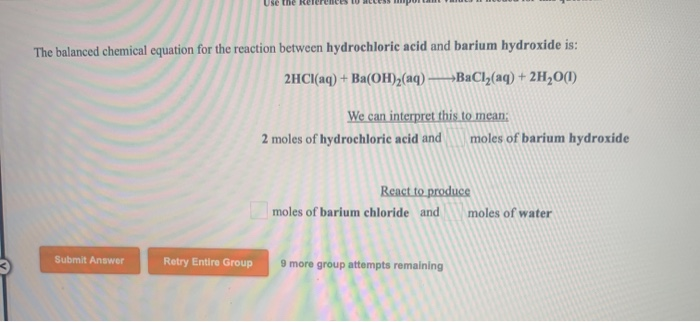
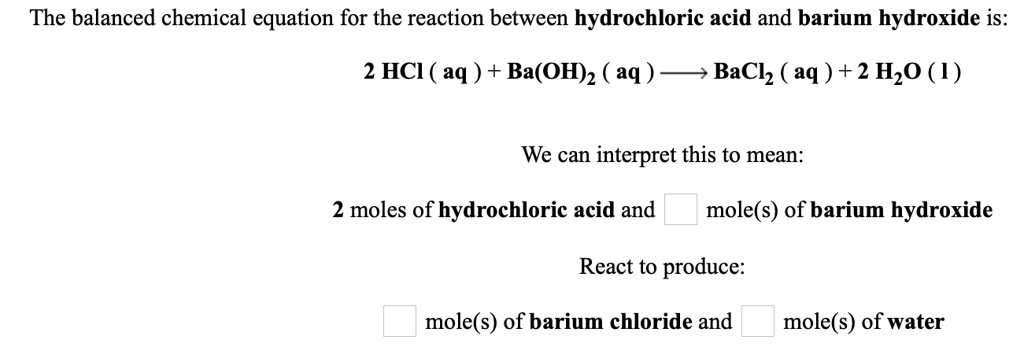
Barium Hydroxide + Hydrochloric Acid Balanced Equation
Imagine you're in your kitchen, whipping up a batch of your famous cookies. You've got your flour, sugar, and eggs all ready to go. But what if, instead of reaching for baking soda, you decided to grab something a little more… exciting? Today, we're diving into the wonderful world of chemistry, where even seemingly ordinary ingredients can have a surprisingly delightful reaction. We're talking about a chemical get-together between Barium Hydroxide and Hydrochloric Acid. Sounds fancy, right? But stick with me, because this is where the magic happens!
So, picture this: Barium Hydroxide is like a very polite, slightly shy host at a party. It's a bit powdery, a little chalky, and generally keeps to itself. It's often found doing its own thing, perhaps in some industrial processes or even in certain types of skincare (don't worry, we're not using that on your face today!). Think of it as the quiet guest who's happy to be invited but isn't the life of the party on its own.
Now, Hydrochloric Acid is a different kind of character. This one’s more like the energetic, enthusiastic guest who bursts through the door with a grin and a story. It’s a strong acid, and in its pure form, it’s not something you’d want to play with. But in the controlled environment of a lab (or our imaginary kitchen!), it’s ready for some action. It’s the kind of ingredient that knows how to get things moving, and it’s not afraid to mingle.
When Worlds Collide: The Grand Unveiling!
When Barium Hydroxide and Hydrochloric Acid finally meet, it’s like a scene straight out of a romantic comedy. They’re introduced, a little hesitant at first, and then… bam! Sparks fly. But instead of dramatic pronouncements of love, these two chemical buddies decide to get really cozy and transform into something entirely new. It's a beautiful dance of atoms and molecules, and the result is a surprisingly neat and tidy outcome.
What do they become? Well, the polite Barium Hydroxide and the energetic Hydrochloric Acid decide to team up and create two new entities. First, they form Barium Chloride. Think of this as the solid, dependable couple that forms from their union. It’s a salt, and it’s quite stable. It’s like they've found their perfect match, settling down and creating something new and lasting.
But the story doesn’t end there! As they’re busy forming Barium Chloride, they also release something else: Water. Yes, good old H₂O! It's like their successful collaboration is so joyous that it overflows, creating this essential and life-giving substance. It’s a heartwarming reminder that sometimes, when different elements come together, they not only create something interesting but also contribute something vital to the world.
It’s a bit like two puzzle pieces that, when fitted together, not only complete a picture but also magically generate a little refreshing drink for everyone to enjoy!
Solved Use the The balanced chemical equation for the | Chegg.com
The Secret Recipe: A Glimpse Behind the Curtain
Now, you might be wondering, how do we know exactly how much of each ingredient to use to get this perfect outcome? This is where the magic of a balanced chemical equation comes in. It’s like having a secret recipe passed down through generations of chemists. It tells us precisely how many molecules of Barium Hydroxide need to dance with how many molecules of Hydrochloric Acid to produce the right amount of Barium Chloride and Water.
For this particular party, the recipe is quite specific. We need one molecule of Barium Hydroxide (let's call it Ba(OH)₂) and two molecules of Hydrochloric Acid (that's 2 HCl). Why two? Well, Barium Hydroxide is a bit like a parent with two helping hands, and Hydrochloric Acid has one hand that’s ready to pair up. So, to make everything work smoothly and ensure no one is left out, we need two of the single-handed Hydrochloric Acid molecules to join up with the two helping hands of Barium Hydroxide.
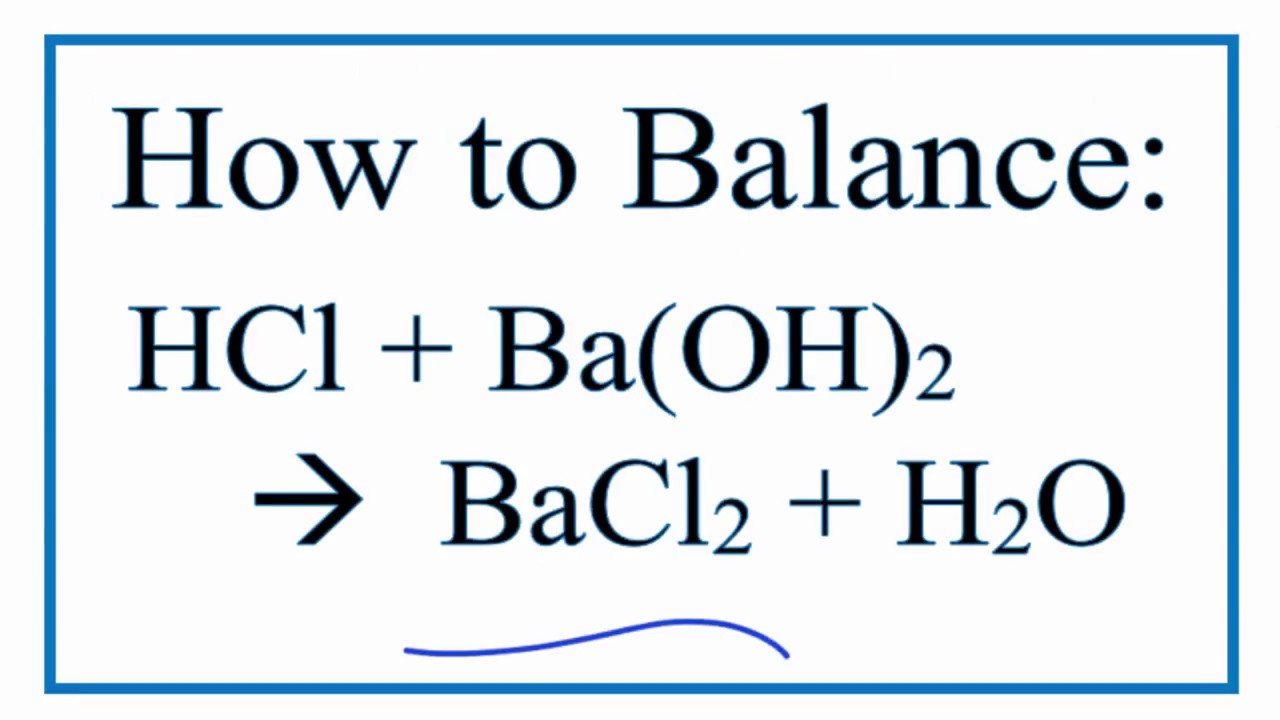
And what’s the reward for this perfect pairing? Two molecules of Barium Chloride (BaCl₂) and two molecules of Water (2 H₂O) gracefully emerge. It’s a beautiful ballet of atoms, perfectly choreographed. The Barium atom, which started out with its Hydroxide friends, finds a new partner in Chlorine. And the Hydrogen atoms, initially attached to the Chlorine, now find themselves happily paired up with Oxygen to form water.
It's a testament to the elegant order that exists in the universe, even at the tiniest levels. So, the next time you hear about Barium Hydroxide and Hydrochloric Acid, don't just think of chemicals. Think of a lively party, a perfect recipe, and the creation of something new and essential, all thanks to a little chemical matchmaking!