Calculate The Delta H For The Following Reaction
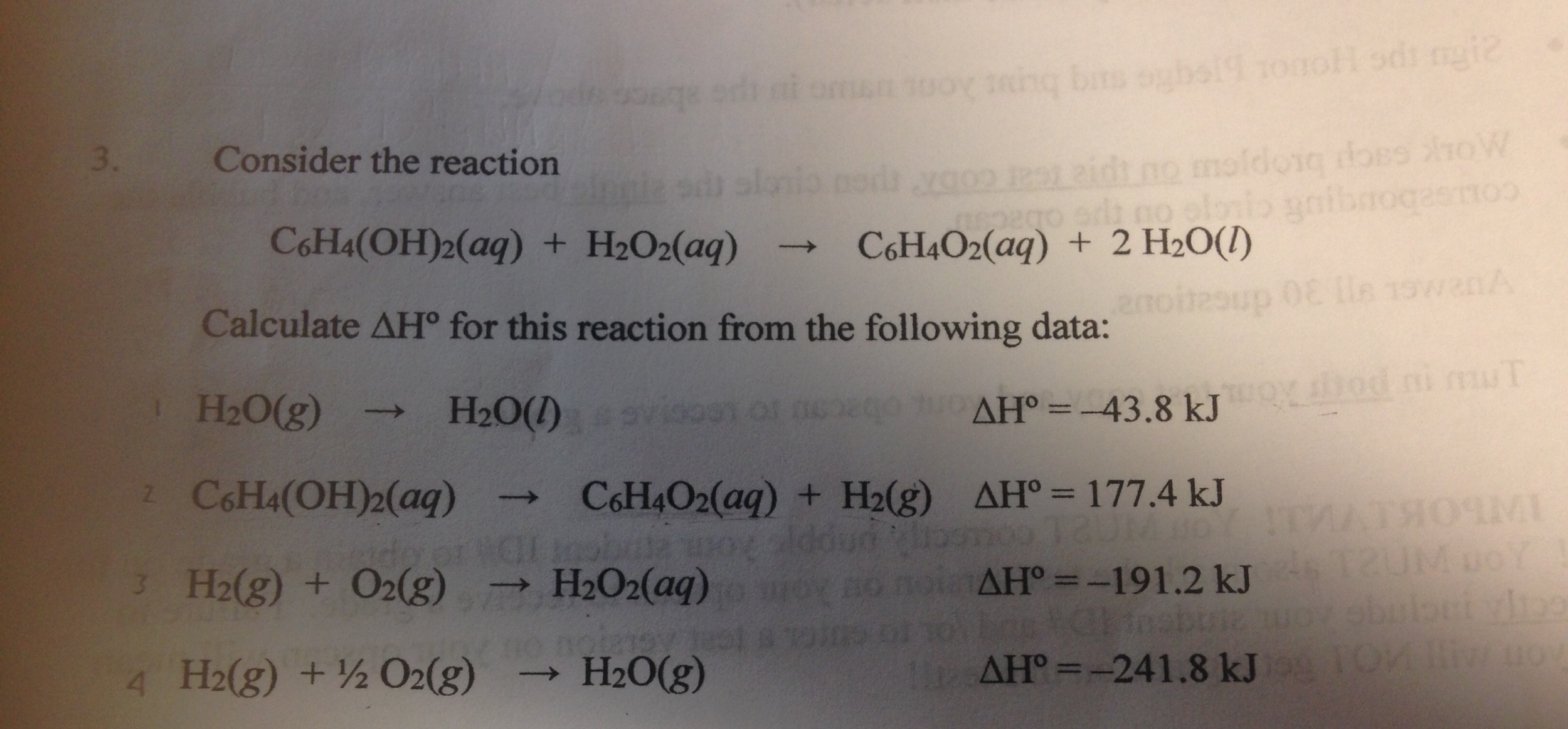
Hey there, you marvelous multitasker! Ever find yourself staring at a complicated equation and thinking, "Is there a chill way to break this down?" Well, buckle up, buttercup, because today we're diving into the wonderfully chill world of calculating Delta H. Don't let the fancy name fool you; it's basically a way to understand how much energy a chemical reaction is going to throw around. Think of it as the reaction's mood swing – is it going to be a cozy, heat-releasing hug (exothermic) or a demanding, energy-sucking vortex (endothermic)? We're about to find out!
Imagine you’re whipping up a batch of your famous chocolate chip cookies. That glorious smell wafting through your kitchen? That's energy being released – a tiny, delicious exothermic reaction. Or maybe you're trying to get that stubborn ice cube to melt on a warm day. You're adding energy to that system, coaxing it into a liquid state. That's an endothermic vibe. Our chemical reactions work on similar principles, just on a much more precise, scientific level.
So, what exactly is Delta H? In the grand theatre of chemistry, Delta H (ΔH) represents the change in enthalpy during a reaction. Enthalpy, in simple terms, is the total heat content of a system. When a reaction happens, some bonds are broken, and new ones are formed. This bond-breaking business costs energy, while bond-forming releases energy. The net result of these energy exchanges is our Delta H. Easy peasy, right?
If ΔH is negative, it means the reaction is exothermic. It’s giving off heat, like a perfectly toasted marshmallow on a campfire. Your kitchen is going to feel warmer, and you might even see some steam. Think of fireworks exploding – lots of heat and light released!
If ΔH is positive, the reaction is endothermic. It’s sucking up heat from its surroundings, making things feel cooler. Think of those instant cold packs you might use for a sprained ankle. They get cold because they’re absorbing heat. It's like a demanding diva who needs all the attention (and energy)!
Now, how do we actually calculate this moody Delta H? There are a few cool ways to do it, depending on the info you've got. Today, we're going to tackle one of the most common and frankly, quite elegant, methods: using standard enthalpies of formation. This is where things get a little like being a detective, piecing together clues to solve the energetic puzzle.
The Standard Enthalpies of Formation Game
Imagine you have a recipe book, but instead of ingredients for a killer lasagna, it’s filled with the energy it takes to create individual compounds from their most basic, stable elements. That, my friends, is essentially what standard enthalpies of formation (ΔH°f) are. They represent the enthalpy change when one mole of a compound is formed from its constituent elements in their standard states, under standard conditions (usually 298 Kelvin or 25°C and 1 atmosphere pressure).
So, if you're looking to calculate the Delta H for a reaction like:
A + B → C + D
The magic formula you'll want to have on your radar is:
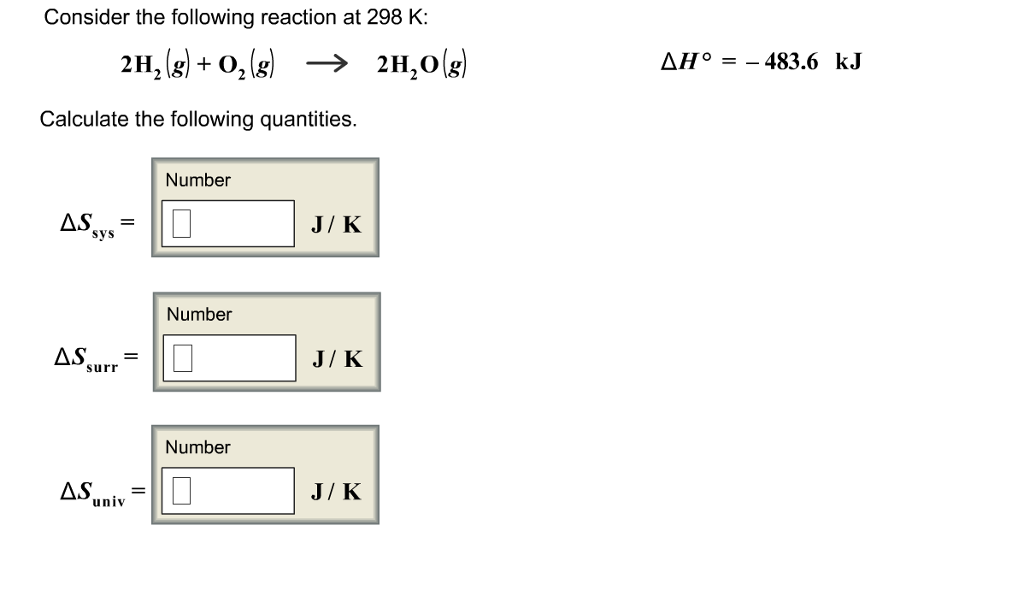
ΔH°reaction = Σ (n * ΔH°f)products - Σ (m * ΔH°f)reactants
Whoa, hold up! Don't panic. Let's break it down. Think of 'Σ' (sigma) as a fancy way of saying "add them all up." 'n' and 'm' are the stoichiometric coefficients – those little numbers in front of the chemical formulas in your balanced equation. They tell you how many moles of each reactant or product are involved. And of course, ΔH°f is our trusty standard enthalpy of formation.
In plain English: You add up the enthalpies of formation of all your products, then you subtract the sum of the enthalpies of formation of all your reactants. It’s like balancing your energetic budget – what’s coming in versus what’s going out.
Let's Get Practical: An Example to Chew On
Alright, theory is great, but let's get our hands dirty with a classic example. Consider the combustion of methane (natural gas) – something we see all the time, whether it's powering our stoves or heating our homes.
The balanced reaction is:
CH4(g) + 2O2(g) → CO2(g) + 2H2O(l)
Now, we need our handy-dandy reference table for the standard enthalpies of formation (ΔH°f) of each species:
- ΔH°f [CH4(g)] = -74.8 kJ/mol
- ΔH°f [O2(g)] = 0 kJ/mol (Elements in their standard state have a ΔH°f of zero. Think of it as them being their own baseline.)
- ΔH°f [CO2(g)] = -393.5 kJ/mol
- ΔH°f [H2O(l)] = -285.8 kJ/mol
See how oxygen gas (O2) has a ΔH°f of 0? That's because it's an element in its most stable form. It doesn't cost or gain energy to form oxygen from oxygen. It's already there, chilling!
Now, let's plug these values into our formula:
ΔH°reaction = [ (1 * ΔH°f [CO2(g)]) + (2 * ΔH°f [H2O(l)]) ] - [ (1 * ΔH°f [CH4(g)]) + (2 * ΔH°f [O2(g)]) ]
Plugging in the numbers:
ΔH°reaction = [ (1 * -393.5 kJ/mol) + (2 * -285.8 kJ/mol) ] - [ (1 * -74.8 kJ/mol) + (2 * 0 kJ/mol) ]
Let's do the math, step-by-step, like we're assembling a bomb playlist:
First, the products:
- CO2: 1 * -393.5 = -393.5 kJ/mol
- H2O: 2 * -285.8 = -571.6 kJ/mol
- Total products: -393.5 + (-571.6) = -965.1 kJ/mol
Next, the reactants:

- CH4: 1 * -74.8 = -74.8 kJ/mol
- O2: 2 * 0 = 0 kJ/mol
- Total reactants: -74.8 + 0 = -74.8 kJ/mol
Now, subtract the reactants from the products:
ΔH°reaction = (-965.1 kJ/mol) - (-74.8 kJ/mol)
Remember, subtracting a negative is the same as adding a positive:
ΔH°reaction = -965.1 kJ/mol + 74.8 kJ/mol
ΔH°reaction = -890.3 kJ/mol
Boom! We just calculated that the combustion of methane is an exothermic reaction, releasing a significant amount of energy (-890.3 kJ per mole of methane burned). This is why natural gas is such a popular fuel source – it packs a lot of energy!
Fun Little Facts and Cultural Tidbits
Did you know that the concept of enthalpy was introduced by Josiah Willard Gibbs in 1876? He was a total rockstar in the world of thermodynamics. And speaking of energy, have you ever been fascinated by the sheer power of volcanic eruptions? That's a seriously exothermic event, and while we can't calculate its ΔH with a simple formula, understanding the principles of energy release helps us appreciate the raw power of nature.
Think about the simple act of boiling water for your morning tea. The energy you're adding to heat the water is an endothermic process. The steam that rises, however, contains a lot of potential energy. When that steam condenses or is used to power something, it releases that energy. It’s a constant dance of energy absorption and release all around us.
And here’s a cool one: In cooking, many reactions are exothermic. Searing a steak, for example, involves complex chemical reactions that release heat and create those delicious browned flavors. The Maillard reaction, responsible for much of that browning, is exothermic! So, when you’re whipping up a culinary masterpiece, you’re essentially performing controlled exothermic reactions.
Another fun fact: the phrase "burn calories" is actually a nod to exothermic reactions. When your body metabolizes food, it’s breaking down complex molecules to release energy, and a byproduct is heat. So, your body is constantly running a series of mini, controlled exothermic processes to keep you going!
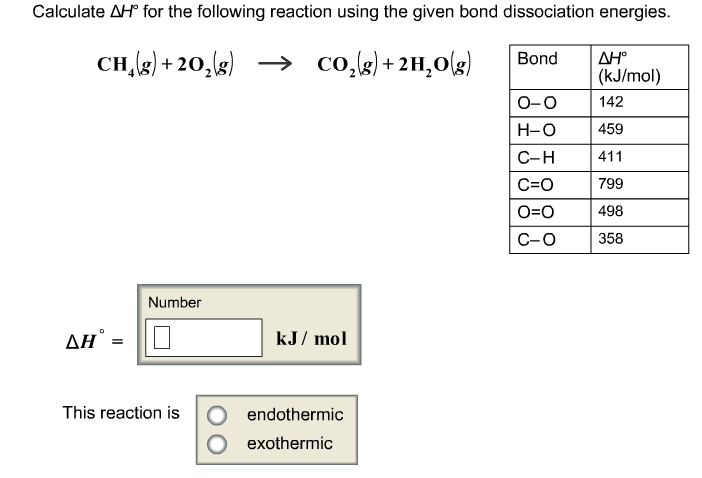
You might also encounter reactions where you need to calculate ΔH using bond energies. This is another cool method, where you look up the average energy required to break specific chemical bonds and the energy released when those bonds are formed. It's a bit like deconstructing a Lego castle and then building a new one – you need to know how much force it takes to pull bricks apart and how much energy is released when they snap together.
The formula for bond energies is similar: you sum the energy required to break bonds in the reactants and subtract the energy released when forming bonds in the products. It’s a different perspective, but it leads to a similar understanding of the overall energy change.
Practical Tips for Your Energetic Journey
- Always balance your equation first! Seriously, this is non-negotiable. Those stoichiometric coefficients (the little numbers) are crucial for getting the right answer.
- Know your standard states. Remember that elements in their standard states (like O2(g), H2(g), C(graphite)) have a ΔH°f of zero. This is a common trick question element.
- Pay attention to the state symbols. Are you dealing with a gas (g), liquid (l), solid (s), or aqueous (aq) substance? The enthalpy of formation can differ significantly between states, especially for water.
- Double-check your signs. Minus signs can be tricky! Make sure you're correctly adding and subtracting, especially when dealing with negative values.
- Keep your reference table handy. You won't be expected to memorize all these values. Most chemistry courses and textbooks provide tables of standard enthalpies of formation.
- Practice makes perfect. The more reactions you calculate, the more comfortable you'll become with the process. It's like learning a new dance move – the more you do it, the smoother it gets.
And for a little extra flair, try to find real-world examples related to the reactions you're calculating. If you're working with a combustion reaction, think about the fuel. If it's a neutralization reaction, picture a science experiment with acids and bases. It helps to make the abstract tangible.
A Moment of Reflection
So, we've learned that calculating Delta H is essentially understanding the energetic personality of a chemical reaction. It tells us whether a reaction is going to be a warm hug or a cool breeze. In our daily lives, we’re constantly interacting with these energetic shifts, often without even realizing it. From the food we eat that fuels our bodies (exothermic metabolism) to the way our homes stay warm (exothermic heating systems), energy is the invisible conductor orchestrating much of our world.
Even something as simple as taking a deep breath and exhaling is a part of the ongoing thermodynamic processes that keep us alive. The air we inhale is processed, and energy is released to power our cells. It’s a beautiful, intricate dance. So, the next time you're feeling a chill or a warmth, remember that behind those sensations are chemical reactions, each with its own unique Delta H, its own energetic story to tell. And now, you've got a little more insight into how to read those stories. Pretty cool, huh?
