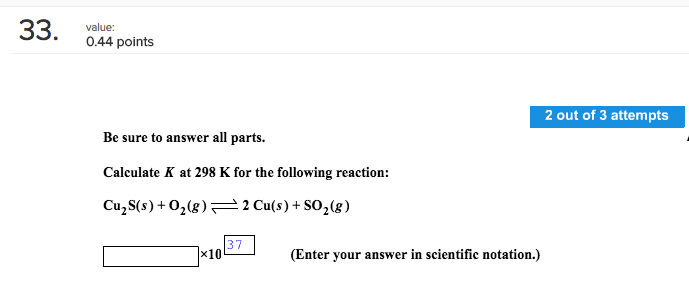
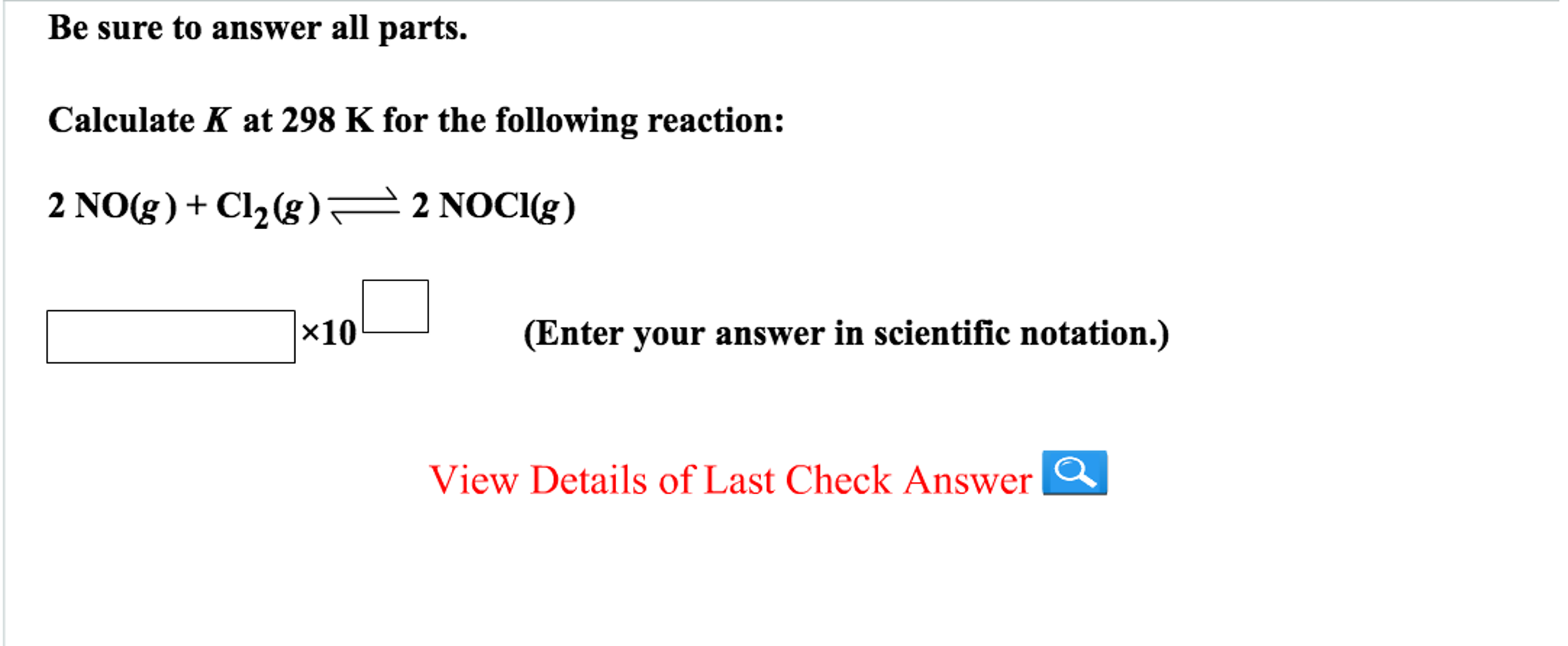
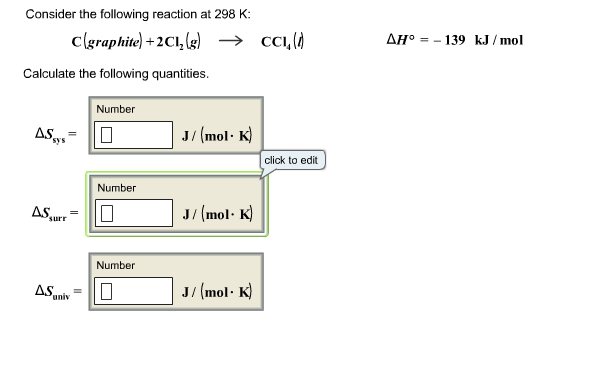
Calculate K At 298 K For The Following Reaction

Hey there! So, you’re staring down a chemistry problem, huh? Don’t worry, we’ve all been there. Today, we’re going to tackle this beast: calculating K at a nice, cozy 298 Kelvin. Think of it like making our favorite coffee blend. We need the right ingredients, right? And a little bit of magic (okay, science!) to get the perfect flavor. This isn't some stuffy lecture, nope. We're just chatting, like we’ve got our mugs and a comfy couch. So, settle in, maybe grab yourself another cuppa, and let's dive in!
First off, what even IS this "K" we're talking about? It’s the equilibrium constant. Super important. Basically, it tells us whether our reaction is feeling more like a party where all the reactants are mingling, or if it's mostly just products chilling on their own. Is it a "go, go, go" reaction, or a "hold up, let's chill" situation? K is our trusty little indicator.
And 298 Kelvin? That's just a fancy way of saying room temperature. It’s like saying "let's make this at a comfortable 25 degrees Celsius." Chemists love their Kelvin scale, though. It’s got some nice mathematical properties, apparently. But for us, just think: nice and chill.
Now, to figure out K, we need a few things. It’s not just pulled out of thin air, you know. We need some juicy data. Usually, this involves something called Gibbs Free Energy. Ever heard of it? It's like the universe’s way of saying, "Is this reaction actually going to happen, or should we just save our energy?" Pretty neat, right?
The magical formula that connects Gibbs Free Energy (we often call it ΔG, pronounced "delta G," for short) and our beloved K is this: ΔG° = -RT ln K. Whoa, looks a bit intimidating, doesn’t it? Like a secret code. But stick with me, it's not that scary.
Let’s break it down. ΔG° (that little circle means "standard conditions," which is usually 298 K and 1 atm pressure, by the way) is our change in Gibbs Free Energy. This is the energy cost or gain for the reaction to happen. It's like the "effort" involved.
Then we have R. That's the ideal gas constant. Think of it as a universal conversion factor. It just helps us keep our units all tidy and happy. We usually use R = 8.314 J/(mol·K). Don't sweat the units too much for now, just know it's a constant we need.
T, of course, is our temperature in Kelvin. We already know that's 298 K for our cozy scenario. Easy peasy!
And then, the star of our show: ln K. This is the natural logarithm of our equilibrium constant. Logarithms are just a way to deal with really big or really small numbers. Think of them as a magnifying glass for numbers, but in reverse. We'll get to K itself later.
So, our mission, should we choose to accept it, is to get K. We need to rearrange that formula. Algebra, anyone? Don't groan! We can do this together. If ΔG° = -RT ln K, then to get ln K by itself, we divide both sides by -RT. So, ln K = -ΔG° / (RT).
Once we have ln K, we need to "undo" the natural logarithm to find K. How do we do that? We use the exponential function, ex. So, K = e(-ΔG° / RT). Ta-da! That’s our ultimate goal. We just need the ΔG° for our specific reaction. It’s like having the recipe, but we need the specific ingredients for our dish.
Where do we get this ΔG° value? Ah, that’s the million-dollar question! Sometimes, it’s given to you directly in the problem. Lucky you! Other times, you might need to look it up in a handy dandy table of standard Gibbs Free Energies of Formation. These tables are like cheat sheets for chemists. They tell you how much energy it takes to form a substance from its elements in their standard states.
If you have to calculate ΔG° yourself (don't panic!), you can use another super useful formula: ΔG° = ΔH° - TΔS°. See, T and ΔS° are back! ΔH° is the standard enthalpy change (think of it as the heat absorbed or released in the reaction, like the "cooking heat") and ΔS° is the standard entropy change (how much "disorder" or "randomness" changes).
Again, you’ll likely find ΔH° and ΔS° values in those helpful tables. You sum up the ΔH° of formation for the products, multiply by their stoichiometric coefficients (those little numbers in front of the chemical formulas), and then subtract the sum of the ΔH° of formation for the reactants. Same goes for ΔS°!
So, let’s imagine our reaction. We’ll just make one up for fun, shall we? Let’s say we’re interested in this little guy: A + B <=> C + D. Just a simple, hypothetical reaction. Now, if we were given the standard Gibbs Free Energy of reaction directly, let’s say ΔG° = -10 kJ/mol. (Note the negative sign! This is a good sign, meaning the reaction is spontaneous. Yay!) And remember, we need to convert kJ to Joules, so that's -10,000 J/mol.
Our temperature T is 298 K. Our R is 8.314 J/(mol·K).
Now, let's plug and chug into our formula: ln K = -ΔG° / (RT).
ln K = -(-10,000 J/mol) / (8.314 J/(mol·K) * 298 K)
ln K = 10,000 / (2477.572)
ln K ≈ 4.036
So, ln K is about 4.036. Remember our next step? To get K, we need to do K = e(ln K).
K = e4.036
K ≈ 56.58
And there you have it! Our equilibrium constant K is approximately 56.58. What does that even mean? Well, a K value greater than 1 (like 56.58!) means that at equilibrium, there are more products than reactants. The reaction likes to hang out in the product zone. It’s basically saying the reaction favors the products. It’s like our coffee brew is mostly rich, dark espresso with just a splash of milk. Delicious!
What if our ΔG° was positive? Let's say ΔG° = +10 kJ/mol, or +10,000 J/mol. Then:
ln K = -(10,000 J/mol) / (8.314 J/(mol·K) * 298 K)
ln K ≈ -4.036
K = e-4.036
K ≈ 0.0177
See? A K value much less than 1 means at equilibrium, there are more reactants than products. The reaction prefers to stay in the reactant phase. It's like a really weak tea, where you can barely taste the tea leaves. Not quite what we were hoping for, eh?
And if ΔG° was zero? Well, then ln K would be zero, and K would be e0, which is 1. This is a special case, where at equilibrium, you have roughly equal amounts of reactants and products. It’s like a perfectly balanced latte, neither too strong nor too weak.
So, the whole point of calculating K is to understand where the reaction "settles down" when it’s done reacting. It’s not about how fast it gets there (that’s kinetics!), but about the final ratio of products to reactants.

Let’s do another example, just to really drill this home. Imagine we have the reaction: 2H₂(g) + O₂(g) <=> 2H₂O(g). This is, of course, the formation of water. Super important, right?
Let’s say we're given the standard Gibbs Free Energy of formation for water vapor (H₂O(g)) is -228.6 kJ/mol. And for H₂ and O₂, it’s 0 kJ/mol (since they are elements in their standard state). So, for our reaction:
ΔG°reaction = (2 * ΔG°f(H₂O(g))) - (2 * ΔG°f(H₂(g)) + 1 * ΔG°f(O₂(g)))
ΔG°reaction = (2 * -228.6 kJ/mol) - (2 * 0 kJ/mol + 1 * 0 kJ/mol)
ΔG°reaction = -457.2 kJ/mol
Woohoo! We got our ΔG°! Now, let's convert it to Joules: -457,200 J/mol.
T = 298 K
R = 8.314 J/(mol·K)
Time for the formula: K = e(-ΔG° / RT).
K = e-(-457,200 J/mol) / (8.314 J/(mol·K) * 298 K)
K = e(457,200) / (2477.572)
K = e184.54
Now, e184.54. That's a HUGE number. Like, astronomically huge. We're talking 10 to the power of... well, a lot! This means that for the formation of water, the equilibrium constant is incredibly, ridiculously, unbelievably large. This reaction absolutely, positively, 100% favors the formation of products. It's like when you mix hydrogen and oxygen, and BOOM, you get water. There's virtually no going back! It's the ultimate "go, go, go" reaction.
So, when you see those calculations, don't get flustered. Just remember the steps:
1. Find your ΔG° for the reaction. Either it’s given, or you calculate it from ΔH° and ΔS°.
2. Make sure your units are consistent! kJ to J, and remember T is always in Kelvin.
3. Plug everything into K = e(-ΔG° / RT). Your calculator will thank you for its powerful exponentiation skills.
4. Interpret your K value! Big K means products, small K means reactants, K near 1 means a mix.
It’s really that straightforward. It’s just a bit of math, a bit of unit conversion, and understanding what those symbols actually mean. Think of it as learning a new language, the language of chemical reactions. And K is one of the most important phrases in that language.
So next time you’re faced with calculating K at 298 K, take a deep breath, maybe make another cup of coffee, and remember: you’ve got this! You’re not just solving a problem, you’re understanding a fundamental aspect of how the chemical world works. Pretty cool, right? Happy calculating!
