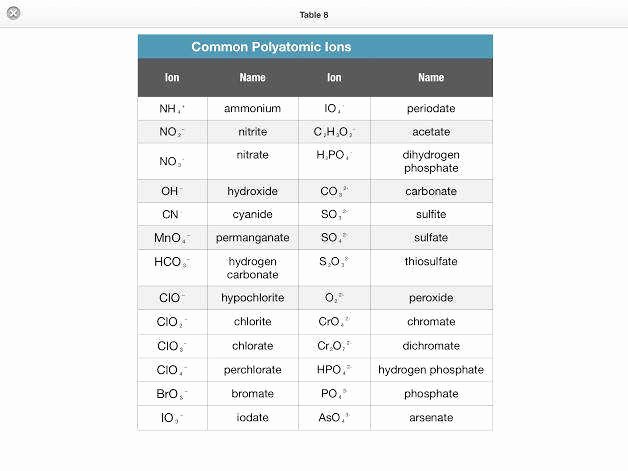
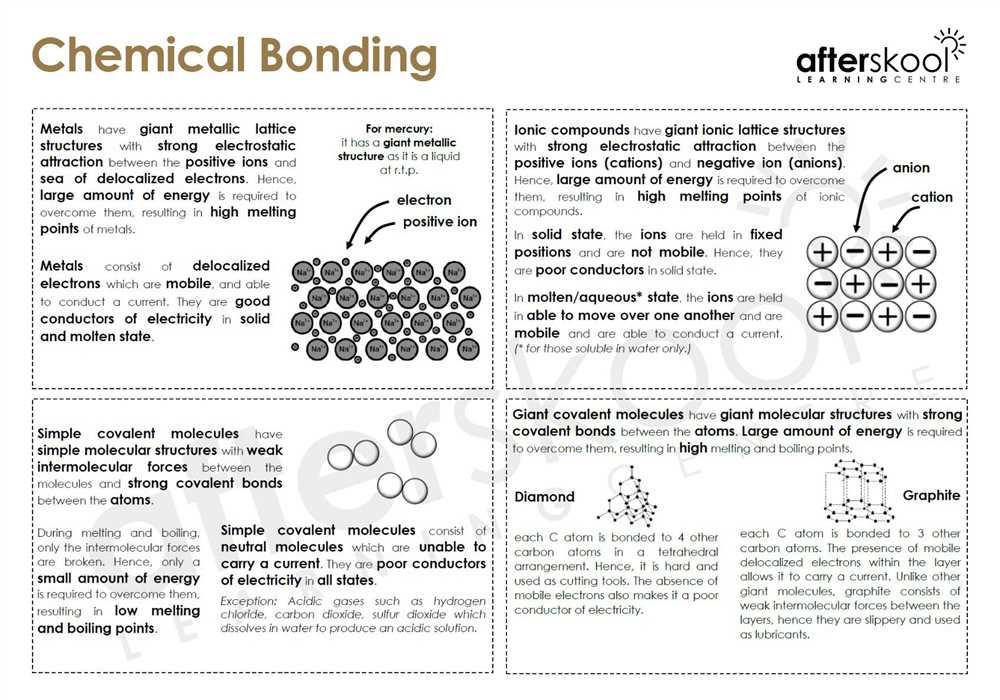
Bonding Basics Covalent Bonds Worksheet Answers

Hey there, ever feel like you're just going through the motions, doing the same things every day? Sometimes, the world of science can feel like that too, full of big words and complicated ideas. But what if I told you that some of the most fundamental building blocks of everything around us are actually tied together in ways that are surprisingly… friendly? Yep, we’re talking about covalent bonds, and no, it’s not as scary as it sounds!
Think about your favorite snack. Maybe it's a chocolate chip cookie. That cookie, from the flour to the sugar to the chocolate, is made up of tiny, tiny things called atoms. And these atoms don't just hang out randomly; they actually like to hold hands, or in science-speak, they form bonds. Covalent bonds are like a special kind of handshake between atoms, where they decide to share something really important: electrons. Electrons are these super energetic little particles that whiz around the outside of an atom. They’re like the atoms' way of saying, “Let’s team up and be stronger together!”
Imagine you and a friend both want to play with a really cool toy, but neither of you has it alone. What do you do? You decide to share, right? You both get to enjoy the toy, and because you’re playing together, it’s probably even more fun. That’s exactly what’s happening with covalent bonds! Atoms share electrons to become more stable, kind of like how you and your friend become more entertained when you share the toy.
Why Should We Even Care About These Atomic Handshakes?
You might be thinking, “Okay, atoms share electrons. So what? How does that affect my Tuesday afternoon?” Well, my friend, it affects everything! Every single thing you can see, touch, smell, or even taste is held together by these tiny, powerful connections. That comfy t-shirt you’re wearing? Covalent bonds. The water you drink? Covalent bonds. Even the air you breathe is packed with molecules held together by them!
Let’s take water, for example. H₂O. You know, the stuff that keeps us alive and makes rain puddles so fun to jump in. A water molecule is made of one oxygen atom and two hydrogen atoms. The oxygen atom needs a couple of electrons to feel complete, and each hydrogen atom needs one. So, the oxygen atom says, “Hey, little hydrogen guys, let’s share!” And they do. The oxygen shares one electron with each hydrogen, and in return, the hydrogens share their electrons with the oxygen. It’s a beautiful, life-sustaining partnership, all thanks to sharing electrons.

It’s like a perfectly balanced potluck. Everyone brings something delicious, and everyone gets to enjoy a fantastic meal. Without these covalent bonds, water wouldn’t exist as we know it, and without water, well, we wouldn’t be here to even talk about it!
So, What About Those Worksheets?
Now, you might have stumbled upon something called a “Covalent Bonds Worksheet” and felt a little pang of dread. Don’t worry! These worksheets are usually just designed to help you see how these sharing arrangements work. Think of them like little puzzles that help you understand the personalities of different atoms and how they like to team up.
When you’re looking at a worksheet and it asks you to draw or identify covalent bonds, you’re essentially being asked to show who’s sharing what. For example, you might see something like “H-Cl”. That little dash in the middle? That’s a scientist’s way of saying, “Hey, the hydrogen atom and the chlorine atom are sharing an electron pair right here!” It's like a little shorthand for their atomic friendship.
Common Scenarios and How to Ace Them (Without Sweating!)
Let's imagine a simple example. You see a molecule like methane. It’s what makes up natural gas, the stuff that heats your home. Methane has one carbon atom and four hydrogen atoms. The carbon atom is a bit of a social butterfly; it likes to share its electrons with four other atoms. So, it forms four covalent bonds, one with each hydrogen. On a worksheet, you'd see something like this:
H
|
H -- C -- H
|
H
See? The carbon (C) is in the middle, and it’s sharing electrons with each of the four hydrogens (H). Each of those dashes represents a shared pair of electrons. It’s like the carbon is the host of a party, and it’s making sure everyone has someone to dance with!
Sometimes, atoms decide to share more than just one pair of electrons. This is called a double bond or a triple bond. Think of it like going from a handshake to a high-five, and then maybe even a full-on hug! For instance, oxygen gas (O₂) that we breathe is held together by a double bond. That means the two oxygen atoms are sharing two pairs of electrons. On a worksheet, this would look like:

O = O
That double dash means they’re sharing twice as many electrons, making their bond super strong! It's like they're really, really committed to being together.
If you see a triple bond, like in nitrogen gas (N₂), it’s like an even more intense commitment. Three pairs of electrons are being shared! They’re practically inseparable.
N ≡ N
The answers on these worksheets are just about identifying these sharing arrangements. If you see a single dash, it’s a single covalent bond. Two dashes? Double covalent bond. Three dashes? Triple covalent bond. It’s like learning a secret code to understand how the universe is put together.
The Magic of Molecules
Covalent bonds are the reason we have molecules. A molecule is just a group of atoms held together by covalent bonds. So, water (H₂O) is a molecule. Methane (CH₄) is a molecule. Even that delicious cookie is made up of countless different molecules, all held together by the magic of sharing!
When you’re filling out a worksheet, and you’re asked to identify the type of bond or the number of bonds, you’re really just learning to recognize these patterns of sharing. It’s not about memorizing a bunch of random facts; it’s about understanding the fundamental way atoms interact to create the world around you. It’s the difference between just looking at a cake and understanding the recipe that made it possible.
So, the next time you see a covalent bond worksheet, don't groan! Instead, think of it as a fun opportunity to peek behind the curtain and see the amazing partnerships that hold everything together. You're not just learning science; you're learning the language of the universe, one shared electron at a time. And that, my friends, is pretty darn cool!
