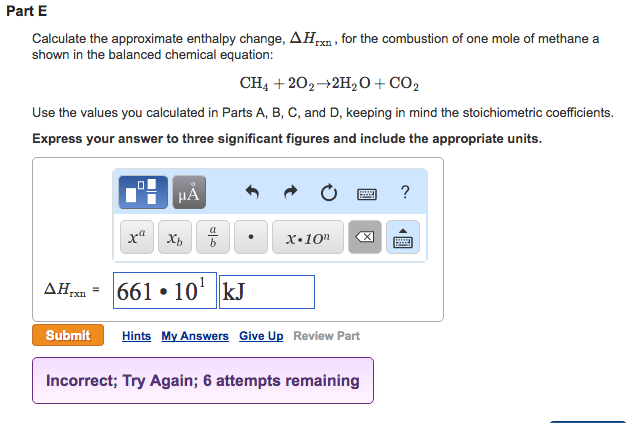
Calculate The Approximate Enthalpy Change For The Combustion Of Methane

Ever wondered about the fiery magic that powers our world? We're talking about burning stuff, plain and simple. But what if we told you there's a way to calculate the heat produced when something like methane, the main ingredient in natural gas, goes up in flames? It sounds like science fiction, right? But it's totally doable, and honestly, it's pretty darn cool.
Think of it like this: when you light a gas stove, you're witnessing a chemical reaction. Methane (which is just carbon and hydrogen hanging out together) meets oxygen from the air, and poof! You get fire. This fire releases energy, and that energy is what we call enthalpy change. It’s essentially the heat hug or heat slap that comes out of the reaction.
Now, calculating this might sound intimidating. We're not talking about complex equations that require a PhD in chemistry. We're aiming for an approximate answer, a good guess that gives you a feel for the energy involved. It's like estimating how many cookies you'll bake based on the flour you have. You might not be exact, but you get a pretty good idea.
So, what’s so special about this calculation? Well, it’s like uncovering a hidden secret of the universe. You’re taking something you see every day – fire – and peering into its energetic soul. It’s a peek behind the curtain of nature's pyrotechnics, and it’s surprisingly accessible.
Let's dive into the fun part. We're going to use something called Hess's Law. Don't let the name scare you; it's more like a clever trick for accountants. Imagine you want to know the total cost of a trip, but you can't remember the exact price of each leg. Hess's Law says you can still figure out the total cost by adding up the costs of smaller, related trips. It’s all about the starting and ending points, not the winding path in between.
In our combustion party, the starting point is methane and oxygen hanging out separately. The ending point is carbon dioxide and water, the happy (or steamy) results of the fire. We can find the "cost" of getting from start to finish by using the "costs" of other, easier-to-calculate chemical reactions. It's like having a cheat sheet for energy transformations.
These "costs" are usually found in tables of standard enthalpies of formation. Think of these as the energy price tags for creating molecules from their basic building blocks (like carbon atoms, hydrogen atoms, and oxygen atoms) in their most stable forms. For example, the table tells you how much energy it takes (or is released) to build a methane molecule from scratch. It's like knowing the price of individual LEGO bricks before you build a whole spaceship.
So, the big idea is to find a series of known reactions that, when added together, exactly equal our combustion of methane reaction. Then, we can just add up the energy changes from those known reactions. It’s a chemical puzzle, and solving it feels incredibly satisfying. You’re playing a game of chemical origami, folding and unfolding reactions until you get the one you want.

Let’s imagine our target reaction: methane burning in oxygen. We write it out like this: CH4 + 2O2 → CO2 + 2H2O. This tells us one molecule of methane plus two molecules of oxygen become one molecule of carbon dioxide and two molecules of water. Simple, right? But the real magic is in the energy exchanged.
Now, for the trick. We need to find other reactions that involve CH4, O2, CO2, and H2O, and whose enthalpy changes we know. A super helpful set of reactions are the formation reactions themselves. Remember those price tags?
For instance, we can find the enthalpy change for forming methane (C + 2H2 → CH4). We can also find it for forming carbon dioxide (C + O2 → CO2) and water (H2 + ½O2 → H2O). These are our building blocks!
But there's a slight twist, a little puzzle piece we need to flip. Our combustion reaction uses up methane, but the formation reaction creates it. So, when we use a formation reaction that creates something our target reaction uses, we have to reverse the sign of its enthalpy change. It's like getting a refund instead of paying! If forming methane releases energy, then breaking down methane requires energy.
Similarly, our combustion reaction creates carbon dioxide and water. The formation reactions for these will tell us how much energy is involved in making them. This is exactly what we want! So, the formation enthalpies for CO2 and H2O will be used as they are.
And what about oxygen? Oxygen (O2) is already in its most stable form. So, the enthalpy of formation for O2 is zero! It’s like getting free fuel in our calculation. This makes things much simpler.

So, the formula we often use for combustion reactions, derived from Hess's Law, looks something like this: ΔHcombustion = Σ(ΔHf° products) - Σ(ΔHf° reactants). This means we add up the standard enthalpies of formation for everything we made (the products) and then subtract the sum of the standard enthalpies of formation for everything we started with (the reactants).
Let's plug in our methane combustion: CH4 + 2O2 → CO2 + 2H2O.
Our products are CO2 and H2O. So we'll need the ΔHf° for CO2 and H2O. Since there are two water molecules, we'll multiply its enthalpy of formation by two.
Our reactants are CH4 and O2. We'll need the ΔHf° for CH4 and O2. Remember, ΔHf° for O2 is zero!
When you look up these values in a trusty chemistry textbook or online, you'll find numbers that usually have a negative sign. This negative sign is a big deal! It means energy is released during these formation processes. For example, forming methane from its elements releases energy. When we use these in our formula, the overall combustion enthalpy for methane will be a large, negative number. This tells us that burning methane releases a lot of energy!
For methane, the standard enthalpy of formation (ΔHf°) is approximately -74.8 kJ/mol. The enthalpy of formation for carbon dioxide (CO2) is about -393.5 kJ/mol. And for liquid water (H2O), it’s around -285.8 kJ/mol. (If it's gaseous water, the value is a bit less negative).
Now, let's do the math: ΔHcombustion = [ (1 * ΔHf° CO2) + (2 * ΔHf° H2O) ] - [ (1 * ΔHf° CH4) + (2 * ΔHf° O2) ]
ΔHcombustion = [ (1 * -393.5 kJ/mol) + (2 * -285.8 kJ/mol) ] - [ (1 * -74.8 kJ/mol) + (2 * 0 kJ/mol) ]
ΔHcombustion = [ -393.5 kJ/mol - 571.6 kJ/mol ] - [ -74.8 kJ/mol ]
ΔHcombustion = [ -965.1 kJ/mol ] - [ -74.8 kJ/mol ]
ΔHcombustion = -965.1 kJ/mol + 74.8 kJ/mol

ΔHcombustion = -890.3 kJ/mol
See that negative sign? It’s a cheering squad for energy release! This number, approximately -890.3 kilojoules per mole, tells us that when one mole of methane burns completely, it releases about 890.3 kilojoules of energy. That’s a significant amount of heat!
Why is this so entertaining? Because you’re not just blindly accepting that fire is hot. You’re understanding why it’s hot, and you’re armed with the tools to predict it. It's like learning the recipe for a chemical reaction's energy output. You’re demystifying a fundamental force of nature.
It’s special because it connects the microscopic world of atoms and molecules to the macroscopic world of flames and heat. You’re seeing how the arrangement of atoms in methane dictates how much energy is unleashed when it transforms. It’s a testament to the order and predictability that lies within the seemingly chaotic dance of chemistry.
And the best part? This skill isn't just for the lab coat brigade. Understanding energy changes is crucial for everything from designing more efficient engines to comprehending climate change. It's a window into how our planet works and how we interact with it. It’s a way to feel a little bit more in control, a little bit more knowledgeable about the world around you.
So, next time you see a flame, whether it’s a candle or a gas stove, remember the hidden calculation happening behind the scenes. Think about the enthalpy change, the energy bargain struck between molecules. It’s a little bit of everyday magic, and now you know how to peek behind the curtain. Give it a try yourself! It's a surprisingly rewarding mental workout, and who knows what other chemical secrets you might uncover.
