Calculate The δg Rxn Using The Following Information

Hello there, fellow enthusiasts of the wonderfully intricate world of chemical reactions! If you're anything like me, there's a certain thrill that comes from understanding the forces that drive change around us. It's like unlocking a secret language of the universe. And when we talk about calculating the Gibbs Free Energy change, or δg Rxn, for a reaction, we're essentially getting a peek into whether a reaction will happen spontaneously or if it needs a little nudge. It’s a bit like forecasting the weather – you want to know if you need to pack an umbrella or if you can enjoy a sunny day!
Why is this so useful, you ask? Well, understanding δg Rxn is fundamental to so many aspects of our lives, even if we don't realize it. Think about it: from the
food we digest
and thebatteries that power our devices
to themedicines that heal us
, chemical reactions are at play. By calculating δg Rxn, scientists and engineers can predict the feasibility and spontaneity of these processes. This means we can design more efficient industrial processes, develop new materials, and even understand the complex biological machinery within our own bodies. It's the science behind why things happen and, crucially, why they happen on their own.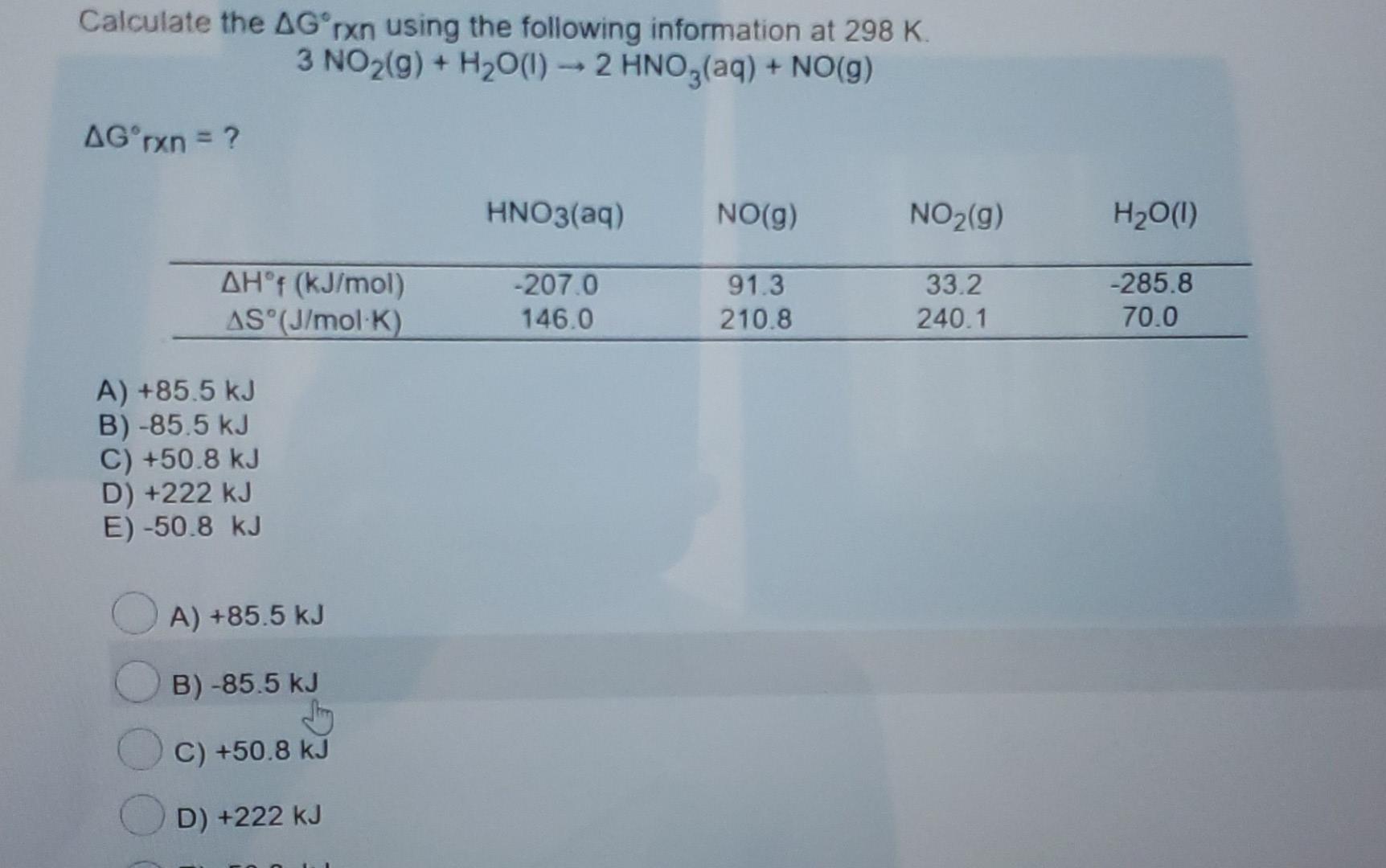
Common examples abound! Consider the simple act of
rusting of iron
. That's a spontaneous process, meaning its δg Rxn is negative. On the flip side, synthesizing a complex molecule in a lab often requires energy input, indicating a positive δg Rxn. Even something as everyday asburning fuel in your car

Now, while the actual calculation of δg Rxn often involves some serious chemistry knowledge and data, there are ways for us to engage with this concept more effectively. If you're a student or just curious, try to relate the abstract principles to tangible examples. Instead of just memorizing formulas, visualize the molecules and the energy involved. When you encounter a chemical process, ask yourself: "Does this seem like something that would happen easily, or would it require effort?" This intuitive approach can be surprisingly insightful. Also, don't be afraid to look up the δg values for common reactions – it can be fascinating to see the numbers behind everyday phenomena. Finally, if you're involved in a science class or club, actively participate in discussions and ask questions. The more you interact with the concept, the more it will solidify and become a powerful tool for understanding the world around you.
