Based On The Data Gathered In Millikan's Oil Drop Experiments
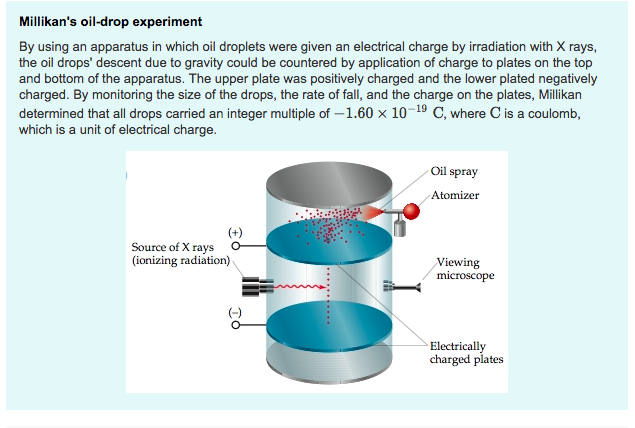
Alright, so picture this: it’s the early 1900s. Think fancy mustaches, horse-drawn carriages, and a whole lot of serious-looking scientists pondering the universe. One of these brainiacs was a chap named Robert Millikan. Now, this guy wasn't just fiddling with beakers and looking smart; he was on a mission. A mission to figure out just how tiny this one specific thing called an electron really was. And the way he went about it? Well, it involved oil, electricity, and a whole lot of patience. Honestly, if he’d asked me, I’d have said, "Dude, just grab a magnifying glass!" But no, he had to go and get all scientific.
Millikan's big idea was to trap tiny, tiny droplets of oil in mid-air. Like, so small you'd need a microscope that could see a dust mote on a gnat's eyelash. He'd spray this oil into a chamber, and these little guys would float around. But here's where it gets good: he'd zap these oil drops with X-rays. Now, X-rays are like the universe's way of saying, "Hey, let's mess with things!" And what they did here was knock electrons off the oil drops. So, suddenly, these otherwise innocent oil droplets were walking around with a negative charge, like they'd just stuck their finger in a light socket.
Then, the real magic happened. Millikan had two metal plates, one above the other. He'd put his charged oil drops in between them. And here’s the kicker: he’d apply an electric voltage. Now, if you’ve ever played with magnets, you know opposite poles attract and like poles repel, right? Well, electricity is a bit like that. He had his negatively charged oil drops. If he zapped the top plate with a positive charge and the bottom plate with a negative charge, the oil drops, being negative, would get pushed UP, away from the bottom plate. It was like giving them tiny little electric trampolines!
But gravity, that stubborn old buzzkill, was still trying to pull the oil drops DOWN. So, Millikan was basically in a cosmic tug-of-war. He could adjust the electric voltage, trying to find that perfect sweet spot where the upward electrical push exactly balanced the downward pull of gravity. When he found that spot, the oil drop would just… hang there. Suspended. Like a tiny, electrified disco ball frozen in time. Imagine doing that with a blob of peanut butter! You’d be there all day.
The really mind-blowing part, though, is what he did next. He painstakingly measured the size of each oil drop, how fast it was falling when the electricity was off, and how much voltage he needed to keep it perfectly still. By doing all this fancy math (which, let’s be honest, most of us would rather go toe-to-toe with a grumpy badger than do), he could figure out the total charge on each individual oil drop. And here’s the kicker: he found that every single oil drop had a charge that was a multiple of a specific, tiny number. Like, some had one unit of this charge, some had two, some had three, and so on. But they never had half a unit, or a quarter of a unit. It was always a whole number.

This was HUGE! It was like discovering that all the LEGO bricks in the world were made of the same basic, tiny, indivisible piece. Millikan’s data showed that the electron, that tiny particle that carries a negative electric charge, was the fundamental unit of electric charge. You couldn’t get a smaller bit of negative charge. It was the universe's smallest electric coin. You can't pay for a candy bar with half a cent, right? Same idea. This was the smallest possible amount of electric charge you could have.
So, basically, this oil drop experiment, which sounds like it could have been a scene from a mad scientist's movie, actually helped us understand that electricity isn't some kind of continuous fluid. It's made of discrete little packets, and the electron is the smallest one. It's like realizing that water isn't just water, but it's made of tiny little H2O molecules. Millikan’s experiment did that for electric charge. Pretty neat, huh?
And get this, the value he got for the charge of an electron? It was astonishingly close to the value we know today. He was off by less than 1%. Think about trying to measure something so tiny that you can't even see it, using methods that would seem primitive today, and being that accurate. It’s like hitting a bullseye on a dartboard from the next continent. He was basically the Usain Bolt of early 20th-century physics – fast, precise, and setting world records.
Millikan’s work didn't just stop there. It laid the groundwork for a whole bunch of other discoveries in the world of quantum mechanics. It showed that the universe, at its most fundamental level, is a bit of a quirky, quantized place. Everything comes in these little packets. So, the next time you flick a light switch or charge your phone, you can thank Robert Millikan and his super-tiny, electrically charged oil drops for helping us understand the fundamental building blocks of the electrical world. He took a gamble, sprayed some oil, played with some electricity, and ended up proving that the electron is one of the universe's most precious, and indivisible, little packages. Cheers to that!
