Calculate The Density Of Each Of The Following Gases

Ever catch yourself staring at a helium balloon, wondering what makes it float so majestically? Or maybe you’ve been at a barbecue, smelling the sweet, slightly metallic scent of propane, and thought, “Huh, what’s actually in there making this work?” Well, my friends, we’re about to dive into a world of invisible wonder – the world of gas density. It sounds a bit science-y, right? Like something you’d only encounter in a dusty textbook or a lab coat-clad documentary. But trust me, understanding density is actually pretty darn cool and surprisingly relevant to our everyday lives, from the air we breathe to the appliances that make our lives a breeze.
Think of density as a sort of “heaviness per volume.” Imagine you have a box. If you fill that box with feathers, it’ll be super light. If you fill the exact same box with lead balls, it’ll be incredibly heavy. The feathers and the lead balls have different densities. Gases are the same way, just on a much smaller, invisible scale. Some gases are lighter than air, making them buoyant and floaty, while others are heavier and tend to sink.
So, how do we actually calculate this elusive property? It’s not as complicated as it sounds! At its core, density is simply mass divided by volume. In the world of gases, we usually express this as grams per liter (g/L) or kilograms per cubic meter (kg/m³). It’s like figuring out how much "stuff" is packed into a specific space. The more "stuff" (mass), the denser the gas.
Let’s break down the key players in our gas density adventure. We’ll be looking at a few common characters you’ve probably encountered, even if you didn’t realize it. We’re talking about the air you’re breathing right now, the stuff that inflates your party balloons, and even the fuel that powers your grill. Get ready to meet your invisible neighbors!
Our Everyday Air: The Undisputed Champion
First up, let’s talk about air. It’s everywhere, all the time, and we rarely give it a second thought. But air isn’t just empty space; it’s a complex mixture of gases, primarily nitrogen (about 78%), oxygen (about 21%), and a smattering of others like argon, carbon dioxide, and trace amounts of noble gases. When we talk about the density of air, we’re usually referring to this average composition.
At standard temperature and pressure (STP – which is a handy scientific shorthand for 0°C or 273.15 K and 1 atmosphere of pressure), the density of dry air is approximately 1.225 grams per liter (g/L). That’s about the weight of a small coin in a liter bottle of air. Pretty light, right? But consider the sheer volume of air surrounding our planet – it’s immense!
Fun Fact: Did you know that the density of air changes with altitude and temperature? As you go higher, there are fewer air molecules packed into the same space, so the air gets less dense. This is why airplanes need wings designed to generate lift even in thinner air, and why mountaineers sometimes struggle to breathe at high altitudes. Also, warmer air is less dense than cooler air because the molecules are moving faster and spread further apart. Think of a hot air balloon – it works precisely because the heated air inside is less dense than the cooler air outside, making it rise!
To calculate this, scientists use the ideal gas law, a fundamental equation in chemistry and physics: PV = nRT. Where P is pressure, V is volume, n is the number of moles (a way to measure the amount of substance), R is the ideal gas constant, and T is temperature. By rearranging this equation and plugging in the molar mass of air and the specific conditions, we can derive its density. For us casual observers, knowing the approximate density is often enough to appreciate its behavior.

Helium: The Life of the Party Balloon
Ah, helium! The gas that turns your voice squeaky and makes balloons defy gravity. Helium is an element, a noble gas, meaning it’s very unreactive. It’s the second lightest element in the universe, after hydrogen. Because it’s so light, it’s significantly less dense than air.
At STP, the density of helium is about 0.1786 grams per liter (g/L). Compare that to air’s 1.225 g/L. Helium is roughly seven times less dense than air! This massive difference is what gives helium balloons their impressive lift. The helium inside the balloon weighs less than the air it displaces, making the balloon buoyant.
Cultural Connection: Helium has a special place in our cultural imagination, often associated with childhood joy, birthday parties, and the iconic Macy’s Thanksgiving Day Parade. Those giant inflatable characters wouldn't float without the low density of helium. It’s also used in MRI machines because of its extremely low boiling point, but let’s stick to the fun stuff for now!
Practical Tip: While it’s fun to inhale helium for a temporary voice change, it's extremely dangerous! You can displace the oxygen you need to breathe, leading to dizziness, unconsciousness, and even death. Stick to blowing up balloons with your own lungs or a dedicated helium tank designed for that purpose!
Propane: The Barbecue Buddy
Now, let’s talk about our grill-master’s best friend: propane. Propane is a hydrocarbon, a gas commonly used as a fuel. It’s a bit more substantial than helium, and importantly, it’s denser than air.
At standard conditions, the density of propane is around 1.94 grams per liter (g/L). This is significantly denser than air (1.225 g/L). This means that if you have a propane leak, the gas will tend to sink and pool along the ground or in low-lying areas. This is a crucial safety consideration.

Safety First! Because propane is denser than air, it can accumulate in basements or crawl spaces, creating a fire or explosion hazard. If you smell propane, do not turn on any lights or appliances, as a spark could ignite it. Open windows and doors to ventilate the area and leave immediately. Call your gas company or emergency services from a safe distance.
The molecular structure of propane (C₃H₈) gives it a higher mass compared to the average molecule in air, resulting in its greater density. It’s this density that makes it behave predictably when stored in tanks and when used as a fuel, settling in the lower part of the container.
Carbon Dioxide: The Fizzy Friend (and Environmental Concern)
We all know carbon dioxide (CO₂) as the stuff that makes our soda fizzy and is a byproduct of our breathing. It’s also a significant greenhouse gas. When it comes to density, CO₂ is also denser than air.
At STP, the density of carbon dioxide is approximately 1.977 grams per liter (g/L). This is very close to the density of propane, and also noticeably heavier than air.
Fun Fact: This density difference is why firefighters often use CO₂ extinguishers. When discharged, the heavy CO₂ gas blankets the fire, displacing the oxygen needed for combustion and smothering the flames. It’s also why you might feel a slight tingling or “bite” from a very cold glass of soda – the dissolved CO₂ is escaping and interacting with your tongue.
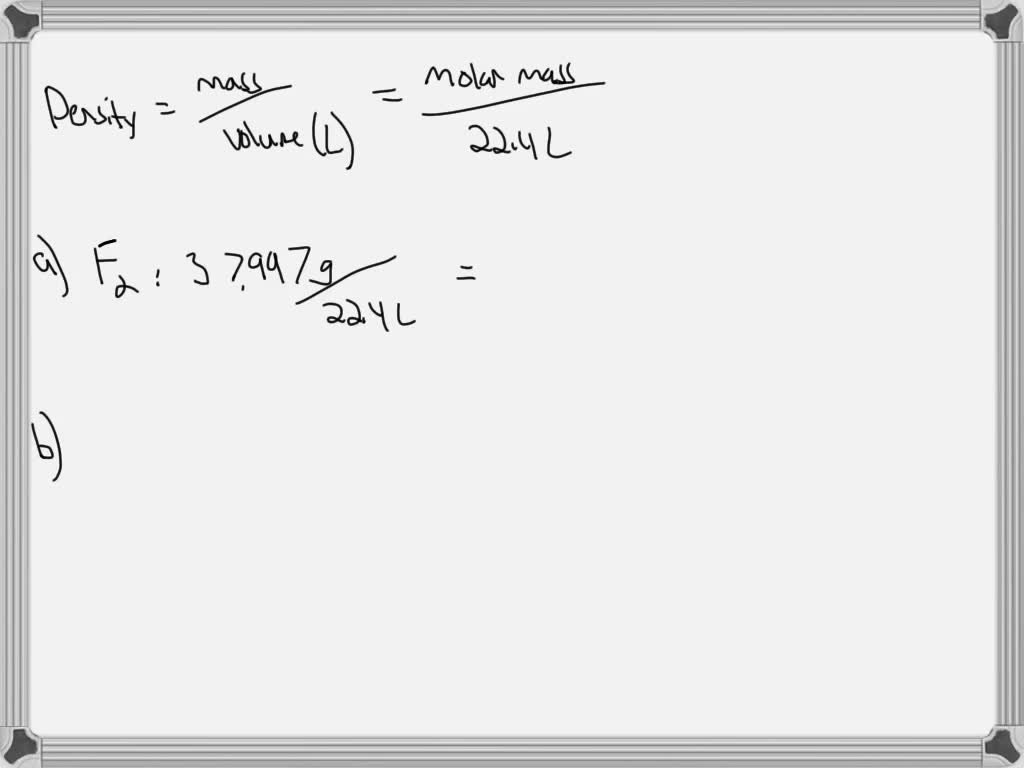
The molecular weight of CO₂ (one carbon atom and two oxygen atoms) is significantly higher than the average molecular weight of air (mostly nitrogen and oxygen), which explains its greater density. This characteristic is important in atmospheric science and climate modeling, as the distribution of CO₂ in the atmosphere affects weather patterns and temperature.
Methane: The Natural Gas Player
Finally, let’s consider methane (CH₄), the primary component of natural gas. It’s a potent greenhouse gas and a key energy source. Like propane and carbon dioxide, methane is denser than air.
At STP, the density of methane is around 0.717 grams per liter (g/L). Wait, what? I just said it’s denser than air! This is where things get a little nuanced, and it highlights the importance of specific conditions. While pure methane at STP is indeed lighter than air, the natural gas we use is often a mixture, and its overall density can be affected by the presence of heavier hydrocarbons.
However, for practical purposes related to leaks and accumulation, methane behaves in a way that can be concerning. While pure methane might rise slightly, it mixes readily with air, and under certain conditions, especially when released in large quantities, it can still pose an explosion risk. Furthermore, the odorants added to natural gas for leak detection are themselves denser, influencing the overall behavior.
Cultural Note: Methane is often associated with cows and their… emissions. While cows do produce methane, it’s just one source. Landfills, wetlands, and the production of fossil fuels are also major contributors. Understanding its properties, including how it disperses in the atmosphere, is vital for addressing climate change.
When we talk about natural gas density in a household context, it's often the safety aspect that’s emphasized, and the fact that it’s flammable is the primary concern. The density differences with air are more relevant in large-scale atmospheric studies or industrial processes.

Putting It All Together: Why Does This Even Matter?
So, we’ve calculated (or at least discussed the approximate densities of) air, helium, propane, carbon dioxide, and methane. Why should you, a person living their best, easy-going life, care about these numbers?
For starters, it’s a fantastic way to understand the invisible forces at play all around us. That helium balloon? Its lightness is a direct consequence of its low density. Your gas stove? It works because propane (or natural gas) is readily available and has specific combustion properties tied to its chemical makeup and density. The air we breathe, a complex and life-sustaining mixture, has a density that changes with temperature and altitude, influencing everything from flight to our ability to hike a mountain.
Understanding these densities also translates into practical safety knowledge. Knowing that propane and carbon dioxide are denser than air helps us understand why leaks can accumulate in low areas, posing a fire or asphyxiation risk. It’s about being informed and prepared, even in subtle ways.
Furthermore, it’s a gateway to appreciating the elegance of science. The simple formula of density = mass / volume, applied to these invisible gases, unlocks a deeper understanding of our world. It’s a reminder that even the most commonplace phenomena have underlying principles that are both fascinating and essential.
Imagine this: the next time you’re at a picnic and see someone inflating a balloon, you can have a little internal monologue about the density of helium versus air. Or when you’re cooking and smell the comforting aroma of your gas stove, you can appreciate the science behind that fuel. It’s about adding a layer of informed appreciation to your daily experiences.
A Moment of Reflection
It’s quite remarkable, isn’t it? We move through a world saturated with gases, their densities dictating their behavior, their interactions, and their impact on our lives. From the lift of a celebratory balloon to the warmth of our homes, these invisible properties are constantly at work. Calculating the density of each gas might seem like a technical exercise, but it’s really an invitation to see the unseen, to appreciate the subtle science that underpins our everyday existence. It’s a gentle reminder that even the most ordinary moments are, in fact, quite extraordinary.
