Barium Hydroxide And Perchloric Acid Balanced Equation
Hello, fellow explorers of the fascinating world around us! Have you ever found yourself staring at a bubbling beaker or a mysterious powder and thinking, "What's going on here?" Well, today we're diving into a chemical reaction that might sound a bit intimidating at first, but trust me, it’s a peek into the incredible precision that makes so much of our modern life possible. We're talking about the balanced equation of barium hydroxide and perchloric acid. Now, before your eyes glaze over, think of this as the secret handshake of two chemical personalities, ensuring they interact perfectly!
Why would anyone care about balancing chemical equations like this? It's all about predictability and control. In everyday life, this principle of balanced reactions is absolutely everywhere, even if we don't see the equations. From the fuels that power our cars to the medications that keep us healthy, chemists and engineers rely on these balanced equations to ensure that the right amounts of ingredients combine to produce exactly what's intended, with no dangerous byproducts. It's the scientific equivalent of following a recipe perfectly – too much of one ingredient, and your cake might be a disaster; too little, and it won't rise!
So, what does barium hydroxide and perchloric acid have to do with us? While you're unlikely to be mixing these two in your kitchen (and please, don't try!), the principles behind their reaction are fundamental. Perchloric acid, in its various forms, is used in rocket propellants and in the manufacture of certain explosives. Barium compounds, on the other hand, have a range of uses, from medical imaging (like barium sulfate for X-rays) to the creation of fireworks. Understanding how these substances react when balanced ensures that these powerful applications are safe and effective.
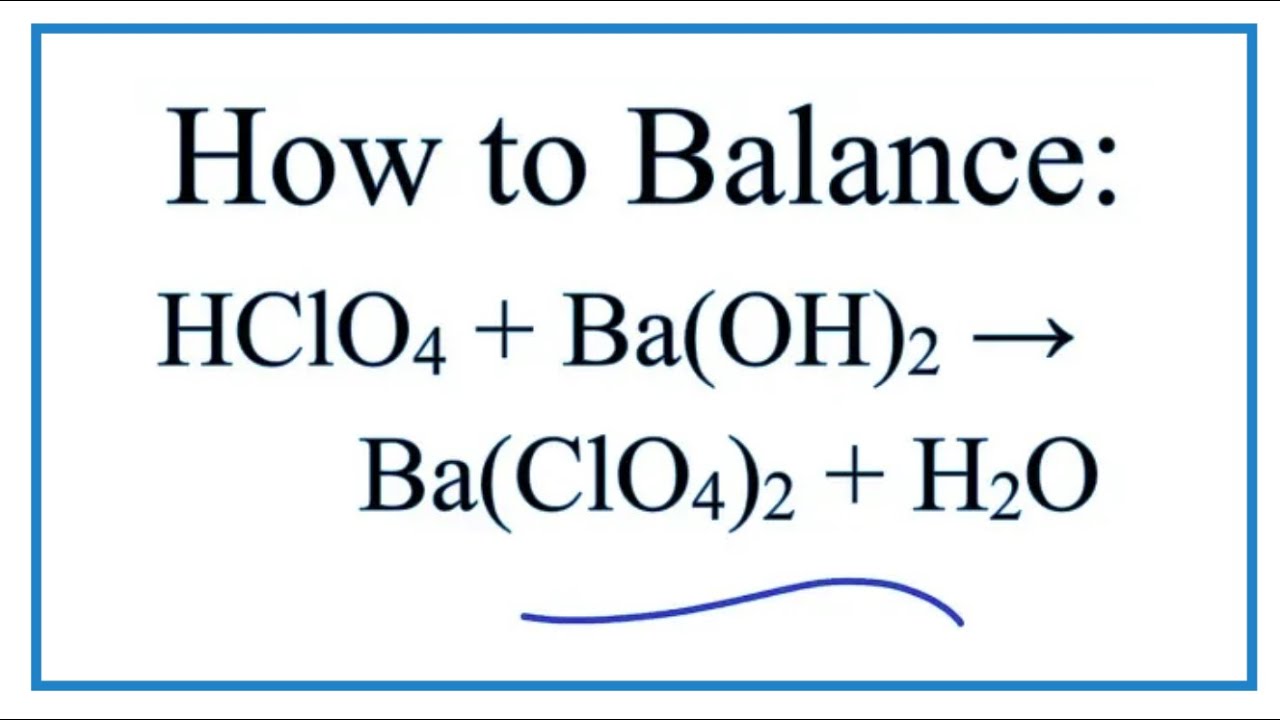
The balanced equation for this reaction is:
Ba(OH)₂ + 2HClO₄ → Ba(ClO₄)₂ + 2H₂O
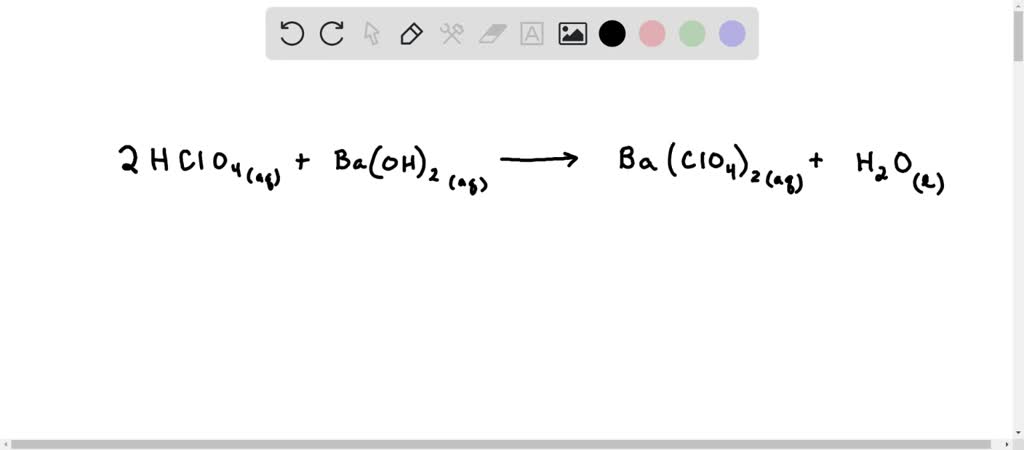
This tells us that one molecule of barium hydroxide reacts with two molecules of perchloric acid to produce one molecule of barium perchlorate and two molecules of water. See how the numbers are crucial? It’s about making sure every atom finds its perfect dance partner. This meticulous balancing act is what allows for the controlled release of energy in rockets or the precise formulation of pharmaceuticals. It's a testament to the elegance and power of chemistry.
Now, how can you "enjoy" this knowledge more effectively? First, don't be afraid of the symbols! Think of them as a shorthand language for understanding the universe. If you're curious, a great way to engage is through online simulations or educational videos that visually demonstrate chemical reactions. You can find resources that animate these precise molecular interactions, making them much easier to grasp. Another tip is to relate it back to familiar concepts. Think about how baking soda and vinegar react – that fizzing is a chemical reaction! While the scale and complexity differ, the underlying principle of balancing reactants to achieve a desired outcome is the same. So, the next time you see something react, whether it’s a spectacular firework display or even just your morning coffee brewing, remember the silent, precise work of balanced chemical equations like our friends, barium hydroxide and perchloric acid. It's a reminder that science is all around us, making our world work!
