Bohr Model And Lewis Dot Diagram Worksheet Answers
Hey there, science curious folks! Ever stare at a worksheet and think, "What even IS this?" Especially when it's all about atoms and their little electron buddies? Yeah, me too. But guess what? Turns out, those seemingly dry "Bohr Model and Lewis Dot Diagram Worksheet Answers" can be a blast to understand. Seriously!
Think of it like this: you're a detective, and atoms are your quirky suspects. The Bohr model is like a super-simple lineup. It’s the old-school, black-and-white mugshot of an atom. You see the nucleus in the center, all smug, with electrons orbiting it like tiny, hyperactive moons.
Niels Bohr, the dude behind this model, basically said electrons hang out in specific “energy levels.” Like little apartments in an atomic building. They can jump between floors, but they can’t just float anywhere. Pretty neat, huh?
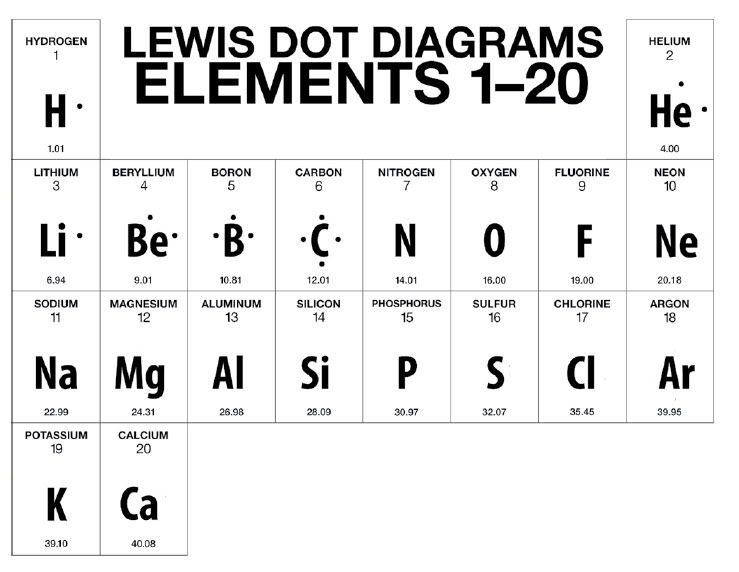
Now, the Lewis dot diagram. This is where things get really fun. Forget the whole orbiting thing. Lewis dot diagrams are all about the outermost electrons. We call these the valence electrons. They're the life of the party, the ones that get to mingle and make friends with other atoms. Think of them as the electrons wearing the loudest hats and the most sparkly shoes.
These diagrams are like super-simplified party invitations. You draw the element's symbol – that’s the name tag, like 'O' for oxygen or 'C' for carbon. Then, you sprinkle dots around it. Each dot is one valence electron. Simple, right? But oh, the stories those dots can tell!
Why are these diagrams even a thing? Because atoms love to connect. They’re social butterflies! They want to fill up their outer electron shells, like collecting enough trading cards to complete a set. Lewis diagrams show you exactly how many electrons an atom has available to do this.

And the “answers” part? That’s where you get to be the smarty-pants who figures out how these atoms are going to party. You look at the dots, you look at the element, and you predict how it will bond. It’s like figuring out who’s going to ask whom to dance at the atomic prom.
The Bohr Model: Atomic Mugshots
So, back to Mr. Bohr. His model is fantastic for visualizing basic electron shells. Imagine a planet with rings – that’s kind of what he pictured. A central sun (the nucleus) and electrons zipping around on specific paths. It’s a bit like an old-school cartoon of an atom, which is actually pretty charming.
A quick quirky fact: Bohr was actually a pretty good soccer player! Who knew our atomic guru had athletic prowess? Probably helped him visualize those speedy electrons.
The Bohr model is great for understanding how electrons gain or lose energy. If an electron absorbs energy, it jumps to a higher energy level (a fancier apartment). If it loses energy, it drops to a lower level (back to a simpler dwelling). This is how we get those beautiful atomic emission spectra – those colorful light shows atoms put on when they're excited!
Lewis Dot Diagrams: The Social Butterflies
Now, Lewis diagrams are the real stars for chemistry. They cut straight to the chase: what electrons are available for bonding? It’s like looking at a group of people and only focusing on those wearing badges that say "Available to Mingle!"
Let's say you have oxygen, 'O'. It has 6 valence electrons. So, you draw an 'O' and then eight dots around it, usually arranged in pairs. This tells you oxygen is almost happy. It wants two more electrons to have a full outer shell. It's like someone at a party who's looking for two more friends to join their circle.
Carbon, 'C', is another superstar. It has 4 valence electrons. So, you draw 'C' with four single dots. Carbon is the ultimate connector! It can make four bonds, linking up with all sorts of other atoms. It's the atom that can never find a date because it's too busy making friends with everyone else!
The beauty of Lewis diagrams is how they predict how atoms will form molecules. You pair up those lonely dots. If oxygen wants two electrons, and another oxygen atom also wants two electrons, they can share them! This is called a covalent bond. It’s like two people agreeing to share their snacks instead of hoarding them.

A funny thought: sometimes I imagine the electrons arguing over who gets to be on the "dot" side of the diagram. They're probably very competitive!
Why This Stuff is Actually Fun
Okay, I know "worksheet answers" doesn't scream "fun." But think about what you're unlocking! You're learning the secret language of the universe, the building blocks of everything!
When you get a worksheet with a Bohr model question, you’re drawing a mini solar system. When you tackle a Lewis dot diagram, you’re predicting how atoms will play house together. It's like a puzzle where the pieces are invisible atoms and the prize is understanding reality.
And the "answers"? They aren't just correct or incorrect. They represent a potential reality. A way for atoms to achieve stability and create all the cool stuff we see around us – from your phone to your favorite pizza topping.

It’s like learning a secret code. Once you know the rules of Bohr and Lewis, you can start decoding how water forms, how sugar is made, or why iron rusts. It’s pretty empowering stuff, don’t you think?
Beyond the Basics: A Sneak Peek
While the Bohr model is a great starting point, real atoms are a bit more complicated. Electrons aren't just on neat, tidy orbits. They exist in these fuzzy cloud-like regions called orbitals. But that's a story for another day, and maybe a more advanced worksheet!
Lewis diagrams, on the other hand, are surprisingly robust for predicting basic bonding. They’re your trusty sidekick for understanding simple molecules. You can even draw Lewis structures for ions, which are atoms that have gained or lost electrons and carry a charge. They're like the atoms that got a bit too excited and changed their wardrobe permanently!
So, next time you see a worksheet with "Bohr Model" or "Lewis Dot Diagram" on it, don't groan! Smile. You've got a chance to be an atomic architect, a molecular matchmaker, and a universe unraveler. And the answers? They're just the exciting conclusions to your scientific adventures. Now go forth and dot those electrons with glee!
