Calculate The Kc Value For The A -protein Binding Reaction.

Hey there, bio-nerd extraordinaire, or maybe just someone who’s stumbled upon this page wondering what all the fuss is about! Ever found yourself staring at a lab report, a scientific journal article, or even a particularly insightful documentary about, say, how your body’s amazing protein machinery works, and hit a wall with a term like “Kc value”? Don’t worry, you’re not alone. It sounds super intimidating, like something only a Nobel laureate would whisper about over a single malt. But guess what? It’s actually a pretty cool concept, and understanding it, even at a casual level, can give you a little peek behind the curtain of the microscopic world that keeps you ticking. Think of it as the secret handshake of molecular interactions. Today, we’re going to demystify the Kc value for an A-protein binding reaction, and I promise, it’ll be less like a pop quiz and more like a chill chat over coffee.
So, what exactly is this “Kc value” we’re talking about? In the grand, bustling metropolis of your cells, proteins are the ultimate multitaskers. They’re the builders, the transporters, the messengers, and even the bouncers at the cellular club. And they rarely work alone. They often need to bind to other molecules, like other proteins, DNA, RNA, or even small drug molecules, to get their jobs done. Think of it like a celebrity needing their entourage, or a superhero needing their trusty sidekick. This binding is crucial for everything from digesting your last meal to remembering that hilarious meme you saw yesterday.
The “A-protein binding reaction” is just a fancy way of saying “protein A is grabbing onto molecule B.” It’s a dance, a connection, a partnership. And the Kc value? Well, it’s basically the measure of how eager or strong that partnership is. In scientific lingo, it’s the equilibrium constant for the association reaction. Don’t let the word “equilibrium” scare you; it just means that at some point, the rate at which A and B are joining up is the same as the rate at which they’re breaking apart. It’s a dynamic balance, like a perfectly orchestrated ballet where dancers are constantly moving, but the overall pattern remains.
Unpacking the Kc: The Affinity Game
Imagine you’re at a party, and you’re trying to strike up a conversation. Some people are super approachable, practically jumping at the chance to chat (high affinity). Others are a bit more reserved, and you might need to work a little harder to get them to engage (lower affinity). The Kc value is kind of like the party host’s report card on how popular certain guests are. A high Kc value means protein A really likes binding to molecule B. They’re like two peas in a pod, inseparable best friends. A low Kc value, on the other hand, suggests that protein A isn’t that thrilled about molecule B. They might interact briefly, but they’re not exactly soulmates.
This concept is super important in biology because it dictates so many things. For example, if a drug needs to bind to a specific protein to have an effect, scientists want that drug to have a high affinity – meaning it binds strongly and reliably. Think of a lock and key. A well-fitting key (high affinity) will easily open the lock, while a poorly fitting one (low affinity) might not work at all. It's also why understanding these binding strengths is crucial for designing new medicines or understanding diseases where these interactions go haywire.
Fun fact: The study of molecular interactions and binding affinities has roots stretching back to the early days of chemistry and pharmacology. Scientists like Wilhelm Ostwald in the late 19th century were already laying the groundwork for understanding chemical equilibria, which are the bedrock of these Kc values. So, while it sounds modern, it’s built on a pretty solid historical foundation!
So, How Do We Actually Calculate This Thing?
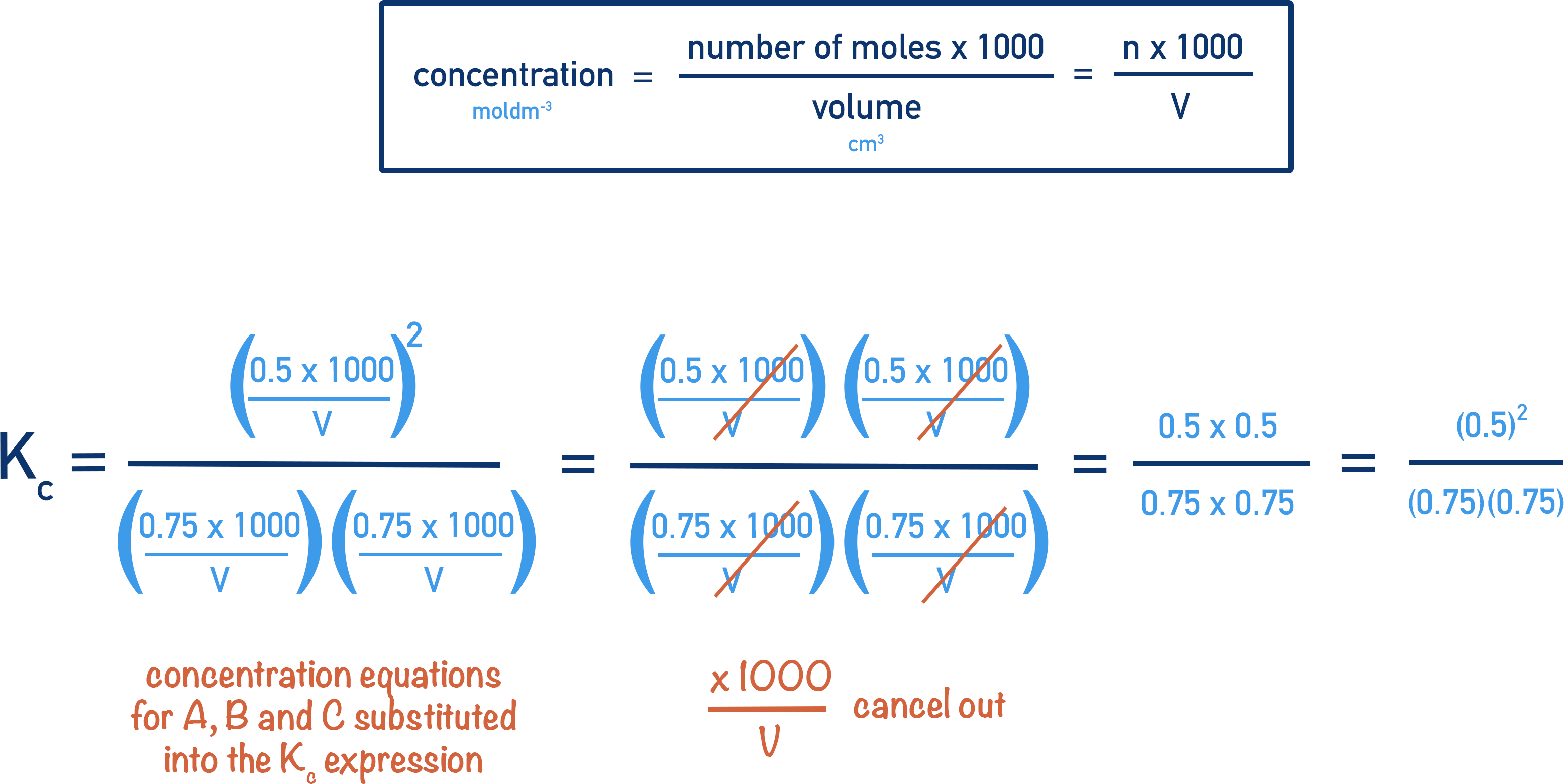
Alright, now for the slightly more hands-on part, without actually needing a lab coat unless you want to rock the look! To calculate the Kc value for your A-protein binding reaction, you’re essentially looking at the concentrations of your molecules at equilibrium. The general formula, which looks a bit like a recipe, is:
Kc = [Products] / [Reactants]
In our specific A-protein binding scenario, let’s say protein A binds to molecule B to form a complex, A-B. The reaction would look something like this:
A + B <=> A-B
(The double arrow, <=> , signifies that the reaction can go both forward – A and B forming A-B – and backward – A-B breaking apart into A and B.)
So, the Kc expression for this would be:
Kc = [A-B] / ([A] * [B])
Where:
- [A-B] is the concentration of the A-B complex at equilibrium.
- [A] is the concentration of free protein A at equilibrium.
- [B] is the concentration of free molecule B at equilibrium.
The units of Kc can vary depending on the reaction order, but for this simple binding, it often ends up being something like “per molar” or “M-1”, indicating how strongly the molecules associate. A larger number means more complex is formed relative to the free molecules.

Practical Tip: To actually get these numbers, scientists use a variety of experimental techniques. One common method is spectroscopy, where you can measure how much light a molecule absorbs or emits. If protein A or molecule B, or the complex A-B, has a unique spectral signature, you can track their concentrations over time as they bind and reach equilibrium. Other methods include techniques like surface plasmon resonance (SPR), isothermal titration calorimetry (ITC), or fluorescence anisotropy, all of which are fancy ways of measuring binding events and their strengths.
Think of it like trying to figure out how many people are sitting at a table versus how many are standing around it at a party. You can count them! Here, we’re counting molecules. It’s a bit more precise, of course.
Why Does This Matter in the Real World?
You might be asking, “Okay, so protein A likes molecule B a lot. So what?” Well, this seemingly simple concept has ripple effects throughout our lives.
Drug Discovery: This is a big one. When pharmaceutical companies develop new drugs, they’re essentially trying to design molecules that will bind with high affinity to specific protein targets in our bodies. Whether it’s an antiviral medication that needs to latch onto a viral protein, or a cancer drug that targets a faulty protein in a cell, understanding the Kc value of these interactions is paramount. A drug with a high Kc value for its target means it will be effective at lower concentrations and potentially have fewer off-target side effects. It’s about precision targeting, like a laser beam versus a floodlight.
Understanding Diseases: Many diseases are caused by proteins not behaving as they should – either they don’t bind to their partners when they should, or they bind too much. For instance, Alzheimer’s disease involves the misfolding and aggregation of certain proteins. Understanding the binding affinities of these proteins can provide clues about how the disease progresses and how it might be treated. It’s like diagnosing a faulty connection in a complex electrical circuit.
Biotechnology and Diagnostics: Think about those rapid COVID-19 tests. They rely on antibodies (a type of protein) binding very specifically to viral proteins. The sensitivity and accuracy of these tests are directly related to the binding affinity, which can be indirectly related to the Kc value of the antibody-antigen interaction. The better the protein partners stick together, the more reliable the test.
Food Science: Even in your kitchen, protein binding plays a role! The way eggs bind together when you cook them, or how proteins in cheese create its texture, involves intricate molecular interactions. While we don't usually calculate Kc values for cooking, the underlying principles of how molecules attract and bind are the same.
Cultural reference: Remember those iconic scenes in sci-fi movies where characters have to precisely dock a spaceship? It’s a bit like that, but on a molecular level. The docking mechanism (binding) needs to be just right. A good dock (high Kc) ensures a stable connection.
Keeping It Simple: An Analogy You Can Grasp
Let’s try another analogy. Imagine you’re at a concert. The stage is where the reaction happens, and the performers are your proteins and molecules. The Kc value is like a measure of how popular the band is. A super famous band (high Kc) will have a massive, tightly packed crowd right up front, all eager to get as close as possible. A lesser-known band (low Kc) might have a more spread-out audience, with people wandering in and out of the prime viewing spots.
The concertgoers who are really close to the stage are like the bound A-B complex. The people who are still milling around the back or getting drinks are like the free A and B molecules. If the band is a huge hit (high Kc), there will be a lot more people packed near the stage (high [A-B]) and relatively fewer people hanging back (low [A] and [B]). If the band is just okay (low Kc), you’ll see more people not bothering to get close, and the ratio of people near the stage to those further away won’t be as dramatic.
So, when scientists measure the Kc value, they are essentially measuring the crowd density right in front of the stage relative to the total number of people at the concert. It tells them how much the audience wants to be up close and personal with the performers.
Navigating the Data: What a High or Low Kc Really Means
High Kc (e.g., 106 M-1 and above): This indicates a very strong binding. Protein A and molecule B are practically glued together. This is desirable for many therapeutic applications, like targeted therapies, or for highly specific diagnostic assays. Think of it as a molecular embrace that’s hard to break.

Medium Kc (e.g., 103 to 106 M-1): This is a moderate binding affinity. The protein and molecule interact readily but can also dissociate under certain conditions. Many natural biological processes rely on these dynamic, reversible interactions. It’s like a friendly handshake – firm but easily released.
Low Kc (e.g., below 103 M-1): This signifies weak binding. Protein A and molecule B have only a fleeting interaction. This might be sufficient for some biological functions or for molecules that need to be easily released, but it’s generally not what you’d aim for if you need a stable, long-lasting molecular partnership.
It's important to remember that Kc is just one piece of the puzzle. Other factors like the concentrations of A and B, the pH of the environment, temperature, and the presence of other molecules can all influence how much binding actually occurs. But the Kc value gives us that fundamental, intrinsic measure of how much two molecules like each other.
A Moment of Reflection
It’s quite remarkable, isn’t it? That on the microscopic level, within the very fabric of our existence, these intricate molecular partnerships are constantly forming and breaking. The Kc value, this seemingly abstract number, is a testament to the elegant efficiency of life. It tells a story of attraction, of compatibility, of purpose.
Thinking about Kc values for protein binding can actually be a gentle reminder for our own lives. How often do we seek connections, collaborations, and relationships? Just like proteins, we too have varying degrees of “affinity” for different people, ideas, and activities. Some connections are deep and enduring (high Kc), providing strength and support. Others are fleeting and superficial (low Kc), perhaps offering a momentary pleasant interaction.
In a world that often feels chaotic, understanding the importance of strong, meaningful “binding” – whether it’s in our personal relationships, our professional endeavors, or our commitment to our passions – can be incredibly empowering. It’s about seeking out those interactions that are mutually beneficial, that create stable and productive “complexes,” and that contribute to the overall health and well-being of our own personal biological systems, or, you know, our lives. So next time you hear about Kc values, don’t just think of it as science jargon; think of it as a microscopic peek into the fundamental principle of connection that resonates even in our everyday human experience.
