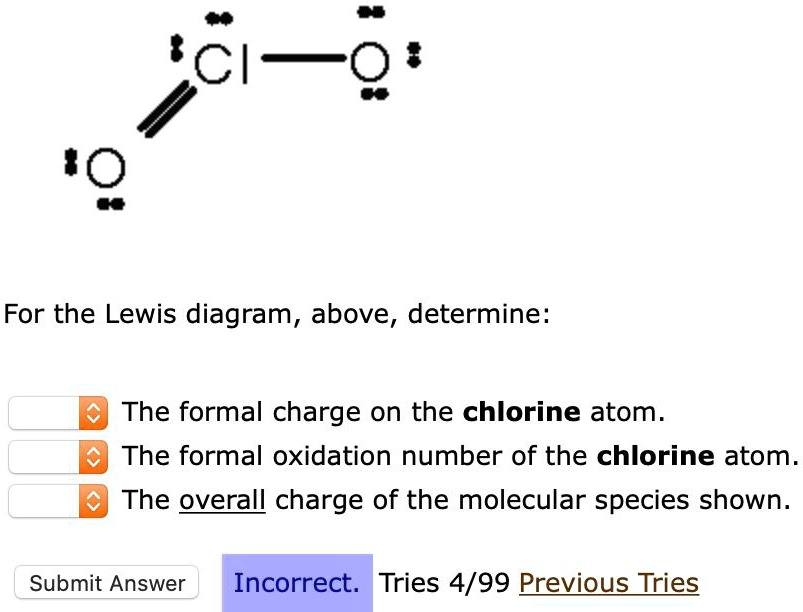
Calculate The Formal Charge On The Chlorine Cl Atom

Hey there, you! Grab your mug, settle in. We're about to dive into something that sounds a tiny bit scary, but I promise, it's more like figuring out who's hogging the remote. We're talking about formal charge, and specifically, how to calculate it on our pal, the chlorine atom. Yeah, chlorine! That stuff in your swimming pool, or maybe the one hanging out in a salt molecule. Super common, right?
So, what even is this "formal charge" thing? Think of it like this: in a molecule, atoms are all sharing electrons, like a bunch of roommates splitting bills. But sometimes, someone's paying a little more than their fair share, or maybe they're getting a sweet deal. Formal charge helps us figure out who's holding the financial reins, electron-wise, in that specific molecular setup. It's not the actual charge, mind you, which is a whole other ballgame involving electronegativity and stuff. Nope, this is more like a scorekeeping system. Fun!
Why do we even bother with this scorekeeping? Good question! It helps chemists predict how molecules will behave. Like, if an atom has a big negative formal charge, it's probably gonna be super attracted to something positive. Makes sense, right? And if it's got a big positive charge? Well, it's probably looking for some electron love. It's all about understanding those molecular vibes.
And when it comes to chlorine, oh boy, chlorine is a drama queen in the world of bonding. It's got a knack for being in all sorts of situations, from super simple to totally bonkers. So figuring out its formal charge in different scenarios is like unlocking its personality traits. We're going to be like molecular detectives, sniffing out the electron distribution.
Ready to get our hands dirty? Or, you know, conceptually dirty? We need a formula, obviously. Don't worry, it's not calculus. It's way more manageable, I swear. The magic formula for formal charge is:
Formal Charge = (Valence Electrons) - (Non-bonding Electrons) - (1/2 * Bonding Electrons)
Let's break that down, piece by piece, like dissecting a really interesting but slightly confusing sandwich.
First up, Valence Electrons. This is just the number of electrons an atom starts with on its outermost shell. For chlorine, it's pretty straightforward. You look at the periodic table, find chlorine (Cl), and bam! It's in Group 17, which means it's got 7 valence electrons. Easy peasy, lemon squeezy. That's our starting point, our baseline. Imagine it's the amount of money each roommate supposedly put into the shared rent pot.
Next, Non-bonding Electrons. These are the electrons that are just chilling on the atom, doing their own thing, not participating in any sharing. They're like the roommate who pays their share on time but never helps with the dishes. We often see these as lone pairs, those little pairs of dots you draw around an atom in a Lewis structure. We need to count all of them, individually.
And finally, Bonding Electrons. These are the ones being shared between atoms. In our Lewis structures, they're the lines connecting the atoms. Each line represents a pair of shared electrons, so two electrons. We count all the electrons involved in bonds. Think of these as the electrons actively used to hold the molecule together, the joint effort in keeping the lights on. And remember, we take half of these. Why half? Because those shared electrons are technically half the responsibility of each atom involved in the bond. It's like splitting the cost of that shared Netflix account. Fair is fair!
Okay, so we've got the formula. Now, let's apply it. The best way to understand this is with examples. Because, let's be honest, abstract formulas can feel like trying to read a foreign language with no translator. We need context! We need to see this chlorine atom in action.
Let's start with a super common one: the chloride ion, Cl⁻. This is what you find when salt (NaCl) dissolves in water. It's gained an extra electron, right? So, it's not a neutral atom anymore. When it's an ion, it usually has a complete outer shell, which is pretty stable for chlorine. Usually, in the chloride ion, the chlorine atom has 8 non-bonding electrons. It's basically packed them all in, no sharing needed anymore. Think of it as that roommate who decided to just buy their own place and be totally independent. So, how do we calculate the formal charge here?
Remember our formula: Formal Charge = (Valence Electrons) - (Non-bonding Electrons) - (1/2 * Bonding Electrons)
For Cl⁻:
- Valence Electrons: 7 (Chlorine always starts with 7)
- Non-bonding Electrons: 8 (In the Cl⁻ ion, it's got 4 lone pairs)
- Bonding Electrons: 0 (It's not bonded to anything else in this form)
Plugging it in: Formal Charge = 7 - 8 - (1/2 * 0)
Formal Charge = 7 - 8 - 0
Formal Charge = -1
See that? The formal charge on the chlorine atom in the chloride ion (Cl⁻) is -1. This makes perfect sense because, well, it is a chloride ion with a charge of -1! The formal charge calculation perfectly matches the actual charge in this simple case. It's like getting the same answer on a math problem multiple ways. Satisfying!
Now, let's move on to something a little more complex. How about chlorine in a covalent bond? Let's take hydrogen chloride, HCl. This is a polar molecule, meaning the electrons are a bit unevenly distributed, but let's stick to our formal charge game for now. In HCl, the chlorine atom forms a single bond with the hydrogen atom. It also usually has 3 lone pairs of electrons. Those lone pairs are the non-bonding electrons we need to count.
So, for the chlorine atom in HCl:
- Valence Electrons: 7 (Still our starting point)
- Non-bonding Electrons: 6 (Those 3 lone pairs, 2 electrons each)
- Bonding Electrons: 2 (The single bond between H and Cl has 2 shared electrons)
Let's crunch those numbers:
Formal Charge = 7 - 6 - (1/2 * 2)
Formal Charge = 7 - 6 - 1
Formal Charge = 0

Ta-da! In HCl, the formal charge on the chlorine atom is 0. This means that, in terms of electron distribution as calculated by this method, the chlorine atom is essentially neutral within the molecule. It's like the roommates are perfectly splitting all the bills, no one owes anyone anything. Pretty balanced!
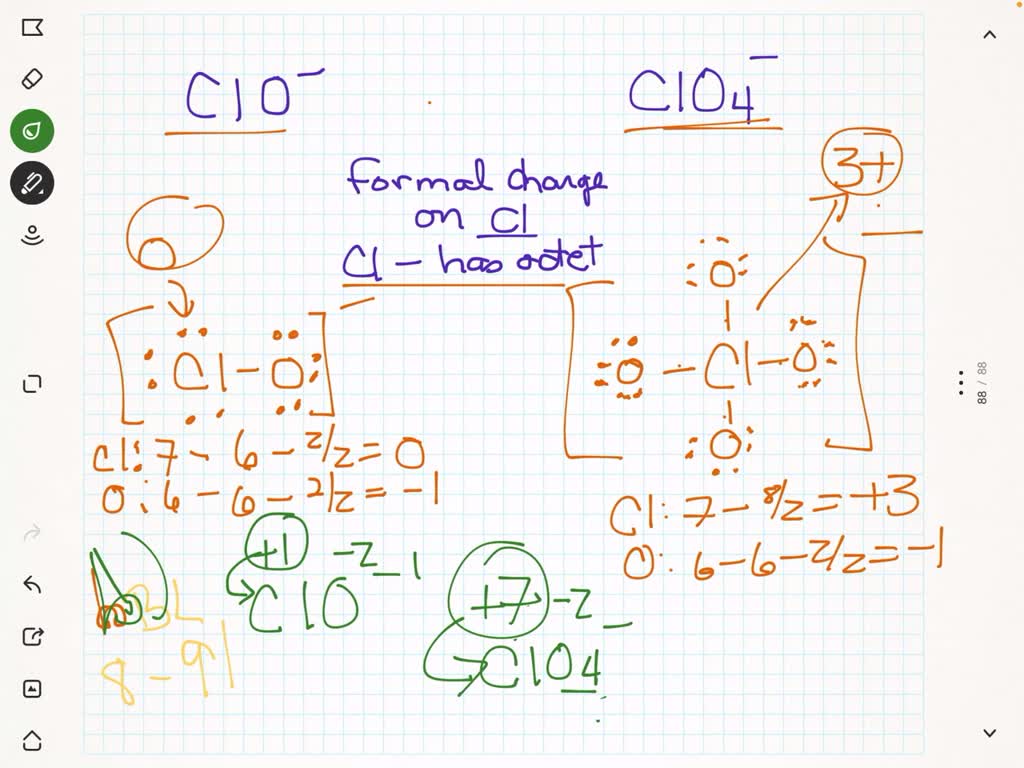
But wait, there's more! Chlorine can get really interesting. Let's think about something like perchlorate ion, ClO₄⁻. This is where things get a bit wild. In perchlorate, the chlorine atom is bonded to four oxygen atoms. And to make it all work out with the overall negative charge, the chlorine often forms double bonds with some of the oxygens. This is where formal charge becomes a superhero, helping us decide which Lewis structure is the best representation of reality.
There are actually a couple of valid Lewis structures for ClO₄⁻. One way to draw it gives the chlorine a formal charge. Let's look at a structure where chlorine is bonded to four oxygen atoms via single bonds, and the overall charge is -1. In this scenario, each oxygen atom would have lone pairs to complete its octet, and the chlorine would also have some lone pairs. This isn't the most stable structure, but let's calculate the formal charge on chlorine just to see.
In a structure with four single bonds to oxygen and a net charge of -1:
- Valence Electrons: 7
- Non-bonding Electrons: 4 (This arrangement might have 2 lone pairs on Cl)
- Bonding Electrons: 8 (Four single bonds, each with 2 electrons)
Formal Charge = 7 - 4 - (1/2 * 8)
Formal Charge = 7 - 4 - 4
Formal Charge = -1
Ooh, that's a negative formal charge on our chlorine! Now, let's look at a better Lewis structure for perchlorate, one that minimizes formal charges. In this more accepted structure, the chlorine atom forms one double bond with two of the oxygen atoms and single bonds with the other two oxygen atoms. This arrangement helps achieve octets and minimizes those pesky formal charges.
So, in the preferred Lewis structure for ClO₄⁻, the chlorine atom has:
- Valence Electrons: 7
- Non-bonding Electrons: 0 (In this structure, chlorine forms multiple bonds and uses all its valence electrons for bonding, no lone pairs left over!)
- Bonding Electrons: 14 (Two double bonds = 4 electrons each * 2 = 8 electrons. Two single bonds = 2 electrons each * 2 = 4 electrons. Total = 12 electrons. Wait, did I count right? Let's re-think. A double bond is 4 electrons, a single bond is 2. So, two double bonds is 8 electrons. Two single bonds is 4 electrons. That's 12 electrons. Hmm, let's pause and verify. Ah, the total number of electrons involved in bonding for chlorine here are from the bonds it forms. It has 4 bonds in total. If we consider each oxygen contributing electrons to these bonds, and chlorine sharing, the count for chlorine's contribution to bonding electrons is a bit trickier when it has double bonds. Let's go back to basics of what electrons are around the Cl atom in its bonds. It's involved in four regions of bonding. Okay, a simpler way: count the electrons in the bonds that involve chlorine. Two double bonds = 8 electrons. Two single bonds = 4 electrons. Total is 12 bonding electrons. Yes, that's right.)
Formal Charge = 7 - 0 - (1/2 * 12)
Formal Charge = 7 - 0 - 6
Formal Charge = +1
Whoa, now the chlorine has a +1 formal charge! And the oxygens that have double bonds? They'll have a formal charge of 0. The oxygens with single bonds will have a formal charge of -1, and those will also have lone pairs. When you add up all the formal charges on all the atoms in ClO₄⁻, you get -1, which is the actual charge of the ion. Phew!
So, why do we prefer the structure with the +1 formal charge on chlorine over the one with the -1 formal charge on chlorine? Because generally, chemists like to minimize the magnitude of formal charges. A +1 is smaller in magnitude than a -1. Also, electronegativity plays a role. Oxygen is more electronegative than chlorine, so it's okay for oxygen to carry a negative charge (or contribute to one) and for chlorine to carry a positive charge. It's all about stability, and minimizing formal charges often leads to a more stable arrangement. It's like finding the most cost-effective way to run a household.
Let's try another one. What about chlorine dioxide, ClO₂? This is a bit of a tricky one. The Lewis structure can be drawn in a few ways, and it's a radical species, meaning it has an unpaired electron. For a common representation, chlorine is bonded to two oxygen atoms. Let's assume one double bond and one single bond, with an unpaired electron on chlorine, and lone pairs on oxygens to complete their octets.
In such a structure for ClO₂:
- Valence Electrons: 7
- Non-bonding Electrons: 1 (That single, unpaired electron)
- Bonding Electrons: 6 (One double bond has 4 electrons, one single bond has 2 electrons)
Formal Charge = 7 - 1 - (1/2 * 6)
Formal Charge = 7 - 1 - 3
Formal Charge = +3
Wowza! A +3 formal charge on chlorine in ClO₂! This indicates that this particular Lewis structure might not be the most accurate representation, or that the electron distribution is quite polarized. It's a reminder that these formal charges are tools, not absolute truths. They guide us, but the actual behavior of molecules is a complex dance.
Sometimes, chlorine can even be bonded to itself! Like in Cl₂, molecular chlorine. This is a simple diatomic molecule where two chlorine atoms are joined by a single covalent bond. Both atoms are identical, so they share the electrons equally.
For each chlorine atom in Cl₂:
- Valence Electrons: 7
- Non-bonding Electrons: 6 (Each Cl atom usually has 3 lone pairs)
- Bonding Electrons: 2 (The single bond between them)
Formal Charge = 7 - 6 - (1/2 * 2)
Formal Charge = 7 - 6 - 1
Formal Charge = 0
And just like in HCl, both chlorine atoms in Cl₂ have a formal charge of 0. This makes perfect sense because they are identical atoms sharing electrons perfectly equally. No one's getting a better deal. It's the epitome of molecular harmony.
So, to recap our adventure into chlorine's formal charges: it's all about counting those valence electrons, the ones chilling on their own (non-bonding), and the ones being shared (bonding). The formula is your trusty sidekick: Valence Electrons - Non-bonding Electrons - 1/2 Bonding Electrons.
We've seen chlorine go from a happy -1 in the chloride ion to a neutral 0 in HCl and Cl₂, and then swing to a rather surprising +1 in the perchlorate ion (in its most stable Lewis structure). And we even saw it hit a theoretical +3 in ClO₂ under one drawing! It just goes to show how versatile and, dare I say, fascinating our good ol' chlorine can be.
The beauty of calculating formal charge is that it's a universal tool. You can use it for any atom in any molecule! It's like learning to read a map; once you know how to read it, you can navigate anywhere. So next time you see a chlorine atom, don't just see a blob. See a potential for charge, a story of electron sharing, a piece of the molecular puzzle. And with a little bit of counting and a dash of formula magic, you can figure out its formal charge. Pretty cool, right?
Keep practicing, keep questioning, and don't be afraid to draw those Lewis structures out. They are your best friends in this electron-counting game. And hey, if you ever get stuck, just remember our coffee-fueled chat. We've got this! Happy calculating!
