Calcium Hydride Reacts With Water To Form Hydrogen Gas

Ever felt like you’re just a sprinkle of stardust away from a little bit of chemical magic? Well, buckle up, because we’re about to dive into a reaction that’s as visually exciting as it is surprisingly useful: when Calcium Hydride meets Water, things get bubbly, and not just with your average soda pop! This isn’t just some dusty old science experiment; it’s a fascinating chemical dance that produces Hydrogen Gas, and understanding it opens up a world of cool possibilities.
Think of it like this: you’ve got this unassuming white powder, Calcium Hydride, which is essentially calcium with a hydrogen atom clinging to it. Then, you introduce the universal solvent, Water. What happens next is a chemical high-five! The calcium and hydrogen decide to part ways with their original partners. The calcium teams up with the oxygen and the other hydrogen from the water to form Calcium Hydroxide (which, by the way, is also known as slaked lime – pretty common stuff!). But the real star of the show, the byproduct that makes this reaction so special, is the pure, unadulterated Hydrogen Gas that bursts free.
Why should you care about this bubbly concoction? Well, the generation of Hydrogen Gas is where the real fun and utility kick in. Hydrogen is an incredibly versatile element. It’s the lightest element there is, and when you produce it cleanly, like in this reaction, it’s a fantastic energy carrier. Imagine a future powered by clean energy; Hydrogen is a major contender, and reactions like the one between Calcium Hydride and Water offer a straightforward way to generate it.
One of the most immediate benefits is its role in creating a portable and on-demand source of Hydrogen. For applications where you need a steady stream of this gas, but can’t easily transport bulky tanks, this reaction is a game-changer. For instance, in remote locations or for specific scientific or industrial processes, being able to generate Hydrogen right where you need it, simply by adding water to Calcium Hydride, is incredibly convenient.

Let’s talk about some of the cool applications. Back in the day, and even in some specialized areas today, Hydrogen produced this way was vital for inflating weather balloons. You need a reliable way to fill those balloons, and this reaction provided just that. It’s a simple, effective method to get a large volume of Hydrogen quickly. Beyond that, the production of Hydrogen Gas has implications for various chemical syntheses. Many industrial processes rely on pure Hydrogen as a reactant, and understanding how to generate it safely and efficiently is paramount.
Safety is, of course, a huge consideration when dealing with any chemical reaction, especially one that produces a flammable gas. While the reaction between Calcium Hydride and Water is relatively straightforward and controllable, it’s important to remember that Hydrogen Gas is highly flammable. This means handling it requires proper precautions, good ventilation, and an awareness of potential ignition sources. However, the very fact that it’s controllable and generates the gas on demand makes it a safer alternative in many scenarios compared to storing large quantities of pre-made Hydrogen.
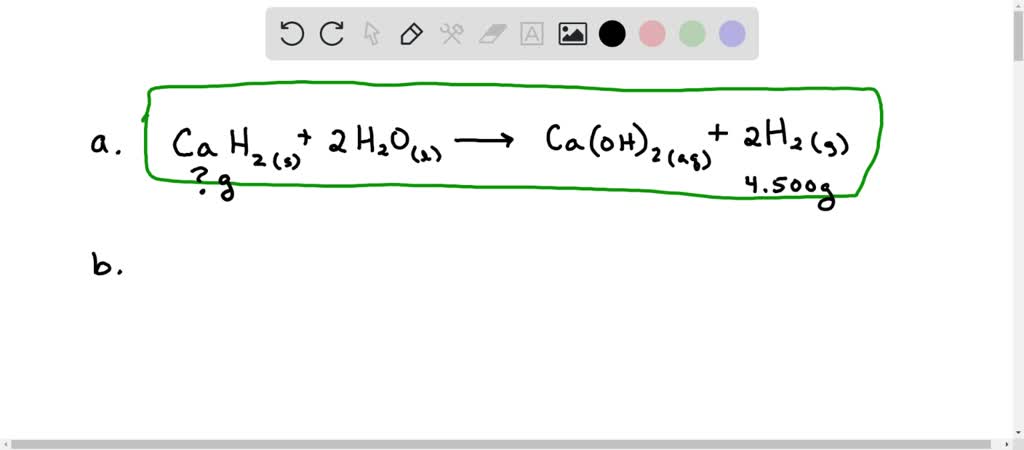
The beauty of this reaction lies in its elegance. It's a simple equation: CaH2 + 2H2O → Ca(OH)2 + 2H2. Calcium Hydride plus Water yields Calcium Hydroxide and Hydrogen Gas. It’s a chemical transformation that’s easy to grasp conceptually, yet it unlocks significant practical benefits. It highlights how seemingly simple interactions between elements can lead to powerful outcomes.
Think about the potential for future innovations. As we continue to explore cleaner energy solutions, the efficient generation of Hydrogen will become even more critical. This reaction, along with others, forms the bedrock of our understanding and ability to harness the power of Hydrogen. It’s a testament to the enduring power of chemistry to solve problems and create new opportunities. So, the next time you hear about Calcium Hydride and Water, remember the exciting bubbly reaction and the valuable Hydrogen Gas it produces – a little chemistry that makes a big difference!
