Calculate The Approximate Number Of Atoms In A Bacterium

Hey there, coffee buddy! Ever looked at one of those tiny little germy things – you know, bacteria – and wondered, "How many bits are actually in there?" It's a question that pops into your head, right? Like, a really tiny, microscopic kind of question. Well, buckle up, because we're going to dive into the wild world of bacterial atom counting. No fancy lab coats needed, just a good ol' dose of curiosity and some clever math. Sounds fun, doesn't it?
So, bacteria. They’re everywhere. Seriously, everywhere. On your phone, in your gut, probably floating around this very room. They’re like the ultimate survivalists of the microscopic world. And they're, well, pretty darn small. We’re talking about things you absolutely cannot see without a microscope. Like, really can’t see. So how on earth do we even begin to guess how many atoms are squished inside one of these little dudes?
It’s not like we can just put a bacterium on a scale, is it? "Oh, this E. coli weighs 0.0000000000001 grams. Easy peasy." Nope. Scales are for, like, vegetables and yourself after a questionable pizza. For bacteria, we gotta get a bit more… creative. Think of it like trying to count the individual grains of sand on a beach. Impossible to count them one by one, right? But you can still get a pretty good idea of the sheer volume, and then make some educated guesses.
The whole process is basically a giant game of "educated guess meets some pretty cool science." We're not getting an exact, down-to-the-last-atom number, okay? Let’s be real, that would be insane. But we can get a really good approximation. Think of it as a scientifically sound ballpark figure. And honestly, for something so microscopic, a ballpark figure is pretty darn impressive.
The Big Picture: What Are We Even Made Of?
Before we cram atoms into our tiny bacterial friends, let's chat about what we’re made of. And, by extension, what everything else is made of. Atoms. Duh. But which ones? Mostly, it’s a handful of the big players. For us humans, and for bacteria too, there's a lot of oxygen. Yep, that stuff we breathe? It’s a major component. Then there’s carbon. Hello, organic life! This is where all the building blocks of cells come from. And hydrogen, another essential ingredient. Don't forget nitrogen, phosphorus, and sulfur – the gang’s all here, making life happen.
These six elements – carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur – make up a huge chunk of any living organism. We’re talking, like, 99% of the mass. The remaining 1% is all the other cool trace elements that do important jobs, but for our big-picture atom count, focusing on the main guys is totally sufficient. It's like trying to count all the people at a concert. You can get a general idea of the crowd size by looking at the general density, rather than stopping to count every single person's shoelaces. You get it?
So, when we talk about a bacterium, we're essentially talking about a tiny, self-contained chemical factory. And like any factory, it has a certain composition. It’s made of proteins, DNA, fats, sugars… all these fancy molecules. And guess what those molecules are made of? You guessed it! Our star elements: carbon, hydrogen, oxygen, and so on.
Breaking Down the Bacterium: A Tiny Package of Elements
Now, let's get a little more specific. Scientists, bless their nerdy hearts, have figured out the approximate elemental composition of a typical bacterium. It’s not like every single bacterium on Earth is identical, of course. There’s a whole universe of bacterial diversity out there! But for the sake of a good approximation, we can use an average. It’s like saying the average human has two arms and two legs. There are exceptions, but it’s a good starting point, right?
On average, a bacterium is roughly 70% water. Water, H2O. So, a lot of hydrogen and oxygen right there. The dry weight of a bacterium? That’s where the other elements really shine. Carbon is usually around 50% of the dry weight. This makes sense, as carbon is the backbone of all organic molecules. Then comes nitrogen, often around 15-20%. This is crucial for proteins and DNA. Oxygen, in its bound form within molecules, is also significant, maybe around 20-25%. Phosphorus and sulfur are also in the mix, playing vital roles.
The percentages might seem a little fluid, and they are! Different bacteria have different diets and lifestyles. A bacterium living in a sulfur-rich environment might have more sulfur. One that’s growing like crazy might have a slightly different protein-to-lipid ratio. But these are generally accepted approximations. Think of them as the fundamental recipe for bacterial life. It’s like a secret menu item that most fast-food joints are working from.

The Size Factor: How Small Are We Talking?
Okay, we know what they’re made of, but how big are these things? This is where the “microscopic” part really hits home. Most bacteria are measured in micrometers. A micrometer, or micron, is one-millionth of a meter. A meter is, like, the length of your arm. So, a micrometer is… tiny. Like, so small you can’t even imagine it. If you could shrink yourself down to the size of a bacterium, the world would look like a giant, alien landscape. Imagine walking through a forest of dust bunnies!
A typical bacterium might be anywhere from 0.5 to 5 micrometers in length. Let’s take a common bacterium, like E. coli. It's usually around 1-2 micrometers long and about 0.5 micrometers wide. So, we’re looking at a little rod-shaped thing. Not exactly a towering skyscraper, is it?
To get a sense of scale, if you laid 1,000 bacteria end-to-end, they might stretch for about a millimeter. A millimeter! That’s the thickness of a fingernail. So, you could fit a thousand of these little guys across the width of your pinky finger. Mind-blowing, right? This is why we need microscopes. Without them, these amazing little life forms would be completely invisible to us.
Volume and Density: Packing 'Em In
Now, to estimate the number of atoms, we need to think about volume. Since bacteria are roughly cylindrical or rod-shaped, we can use some basic geometry to estimate their volume. Let’s say our average bacterium is a cylinder with a length (L) of 1 micrometer and a diameter (D) of 0.5 micrometers. The radius (R) would then be half the diameter, so 0.25 micrometers.
The formula for the volume of a cylinder is V = π * R² * L. Plugging in our numbers (and keeping everything in micrometers for now): V = π * (0.25 µm)² * (1 µm) ≈ 0.196 cubic micrometers (µm³).
This is the volume of one bacterium. It’s a very small volume, obviously. But what about the mass of that volume? That’s where density comes in. The density of a bacterium is roughly similar to the density of water, which is about 1 gram per cubic centimeter (g/cm³). However, since a bacterium is mostly organic molecules, its density might be slightly higher, let’s estimate around 1.1 g/cm³ to be a bit more precise. This is where things get a little tricky with units. We need to convert cubic micrometers to cubic centimeters. And let me tell you, that’s a lot of zeros involved!
1 centimeter = 10,000 micrometers. So, 1 cubic centimeter (cm³) = (10,000 µm)³ = 1,000,000,000,000,000 µm³ (that’s a quadrillion!).

So, our 0.196 µm³ bacterium has a mass of: Mass = Density * Volume. Mass ≈ 1.1 g/cm³ * (0.196 µm³ / 10¹⁵ µm³/cm³) Mass ≈ 1.1 g/cm³ * 0.000000000000196 cm³ Mass ≈ 0.0000000000002156 grams.
See? We’re already getting into numbers so small they’re practically invisible. This is why we need fancy tools and methods!
From Mass to Atoms: The Magical Calculation
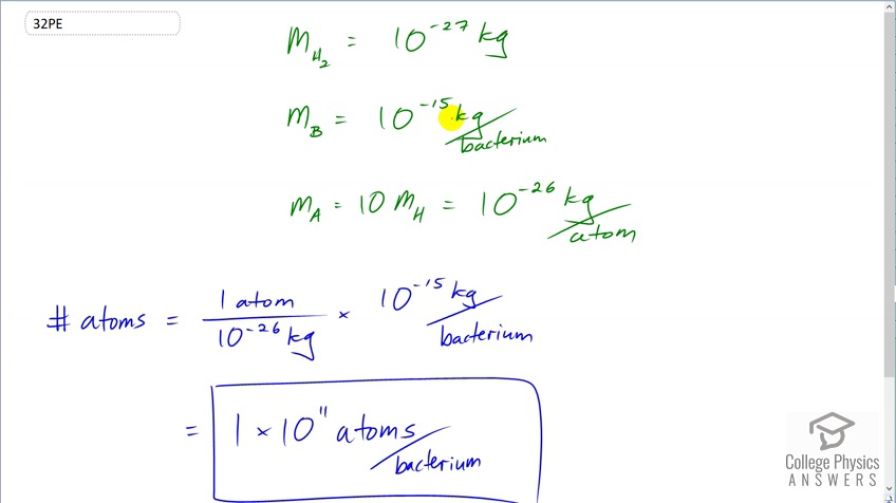
Okay, we’ve got the approximate mass of a bacterium. Now, how do we go from mass to atoms? This is where the concept of the mole and Avogadro's number comes into play. Don’t let the fancy terms scare you; it's actually a really neat idea. A mole is just a specific amount of a substance, like a dozen is 12 of something. A mole is a huge number of something: Avogadro's number, which is approximately 6.022 x 10²³. That’s a 6 followed by 23 zeros!
The beauty of the mole is that it links the mass of a substance to the number of atoms or molecules it contains. For any element, its molar mass (the mass of one mole) in grams is numerically equal to its atomic weight on the periodic table. For example, carbon has an atomic weight of about 12. So, 12 grams of carbon contains about 6.022 x 10²³ carbon atoms.
Since we know the approximate elemental composition of a bacterium, we can estimate the number of atoms of each element and then add them all up. This is where it gets a little bit of estimation upon estimation, but it’s still a solid approach.
Let's Crunch Some Numbers (Roughly!)
Let’s assume our bacterium (with its approximate mass of 2.156 x 10⁻¹³ grams) is composed as follows (percentages are of total mass, not dry weight here):
- Water (H₂O): ~70%
- Proteins, nucleic acids, etc. (rich in C, N, O, P, S): ~30%
This gets complicated quickly if we try to break down every single molecule. So, a more common and simpler approach is to use an average atomic mass for the bacterium, based on its elemental composition.
If we consider the main elements (H, C, N, O, P, S), and their approximate proportions, we can calculate an average atomic mass. A common approximation for the average atomic weight of a living cell is around 10-15 atomic mass units (amu). Let's use an average of 12 amu for our simplified calculation. This means, on average, each "atom" in our bacterium weighs about 12 times as much as a hydrogen atom (which has an atomic mass of about 1 amu).

To convert this average atomic mass to grams, we divide by Avogadro’s number. Mass of one "average atom" ≈ 12 amu / (6.022 x 10²³ atoms/mole) ≈ 1.99 x 10⁻²³ grams/atom.
Now, we can divide the total mass of the bacterium by the average mass of an atom to get the approximate number of atoms:
Number of Atoms ≈ Total Mass of Bacterium / Average Mass per Atom Number of Atoms ≈ (2.156 x 10⁻¹³ grams) / (1.99 x 10⁻²³ grams/atom)
Let’s do the math… Drumroll please… 🥁
Number of Atoms ≈ 1.08 x 10¹⁰ atoms!
So, that’s roughly ten billion atoms in a single, average bacterium. Ten billion! That’s a lot of tiny, tiny building blocks crammed into something invisible to the naked eye. It’s like a super-miniature, super-dense universe. Wild, isn't it?
A Little Caveat (Because Science is Never That Simple)
Now, I want to stress that this is an approximation. A really, really good approximation, but still an approximation. The actual number can vary depending on the size, shape, and specific composition of the bacterium. Some might have more water, some might have more protein. Some might be bigger, some might be smaller. It’s like trying to estimate how many jellybeans are in a jar – you can get a good guess, but it’s not going to be perfect down to the last bean unless you’re willing to spill them all out and count!
Also, the "average atomic mass" is a simplification. We’re not really dealing with individual atoms with a mass of exactly 12 amu. We have a mix of hydrogen (1 amu), oxygen (16 amu), carbon (12 amu), and so on. Calculating the exact number of each element and then summing them up would be more precise, but this average method gives us a very strong ballpark figure.
But even with the approximations, the takeaway is huge: bacteria are incredibly complex, packed with an astronomical number of atoms. It’s a testament to the incredible efficiency and intricate design of life at its smallest scales. It really makes you appreciate the complexity of even the most seemingly simple organisms. Who knew something so small could contain so much?
The Bigger Picture: Why Does This Even Matter?
Okay, so we’ve calculated that a bacterium has roughly 10 billion atoms. Cool, right? But why do we even bother with these calculations? Is it just to impress your friends at your next science-themed party? (Though that would be a pretty killer party trick!) There are actually some really important reasons why understanding the atomic composition and scale of bacteria is crucial.
Firstly, it helps us understand how life works at its most fundamental level. Knowing the building blocks allows us to understand how cells function, how they replicate, and how they interact with their environment. It’s like understanding the ingredients in a cake to know how it’s baked and what it will taste like.
Secondly, this knowledge is vital for fields like medicine and biotechnology. If we want to develop antibiotics, for example, we need to understand the molecular machinery of bacteria. How do these drugs interfere with essential processes? How can we target them effectively without harming our own cells?
And don’t forget environmental science! Bacteria play a massive role in nutrient cycling and breaking down organic matter. Understanding their scale and composition helps us model these processes and understand their impact on ecosystems. They are the unsung heroes of our planet’s recycling system!
A Universe in a Cell
Thinking about 10 billion atoms in something invisible is, frankly, mind-boggling. It really puts things into perspective, doesn't it? We are all made of atoms, from the largest whale to the tiniest bacterium. And the universe itself is just a massive collection of these fundamental building blocks, organized in countless ways. It’s a beautiful, interconnected dance of matter and energy.
So, the next time you see a picture of a bacterium or hear about them in the news, remember this little calculation. Remember that within that minuscule speck of life lies an astonishing universe of atoms, working together to keep the wheels of life turning. It’s a constant reminder of the incredible complexity and wonder that exists all around us, even in the places we can’t see. Pretty neat, huh? Now, where's that refill of coffee? This atom-counting talk has made me thirsty!
