Brainpop Periodic Table Of Elements Worksheet Answers

Hey there, fellow science adventurers! So, you’ve been diving into the wild and wonderful world of the Periodic Table of Elements, huh? Maybe you’ve got a BrainPOP video playing in the background, and now you’re staring at a worksheet, wondering, "What in the elemental heck am I supposed to do with this?" Don't sweat it! It's like trying to build a LEGO castle without the instruction booklet sometimes, right? But guess what? We're about to unlock the secrets to those BrainPOP Periodic Table worksheet answers, and trust me, it's gonna be a breeze (or maybe a noble gas, if you’re feeling fancy!).
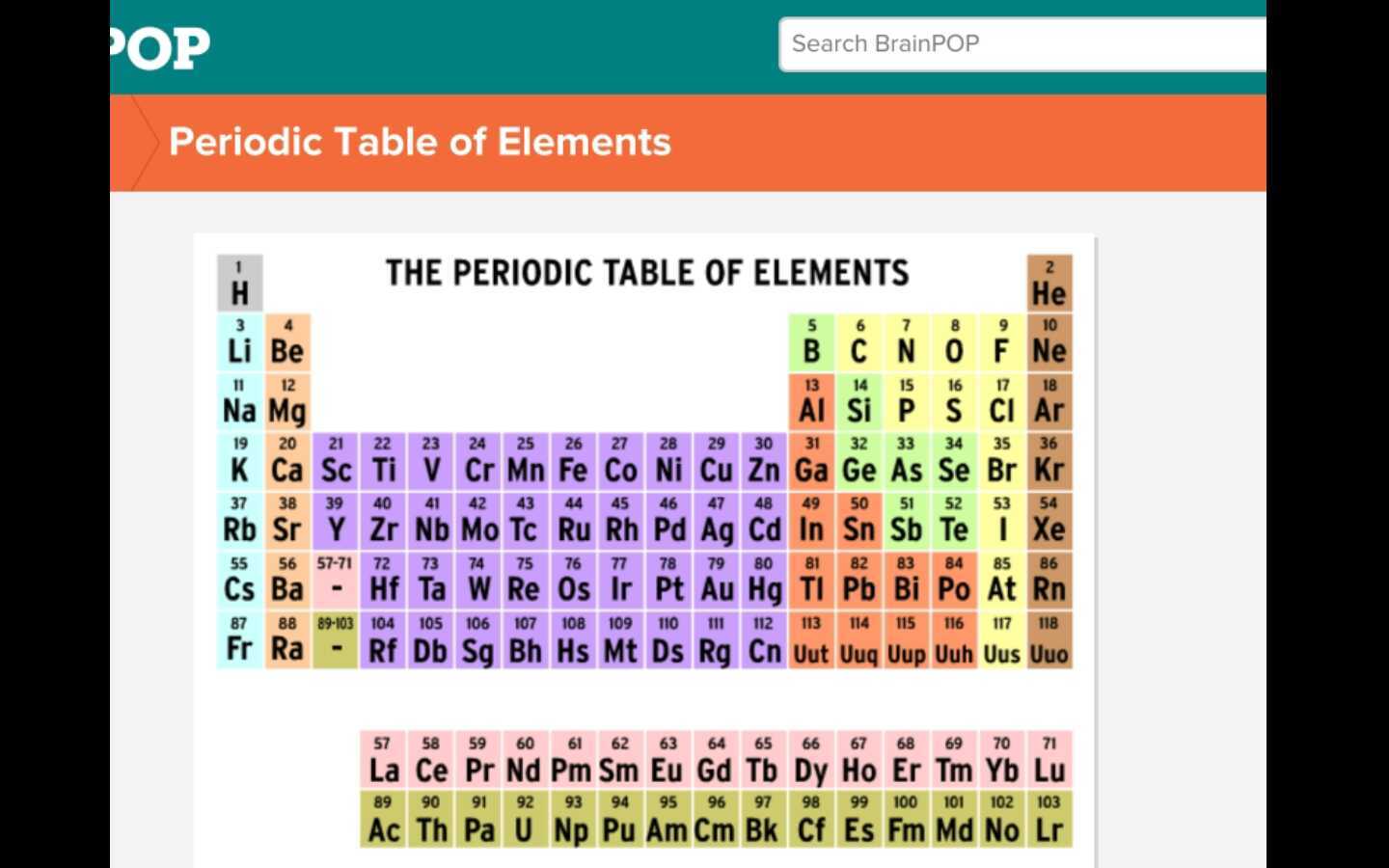
Think of the Periodic Table as the ultimate elemental party. Every element is invited, and they're all lined up in a super organized way. It's not just a bunch of random letters and numbers thrown together; oh no, this table has meaning, like a secret code waiting to be cracked. And those BrainPOP worksheets? They're your little keys to understanding that code!
Let's be real for a sec. Sometimes, science can feel a bit like trying to herd cats. You've got protons doing their thing, electrons zipping around like hyperactive hummingbirds, and neutrons just chilling in the nucleus, being all stoic. But the Periodic Table helps us make sense of it all. It’s like a family tree for atoms, showing us who’s related to whom and why they behave the way they do.
So, you’ve probably got questions about atomic numbers, mass numbers, symbols, and maybe even electron configurations. Don't let those terms send you running for the hills! BrainPOP usually breaks it down so nicely, like a perfectly peeled banana. And the worksheet? It’s just there to reinforce what you’ve learned. It's like practicing your multiplication tables; the more you do it, the easier it gets!
Let's tackle some common worksheet scenarios. You might see a question asking for the atomic number of an element. What’s that, you ask? Easy peasy! The atomic number is simply the number of protons in an atom's nucleus. It's like an element's ID number. Every element has a unique atomic number. So, if you're looking at Hydrogen (H), its atomic number is 1. That means it has 1 proton. Simple, right? No need to overcomplicate it!
Then there's the mass number. This one's a little different. The mass number is the total number of protons and neutrons in an atom's nucleus. Think of it as the element's weight, in a super simplified way. So, if an element has 6 protons and 6 neutrons, its mass number is 12. See? It’s just adding two numbers together. You got this!

And the element symbol? This is usually the easiest part. It's just a one or two-letter abbreviation for the element's name. Like ‘O’ for Oxygen, or ‘Fe’ for Iron (that one’s a bit of a curveball because it comes from the Latin word 'ferrum,' but hey, we’re learning!). BrainPOP usually shows you these right on the table itself. It's like the element's nickname!
Now, let's talk about groups and periods. These are the rows and columns of the Periodic Table. The periods are the horizontal rows. They tell you something about the element's electron shells. The groups are the vertical columns. Elements in the same group often have similar chemical properties. It's like they're all part of the same elemental club, sharing common traits!
BrainPOP might ask you to identify which group or period an element belongs to. All you need to do is find the element on the table and count its row (for periods) or column (for groups). It's like playing a game of "I Spy" on the Periodic Table. "I spy with my little eye... an element in Group 1!"
Sometimes, worksheets will get a bit more advanced and ask about electron configurations. Woah, don't panic! This just describes where the electrons are located around the nucleus. Think of it like assigning seats in a theater for the electrons. They fill up different "shells" or energy levels. BrainPOP usually explains this by showing you how many electrons are in each shell. It's like saying, "Okay, the first row has 2 electrons, the second row has 8, and so on." This is super important because it dictates how elements interact with each other – which is basically the whole point of chemistry!

A common task is identifying an element based on its atomic number. If the worksheet says, "What element has an atomic number of 7?" you just look on the Periodic Table for the element with 7 protons. Boom! It's Nitrogen (N). It’s like knowing your friend’s phone number and being able to call them. The atomic number is the key!
Another type of question might be: "An element has 11 protons and 12 neutrons. What is its atomic number and mass number, and what is the element's symbol?" For the atomic number, you know it's the number of protons, so that's 11. For the mass number, you add protons and neutrons: 11 + 12 = 23. Now, find the element with atomic number 11 on the Periodic Table. Ta-da! It's Sodium (Na). See? You're basically a detective now, sniffing out elemental clues!
You might also be asked to classify elements. Is it a metal, a nonmetal, or a metalloid? The Periodic Table is helpfully color-coded (usually!). Metals are generally shiny, good conductors of heat and electricity, and tend to lose electrons. Nonmetals are the opposite – dull, poor conductors, and tend to gain electrons. Metalloids are the in-betweeners, having properties of both. BrainPOP will definitely walk you through these categories, making it super clear which sections of the table are which.

So, when you see a question asking, "Which of the following elements is a noble gas?" and you see options like Helium (He), Neon (Ne), and Argon (Ar), you'd look for Group 18 on the Periodic Table. These guys are famously unreactive, like they're too cool to hang out with anyone. They've got a full outer electron shell, so they're perfectly content being left alone. Imagine a party where everyone's already got a plus-one and doesn't need to mingle. That's the noble gases!
What about halogens? They're in Group 17, and they're very reactive. They’re like the social butterflies of the Periodic Table, always looking to snag an electron from someone else to complete their own outer shell. Think Fluorine (F) or Chlorine (Cl). They’re eager to make friends, even if it means stealing something!
Sometimes, the questions might be a bit more conceptual. Like, "Why do elements in the same group have similar chemical properties?" The answer, my friends, is all about those pesky valence electrons – the electrons in the outermost shell. Elements in the same group have the same number of valence electrons, which is the main driver behind their chemical behavior. It's like siblings from the same family – they might have different personalities, but they often share some core traits passed down from their parents.
Don't get discouraged if you don't get every single answer right away. That’s what the learning process is all about! The BrainPOP videos are your teachers, and the worksheets are your practice problems. If you’re stuck on a question, go back to the video! Rewind it, rewatch the section that explains that concept. It’s like re-reading a chapter in a book when you’re unsure about a plot point.
And if you’re really stuck, don’t be afraid to ask for help! Your teacher, a classmate, or even a friendly online science forum can be a lifesaver. We’re all in this elemental adventure together!
Let’s think about the sheer brilliance of the Periodic Table. It’s a testament to human curiosity and the desire to organize the universe around us. It's a map that guides us through the building blocks of everything we see, touch, and even are. From the air we breathe (Oxygen!) to the phones in our hands (Lithium, Silicon!) and the stars in the sky (Hydrogen, Helium!), the Periodic Table holds the key to understanding it all.
So, as you tackle those BrainPOP Periodic Table worksheets, remember you’re not just filling in blanks. You’re decoding the language of the universe. You’re learning about the fundamental ingredients that make up everything. Every element has a story, a personality, and a role to play in the grand cosmic drama. And by understanding the Periodic Table, you’re becoming a part of that story, a master of the elemental alphabet!
Don’t let a few tricky questions dim your shine. Embrace the challenge, celebrate the little victories when you get an answer right, and keep that curious spirit alive. You’re doing awesome, and with a little practice and the awesome resources like BrainPOP, you'll be a Periodic Table pro in no time! Keep exploring, keep questioning, and keep discovering the incredible world of elements. You’ve got this, and the universe of atoms is waiting for you!
