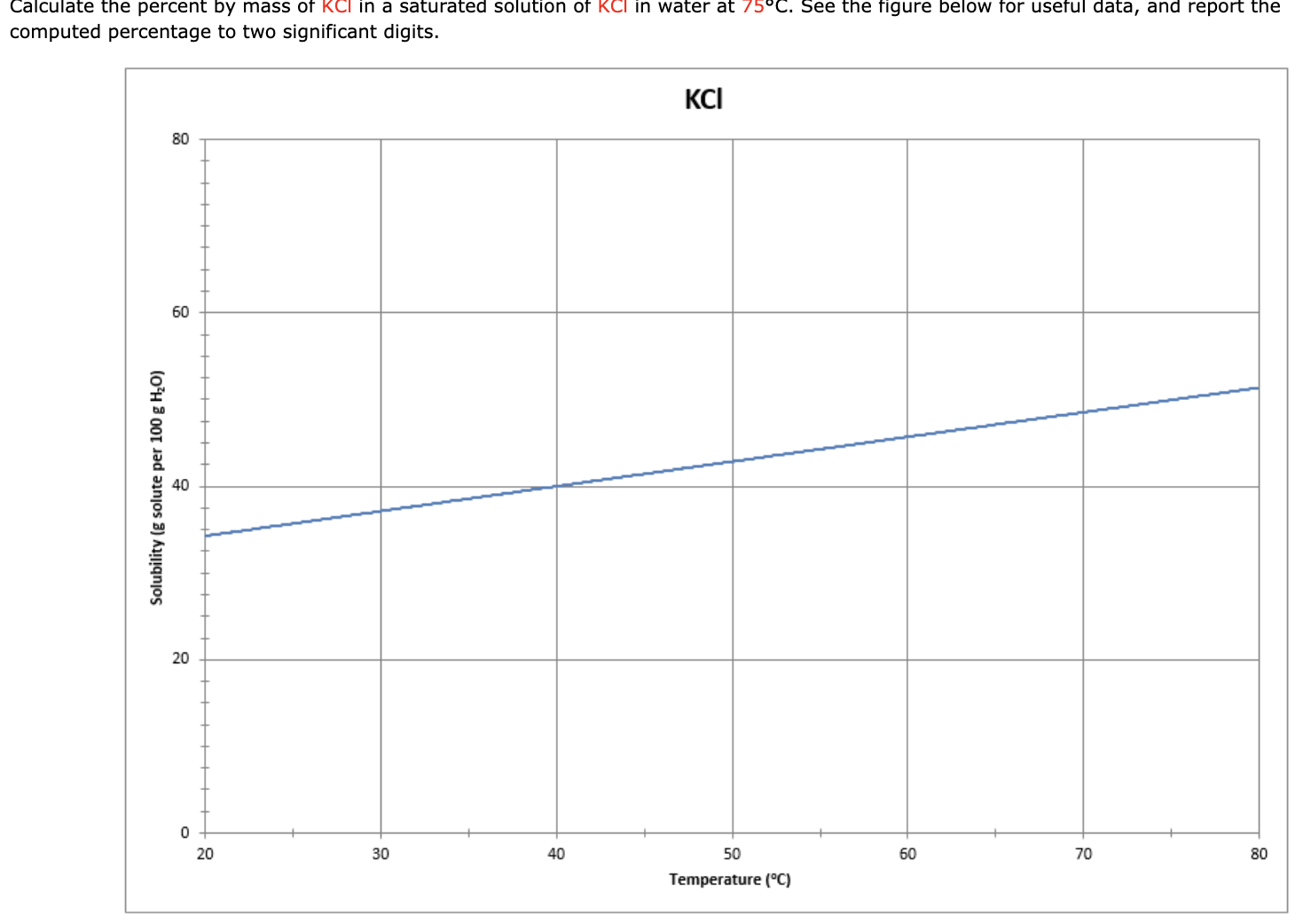
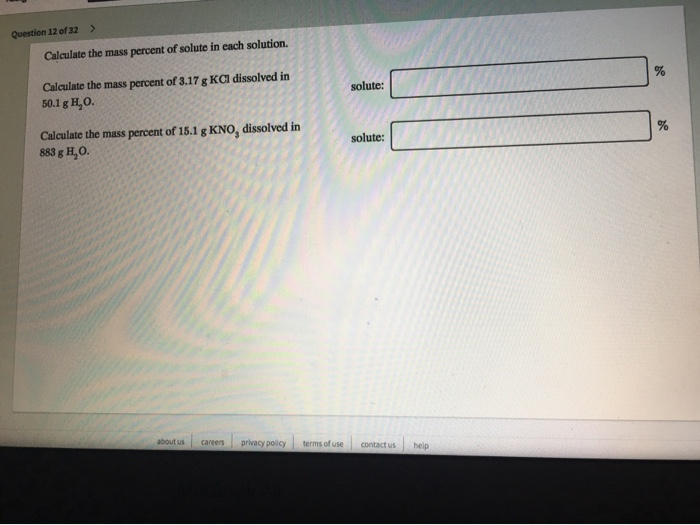
Calculate The Mass Percent Of Kcl In The Solution
Hey there, science adventurer! Ever found yourself staring at a label, maybe on a bottle of something fizzy or even a fancy skincare product, and wondered, "What's really in this stuff?" Well, today we're diving into a little bit of kitchen chemistry that's surprisingly fun and, dare I say, empowering. We're going to unlock the secret to calculating the mass percent of KCl in a solution. Sounds a bit technical, right? But stick with me, because this isn't about memorizing complicated formulas (though we'll touch on one, don't worry!). It's about understanding how we quantify what's dissolved in our everyday liquids, and trust me, that's pretty cool!
So, what exactly is mass percent? Think of it like this: if you bake a cake, and you want to know how much of your delicious chocolate chips are in the whole cake, you'd figure out the weight of the chips and divide it by the total weight of the cake, then multiply by 100. Easy peasy, right? That's essentially what mass percent is all about. It tells you, out of the total weight of your solution, how much of a specific ingredient (in our case, potassium chloride, or KCl) is hanging out there. It's like knowing the exact concentration of sunshine in your lemonade!
Why KCl, you ask? Well, KCl, or potassium chloride, is a pretty common compound. You might find it in some table salt substitutes, fertilizers, or even in medical solutions. Understanding its concentration in a liquid can be super important in all sorts of fields, from agriculture to healthcare. And honestly, just knowing you can figure this out? That's a little superpower you get to add to your belt!
Let's imagine you're making a simple brine. Maybe for pickling some cucumbers, or perhaps you're a budding scientist experimenting with different salt concentrations. You've got your water, and you've dissolved some KCl in it. Now, how do you know how much KCl is actually in there, percentage-wise? This is where our little calculation comes in!
The magic formula, which is honestly less magic and more just common sense, is this:
Mass Percent of KCl = (Mass of KCl / Mass of Solution) * 100%
See? It's just like our cake example! You need two key pieces of information: the mass of the KCl you added and the total mass of the solution. The "solution" is everything combined – the KCl and the solvent, which in this case is usually water.
Let's break down how you'd get those numbers. First, the mass of KCl. This is the easy part if you're doing it yourself. You'd grab a trusty kitchen scale, weigh out your potassium chloride, and voilà! You have your first number. Let's say you added 10 grams of KCl. Easy, right?
Now, the mass of the solution. This is where a tiny bit of thought comes in. If you simply weighed the water before you added the KCl, that's not the mass of the solution. You need the combined weight. So, the best way is to weigh your container (say, a beaker or a jar), then weigh it again with the water in it to get the mass of the water. Then, add your KCl, and weigh it one last time to get the total mass of the solution. Or, a bit of a shortcut: weigh your container, add your water, weigh again to get water's mass. Then, add your KCl, stir it all up, and weigh the whole shebang! That final weight is your mass of the solution.

Let's say you added 90 grams of water to your 10 grams of KCl. What's the mass of the solution? Drumroll, please... it's 100 grams! (10g KCl + 90g water = 100g solution). So, using our handy formula:
Mass Percent of KCl = (10 grams / 100 grams) * 100% = 10%
Ta-da! You've just calculated that your solution is 10% KCl by mass. How cool is that? You took raw ingredients and, with a little bit of weighing and a simple calculation, you’ve quantified its composition. It’s like being a culinary alchemist, but with actual, useful science!
What if your numbers are a bit different? Let's say you added 25 grams of KCl to 150 grams of water. The mass of the solution would be 175 grams (25g + 150g). So, your mass percent would be:
Mass Percent of KCl = (25 grams / 175 grams) * 100% ≈ 14.3%
See? The math is still straightforward, even with different quantities. It's all about that ratio. This skill isn't just for textbooks, either. Imagine you're trying to make a perfect bath soak with Epsom salts (which is magnesium sulfate, but you get the idea!). Knowing the concentration can help you achieve the exact effect you're going for. Or maybe you’re a gardener and need to mix a fertilizer solution – getting the concentration right is key for happy plants!
It’s also a fantastic way to make sense of recipes or instructions that might seem a bit vague. When you can translate “dissolve some salt in water” into a precise percentage, you gain a whole new level of control and understanding. It’s about moving from passively following instructions to actively understanding the why behind them.
Think about the satisfaction of knowing you can accurately measure and understand the components of the liquids around you. It's a small step, but it opens doors to understanding bigger scientific concepts. It’s the kind of knowledge that builds confidence and makes you feel a little more connected to the world of science. It’s not just about numbers; it’s about the power of observation and measurement. It’s about seeing the world with a little more clarity and a lot more curiosity.
So, next time you're mixing something up, whether it's a science experiment for fun or just a home remedy, remember this little trick. Give it a try! Grab some KCl (or even just regular table salt, NaCl, and calculate its mass percent!), some water, and your scale. Play around with it. See what different concentrations you can create. You might be surprised at how much fun you have, and how much smarter you feel!
And remember, this is just the tip of the iceberg! The world of chemistry is vast and full of fascinating discoveries. By understanding simple concepts like mass percent, you’re building a foundation for exploring even more complex and exciting ideas. So, keep that curiosity buzzing, keep experimenting, and never stop learning. You’ve got this!
