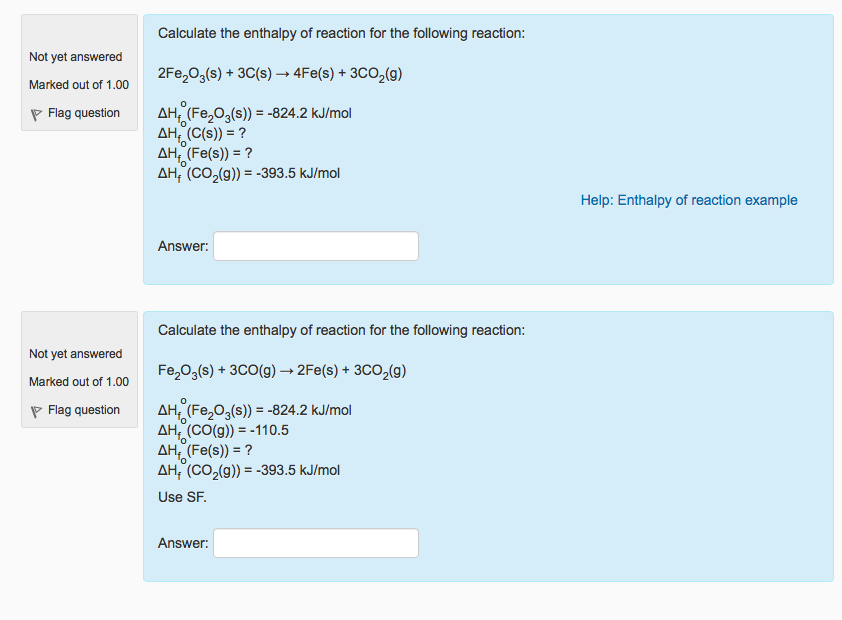
Calculate The Enthalpy For The Following Reaction
Hey there, amazing curious minds! Ever find yourself staring at a chemical reaction and thinking, "What's the deal with all this energy stuff?" Well, buckle up, buttercups, because we're about to dive into something super cool: calculating the enthalpy for a reaction. And guess what? It's not just for scientists in lab coats! It can actually make understanding the world around you a whole lot more… sparkly.
So, what exactly is enthalpy? Think of it as the total heat content of a system. When a chemical reaction happens, energy is either released (like a little explosion of warmth!) or absorbed (making things feel chilly). Enthalpy change, often shown as 'ΔH' (pronounced "delta H," which sounds way cooler than it looks, right?), tells us exactly how much heat is involved in that transformation. It's like the reaction's energy budget!
Why should you care? Well, imagine you're baking a cake. You're adding energy (heat!) to transform flour, eggs, and sugar into something delicious. Understanding enthalpy helps us predict how much energy is needed or released. It's the science behind why your oven gets hot, why a fire burns, or even why your body uses energy to keep you going!
Now, the prompt here is to "Calculate The Enthalpy For The Following Reaction." That sounds a tad intimidating, doesn't it? Like a pop quiz you weren't prepared for. But fear not! While the prompt itself is a bit of a cliffhanger (what is the reaction?!), the process of figuring out that enthalpy is where the fun really begins. It’s a bit like being a detective, gathering clues to solve an energy mystery.
Unlocking the Energy Secrets!
So, how do we actually do this calculation thing? There are a few neat tricks up our sleeves. One of the most common ways is by using something called standard enthalpies of formation. These are like the energy "building blocks" for individual substances. Each molecule has its own pre-determined enthalpy of formation, representing the energy change when one mole of that substance is formed from its elements in their standard states. Pretty neat, huh?

Think of it this way: If you want to build a Lego castle (your reaction), you need to know how much energy it takes to get each individual Lego brick (your reactants) and how much energy is released when you snap them together to form the castle walls and towers (your products). Standard enthalpies of formation give us that "brick energy" information.
The formula for calculating the enthalpy change of a reaction using these building blocks is actually quite elegant. It's generally: ΔH°reaction = Σ(n * ΔH°f products) - Σ(m * ΔH°f reactants).
Whoa, hold on! Don't let those Greek letters and symbols scare you. It just means: "Sum of (number of moles of each product multiplied by its standard enthalpy of formation) MINUS the sum of (number of moles of each reactant multiplied by its standard enthalpy of formation)." So, you take all the energy "building blocks" of what you're making, and you subtract all the energy "building blocks" of what you started with. Voilà! You've got your reaction's energy change!

A Little Example to Lighten the Mood
Let's pretend our (unspecified!) reaction was something simple and everyday, like the formation of water from hydrogen and oxygen. We know that 2H2(g) + O2(g) → 2H2O(l). This is a simplified version, of course. To do the actual calculation, we'd need the specific standard enthalpies of formation for H2, O2, and H2O.
Now, the standard enthalpy of formation for elements in their standard states (like H2 and O2 as gases) is zero. It's like they're already "formed" and don't require any energy input to get them ready. The magic happens with the compound, water (H2O). Let's imagine (because we don't have the actual numbers here) that the standard enthalpy of formation for liquid water is -285.8 kJ/mol.

So, applying our formula: ΔH°reaction = [2 * ΔH°f(H2O)] - [2 * ΔH°f(H2) + 1 * ΔH°f(O2)] ΔH°reaction = [2 * (-285.8 kJ/mol)] - [2 * (0 kJ/mol) + 1 * (0 kJ/mol)] ΔH°reaction = -571.6 kJ
See? The reaction releases 571.6 kilojoules of energy! That's why burning hydrogen is used as a fuel – it gives off a lot of heat. Isn't it cool how just a few numbers can tell us so much about a process?
Beyond the Formula: Why It's Actually Fun!
Calculating enthalpy isn't just about memorizing formulas, oh no! It's about understanding the energy flow of the universe. Think about it:

- Cooking: Why does your toast get brown and crispy? Enthalpy changes are at play!
- Nature's Wonders: Geysers erupting, volcanoes spewing lava – these are all driven by massive energy releases, tied to enthalpy.
- Your Own Body: Every single process, from breathing to thinking, involves chemical reactions with corresponding enthalpy changes. You're a walking, talking, energy-transforming marvel!
When you start looking at reactions and thinking about their ΔH, you're not just looking at symbols on a page; you're peeking into the fundamental forces that shape everything around us. It makes everyday observations so much more profound. That warm mug of coffee? Enthalpy. That chilly evening air? Also enthalpy!
And the best part? The more you learn, the more you realize how interconnected everything is. Chemistry, physics, biology – they all dance together, and enthalpy is a key partner in that dance. It’s like gaining a superpower: the ability to understand and predict energy transformations!
So, even though the specific reaction for our calculation was a bit of a mystery, the process of calculating enthalpy is a gateway to understanding so much more. It’s a journey of discovery, and every step reveals a new layer of how our universe works. Don't be shy to explore! Grab a textbook, watch a video, ask questions. The world of energy is waiting, and it’s far more exciting and inspiring than you might imagine. Go forth and calculate (or at least understand what you're calculating)! You’ve got this!
