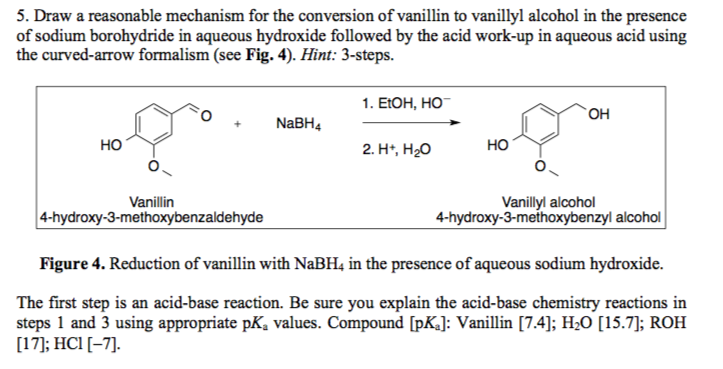
Borohydride Reduction Of Vanillin To Vanillyl Alcohol

Ever wondered how those delightful vanilla scents in perfumes or even the subtle notes in certain foods come to be? Well, today we're diving into a fun little chemistry trick that helps us get there: turning vanillin into vanillyl alcohol. It might sound fancy, but think of it like transforming a delicious spice into a slightly different, yet still wonderfully aromatic, compound. This process, called borohydride reduction, is a fantastic way to explore a bit of organic chemistry in a way that's accessible and, dare we say, enjoyable!
So, why would you want to do this? For beginners in chemistry, it's a brilliant introduction to reduction reactions – a fundamental concept. You get to see how one molecule can be gently changed into another, creating something new. For families looking for a science project, it's a safe and relatively straightforward experiment that can be quite visually rewarding as you see the transformation happen. Hobbyists, particularly those interested in fragrance creation or flavor chemistry, might find this a valuable step in understanding how different vanilla-related compounds are made, opening up new possibilities for their creations.
The core idea is taking vanillin, which is the primary component of vanilla bean extract and has that distinct vanilla smell, and adding a special ingredient, a borohydride, that essentially "adds hydrogen" to a specific part of the molecule. This changes its structure slightly, turning the aldehyde group in vanillin into a primary alcohol in vanillyl alcohol. Vanillyl alcohol still retains a pleasant, somewhat vanilla-like aroma, but it's often described as having a sweeter, more balsamic, or slightly smoky note compared to vanillin. It's also a precursor to other interesting compounds, like capsaicin, the active ingredient in chili peppers, which is a fun fact to impress your friends with!
Think of it like this: vanillin is like a ready-made cookie dough, and the borohydride reduction is like gently baking it in a special oven to get a slightly different cookie. You're not fundamentally changing the 'vanilla' nature, just refining it. You could even explore variations by using different types of borohydrides, although for a simple, everyday introduction, sodium borohydride is usually the go-to. It’s readily available and relatively safe to handle with proper precautions.
Getting started is easier than you might think. For a home experiment, you'll typically need vanillin (easily found online or in baking supply stores), sodium borohydride, a suitable solvent like ethanol or methanol (again, available from chemical suppliers or even some pharmacies), and some basic labware like a flask and stirring rod. Always remember to work in a well-ventilated area and wear safety goggles. The reaction is usually quite quick, and you can often isolate the vanillyl alcohol afterwards with simple techniques. A good tip is to start with small quantities to get a feel for the process before scaling up.
Ultimately, this little chemical journey from vanillin to vanillyl alcohol is a charming way to connect with the science behind everyday aromas and flavors. It’s a reminder that even simple-looking molecules can be transformed, opening up a world of possibilities for those curious enough to explore. It’s a satisfying and enlightening experience, adding a touch of scientific wonder to your day!
