Boron Electron Configuration Noble Gas Notation

Alright, gather ‘round, you magnificent collection of curious minds! Let’s talk about something that sounds super serious, like it’s going to involve white lab coats and ominous bubbling beakers, but I promise, it’s way more fun than that. We’re diving into the wild, weird world of Boron, and more specifically, its electron configuration and this fancy thing called noble gas notation. Think of it as Boron’s cosmic address, its celestial ZIP code, the secret handshake of its electrons.
So, Boron. What is this guy? Is it a type of fancy cheese? A new superhero? Well, it’s actually an element, number 5 on the periodic table. It’s that little guy chilling between Helium and Carbon. And, believe it or not, Boron is actually pretty important. It’s in things like detergents (making your whites whiter than a polar bear’s dental floss), and it’s even used in some really cool high-tech ceramics. So, next time you’re battling a stubborn stain, give a little nod to Boron. You owe it one.
Now, before we get to the electron configuration, we need to understand what electrons are. Imagine tiny, hyperactive popcorn kernels zipping around the nucleus of an atom. They’re not just randomly bouncing around, oh no. They have rules! They live in different "shells" or energy levels, kind of like apartments in a ridiculously tall skyscraper. The closer to the nucleus (the penthouse suite), the lower the energy. The further away, the more energetic they get, probably fueled by too much atomic espresso.
Boron, being element number 5, has 5 protons in its nucleus (that’s what defines it as Boron, its atomic number) and, in a neutral atom, it also has 5 electrons. These little guys need to find their spots in the atomic apartment building. This is where the electron configuration comes in. It’s basically a list of where all 5 of Boron’s electrons are hanging out.
We start filling up the lowest energy levels first, because, duh, who wouldn’t want the best real estate? The very first shell, the one closest to the nucleus, can only hold 2 electrons. Think of it as a studio apartment – cozy, but limited space. So, Boron’s first two electrons zoom into the 1s orbital. We denote this as 1s². The ‘1’ is the shell number, ‘s’ is the type of orbital (they’re like different room shapes), and the ‘²’ means there are two electrons there. Simple enough, right?
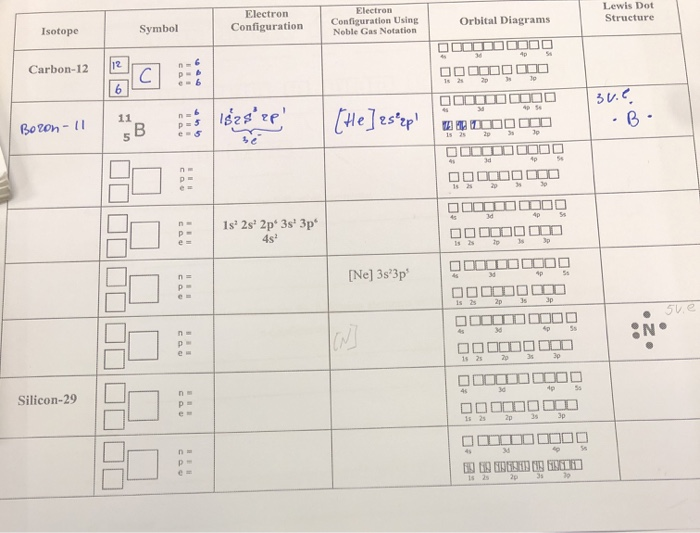
Now we have 3 electrons left. The first shell is full, like that tiny studio is now occupied. So, our remaining electrons have to move up to the next floor, the second energy level. This second level is a bit more spacious. It has a few different types of rooms, or orbitals. There’s the ‘s’ orbital again, which can hold 2 electrons, and then there are the ‘p’ orbitals, which are like suites with multiple rooms, capable of holding up to 6 electrons in total (3 ‘p’ orbitals, each holding 2).
So, Boron’s next two electrons go into the 2s orbital. That’s 2s². We’ve now accounted for 2 + 2 = 4 electrons. We’ve got just one electron left. Where does this little guy go? He’s the last one at the party, the odd electron out!

He has to move to the next available spot, which is in the 2p orbitals. Since there are plenty of ‘p’ orbitals available, this lone electron takes up residence in one of them. So, we write this as 2p¹. And there you have it, the full electron configuration for Boron: 1s² 2s² 2p¹. It’s like Boron’s apartment floor plan: two in the first floor studio, two in the second floor ‘s’ suite, and one in a ‘p’ suite on the second floor. It's basically saying, "I'm here, and I'm kinda just chilling in this one ‘p’ room."
But wait, there’s more! Sometimes, writing out the entire electron configuration for every element can be a bit like writing a novel for every person you meet. Imagine if you had to list every single thing about your neighbor, down to their favorite brand of socks, just to say "Hi, I live next door." Exhausting, right? This is where our hero, noble gas notation, swoops in like a superhero with a shortcut.
Noble gases are those super chill, unreactive elements at the very end of the periodic table: Helium, Neon, Argon, Krypton, Xenon, Radon. They’re called noble because they’re like the divas of the element world – they’ve got a full outer shell, they’re incredibly stable, and they couldn’t be bothered to react with anyone. They’re the atoms that just don’t need anyone else. They’ve achieved electron nirvana.
Noble gas notation uses the electron configuration of the preceding noble gas as a shorthand. Think of it as saying, "Okay, I’m basically like this really stable, boring element, but with a few extra bits tacked on." For Boron, the noble gas that comes before it (remember, Boron is #5) is Helium (#2). Helium’s electron configuration is 1s². This is exactly the first part of Boron’s electron configuration!
So, instead of writing 1s² 2s² 2p¹, we can say: "Okay, Boron’s first two electrons are like Helium’s electrons (1s²). Then, after that, it’s got 2s² 2p¹." We represent the Helium part by putting Helium’s symbol in brackets: [He].
Then, we just add the remaining parts of Boron’s electron configuration. So, Boron’s noble gas notation is [He] 2s² 2p¹. See? It’s like saying, "Boron is basically Helium, plus a little extra flair on the second floor." It’s way shorter, way cleaner, and lets us focus on the electrons that are actually doing stuff and are responsible for Boron’s personality (its chemical behavior, if you want to be formal).
Why is this so cool? Well, imagine you’re studying a giant molecule made of hundreds of atoms. If you had to write out the full electron configuration for every single one, your brain would melt faster than an ice cream cone on a July day in the Sahara. Noble gas notation makes it manageable. It’s the difference between having to read War and Peace to understand a single character’s motivation versus just getting a concise summary.
So, there you have it. Boron, the element that’s surprisingly useful, has a specific electron arrangement (1s² 2s² 2p¹), and we can make it even easier to understand using the noble gas notation: [He] 2s² 2p¹. It’s a little bit of atomic shorthand that saves us a whole lot of mental energy. Next time you see Boron on a label or hear about it in a science documentary, you’ll know its electrons are just like, "We’re basically Helium, but with some extra rooms we’re currently occupying. No big deal." And that, my friends, is pretty darn fascinating for something so small!
