Ca Subshell From Which Electron Removed To Form Cation

Ever wondered what makes an atom decide to become a positive ion, or a cation? It’s like a tiny chemical drama playing out, and the star of the show is often an electron making a daring escape! Today, we’re going to peek behind the scenes and explore which electron, from which special part of the atom, is usually the first to go when an atom wants to charge up. Think of it as a backstage pass to the electrifying world of ions!
This might sound like pure science fiction, but it’s actually a fundamental concept in chemistry that helps us understand everything from why metals conduct electricity to how our bodies use vital minerals. Knowing which electron is the "sacrificial lamb" is super useful for predicting chemical reactions, designing new materials, and even understanding the intricate processes happening within living cells. It’s the key to unlocking why some elements are more reactive than others and how they bond together to form the world around us.
"The outermost electrons are the 'party animals' of the atom – always ready to mingle and, in this case, to leave!"
So, where do these adventurous electrons live within an atom? Imagine an atom as a miniature solar system. At its center is the nucleus, a dense core containing protons (positively charged) and neutrons (no charge). Whizzing around this nucleus are electrons, like tiny planets in orbits. But these orbits aren’t all the same distance from the sun (the nucleus). They exist in different energy levels, or shells.
The shells closest to the nucleus are the innermost shells. These electrons are held tightly by the positive pull of the protons. Think of them as being comfortably tucked in, enjoying the strong parental embrace. They require a lot of energy to be coaxed away. The further out you go, the weaker the pull from the nucleus becomes. These are the outermost shells, also known as the valence shells.
Now, here’s where the fun really begins! When an atom wants to become a cation – meaning it wants to lose a negatively charged electron to become positively charged – it doesn’t usually give up just any electron. It’s far more efficient and energetically favorable to lose one from the valence shell. Why? Because these valence electrons are the furthest away from the nucleus and experience the weakest attraction. It’s like trying to grab a toy from a child who’s right next to you versus one who’s way across the playground; the latter is much easier!

The specific subshell from which an electron is removed to form a cation is almost always the outermost subshell within the valence shell. In the grand scheme of electron arrangement, we talk about orbitals (regions where electrons are likely to be found) grouped into subshells (like s, p, d, and f). Each subshell has a different shape and capacity for holding electrons. The s subshell is the simplest, with a spherical shape. The p subshell is dumbbell-shaped and has three orbitals. Then comes the more complex d subshell (five orbitals) and the highly intricate f subshell (seven orbitals).
For most elements, especially the main group elements (those in the s-block and p-block of the periodic table), the electron that's easiest to remove resides in the outermost p subshell. If the outermost shell doesn't have any p electrons, then it would be the outermost s subshell. Think of it as a hierarchy of "easy escapes." The electrons in the p orbitals of the valence shell are typically the first to go because they are the furthest out and least attracted to the nucleus.
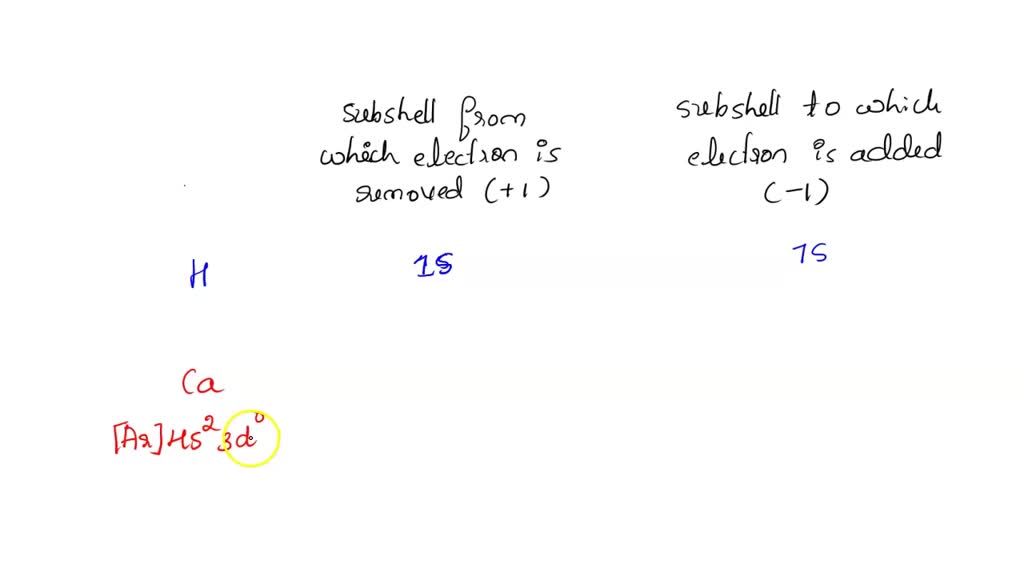
However, things get a bit more interesting when we look at transition metals (the elements in the d-block). These metals have electrons in both their outermost s subshell and their underlying d subshell. When transition metals form cations, it’s often the electrons from the outermost s subshell that are removed first, even though the d subshell might be further from the nucleus in terms of energy level. This is because the s electrons are generally more exposed and thus more readily lost. After the s electrons are gone, then electrons from the d subshell can be removed, leading to the formation of ions with different charges for transition metals, which is why they are so versatile in their chemistry.
So, the next time you see a chemical reaction where an atom transforms into a positively charged ion, you can picture those electron "party animals" in the outermost shells, particularly those in the s or p subshells (or the d subshell for transition metals, though often after the s electrons have gone), making their grand exit. It’s this willingness of valence electrons to be removed that makes elements react and bond, creating everything from the salt on your table to the complex molecules that keep us alive. Pretty neat, right? It’s a simple rule, but it has profound implications for the entire universe of chemistry!
