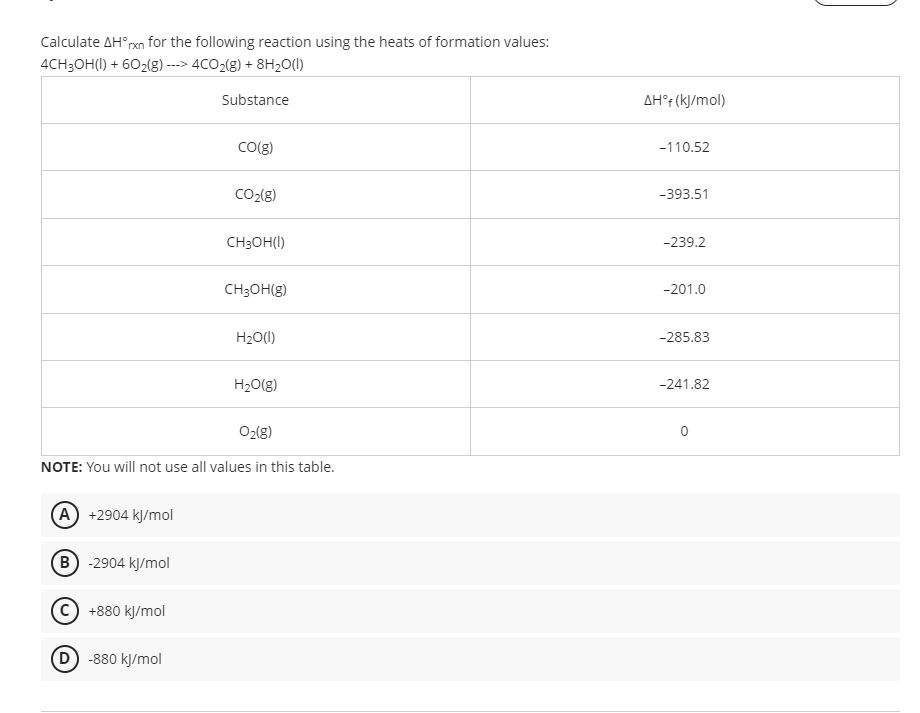
Calculate Delta H Rxn For The Following Reaction

Hey there, science explorers! Ever found yourself staring at a chemical reaction and wondering, "What's the deal with the energy here?" You know, like, is it giving off heat, or does it actually suck heat out of the room? Well, buckle up, because today we're diving into the wonderfully chill world of calculating something called Delta H Rxn. Sounds fancy, right? But trust me, it's just a cool way to figure out the energy change in a chemical reaction.
Think of it like this: every time you see chemicals doing their thing, rearranging themselves into new stuff, there's an energy exchange happening. It's like a little cosmic handshake. Sometimes the handshake gives off a warm hug (exothermic), and sometimes it feels a bit like a chilly breeze (endothermic). Delta H Rxn is our way of putting a number on that hug or that breeze. Pretty neat, huh?
So, What Exactly IS Delta H Rxn?
Let's break it down. Delta, that little triangle symbol (Δ), in science basically means "change." And H? That stands for enthalpy, which is basically a fancy word for the total heat content of a system. So, Delta H Rxn is the change in heat content during a chemical reaction. Simple as that!
When we calculate Delta H Rxn, we're essentially asking: "How much heat did this reaction either release or absorb?" If Delta H Rxn is negative, it means the reaction gave off heat – it’s exothermic. Think of it like lighting a match; you feel the warmth, right? That's an exothermic process.
If Delta H Rxn is positive, it means the reaction absorbed heat from its surroundings – it’s endothermic. Imagine dissolving some instant ice packs; that sudden chill you feel? That’s an endothermic reaction at work.
Why Should We Even Care About This Energy Stuff?
Honestly, it's kind of a big deal! Understanding these energy changes helps us predict how reactions will behave. For example, engineers use this information all the time. They need to know if a reaction is going to produce a ton of heat (which could be dangerous if not managed!) or if it requires a lot of energy to get going (meaning you'll need to supply it). It's like knowing if you're planning a bonfire or a chilly camping trip – you prepare differently for each!
Plus, it’s just plain fascinating! It's like unlocking a secret code of the universe. Every reaction has its own energetic fingerprint, and Delta H Rxn helps us read it. It’s the difference between just seeing a car move and understanding the engine that's making it go.
Okay, Enough Chat, How Do We Actually Calculate It?
Alright, alright, I know you're eager to get to the good stuff. There are a few ways to tackle this, but one of the most common and accessible methods involves using something called standard enthalpies of formation. Don't let the big words scare you!

Think of standard enthalpies of formation (we often write this as ΔH°f) as the "building blocks" of energy for different substances. It's the energy it takes to create one mole of a compound from its basic elements in their most stable form, under standard conditions (which are usually 25°C and 1 atmosphere of pressure). It's like knowing the energy cost to assemble a Lego model from individual bricks.
We usually have tables of these values, kind of like a cheat sheet, ready for us. Chemists have spent ages figuring these out, so we don't have to reinvent the wheel every time.
The Magic Formula: Using Enthalpies of Formation
The actual calculation is surprisingly straightforward once you have your values. The formula looks a little something like this:
ΔH°rxn = ΣnΔH°f (products) - ΣmΔH°f (reactants)
Whoa, what does that mean? Let's break it down piece by piece:
- Σ (Sigma): This is the Greek letter sigma, and it just means "sum of." We're going to be adding things up!
- n and m: These are the stoichiometric coefficients from your balanced chemical equation. Think of them as the numbers in front of the chemical formulas that make sure everything balances out. They tell you how many "units" of each molecule are involved. It’s like counting how many of each Lego brick you need for your model.
- ΔH°f: We already talked about this – the standard enthalpy of formation for each substance.
- Products: These are the new things that are made during the reaction.
- Reactants: These are the starting materials that get mixed together.
So, in plain English, the formula means: "Take the sum of the enthalpies of formation for all your products, and then subtract the sum of the enthalpies of formation for all your reactants."

It’s like balancing your budget! You look at all the money coming in (products) and subtract all the money going out (reactants) to see if you’re in the black or in the red (energetically speaking).
Let's Walk Through an Example (Without Getting Too Gritty)
Imagine we have a simple reaction like the combustion of methane (natural gas):
CH₄(g) + 2O₂(g) → CO₂(g) + 2H₂O(g)
To find the ΔH°rxn for this, we'd need to look up the ΔH°f values for CH₄, O₂, CO₂, and H₂O. Let's pretend we found these (these are approximate values for illustration!):
- ΔH°f (CH₄) = -74.8 kJ/mol
- ΔH°f (O₂) = 0 kJ/mol (The enthalpy of formation for an element in its standard state is always zero. Oxygen is already in its most stable form!)
- ΔH°f (CO₂) = -393.5 kJ/mol
- ΔH°f (H₂O) = -241.8 kJ/mol
Now, let's plug these into our formula:

ΔH°rxn = [1 * ΔH°f(CO₂) + 2 * ΔH°f(H₂O)] - [1 * ΔH°f(CH₄) + 2 * ΔH°f(O₂)]
See how we used the coefficients (1 for CO₂, 2 for H₂O, etc.)? Now, let's plug in the numbers:
ΔH°rxn = [1 * (-393.5 kJ/mol) + 2 * (-241.8 kJ/mol)] - [1 * (-74.8 kJ/mol) + 2 * (0 kJ/mol)]
Let's do the math:
ΔH°rxn = [-393.5 kJ/mol - 483.6 kJ/mol] - [-74.8 kJ/mol + 0 kJ/mol]
ΔH°rxn = [-877.1 kJ/mol] - [-74.8 kJ/mol]
ΔH°rxn = -877.1 kJ/mol + 74.8 kJ/mol
ΔH°rxn = -802.3 kJ/mol
Ta-da! We calculated that this reaction releases a whopping 802.3 kilojoules of energy per mole of methane burned. That's why burning natural gas feels warm – it's giving off a lot of heat!
It's Not Just About Burning Things!
This method is super versatile. It works for all sorts of reactions, from the formation of water to more complex biological processes. It's the foundation for understanding so much about how the world around us works, chemically speaking.
So, next time you see a chemical equation, don't just see a jumble of letters and numbers. See a story of energy exchange, a dance of molecules. And with the power of Delta H Rxn, you can even put a number on how much energy is involved in that dance. Pretty cool, right?
Keep exploring, keep asking questions, and remember, even the most complex science can be understood with a little curiosity and a chill attitude!
