Barium Hydroxide And Hydrochloric Acid Balanced Equation
Ever wondered what happens when you mix a couple of common household ingredients and things get a little bubbly? It might sound like kitchen science, but understanding chemical reactions like the one between barium hydroxide and hydrochloric acid can be surprisingly fascinating and even a little bit useful. Think of it as a tiny, controlled explosion of knowledge that’s easy to grasp and can spark curiosity in all sorts of everyday discoveries.
So, why dive into the world of balanced chemical equations? For beginners, it’s like learning the secret handshake of chemistry. It shows you how ingredients combine in precise amounts to create something new. Imagine baking a cake – you need just the right amount of flour and sugar to get it right! Chemical equations are similar, just a bit more… scientific. For families looking for fun and educational activities, this is a great starting point. It’s a chance to explore together, ask "what if?" questions, and maybe even demystify some of the "magic" that happens in science labs. Even for hobbyists, like those interested in DIY projects or understanding how things work, grasping these fundamental concepts can open up a new understanding of the materials they're working with.
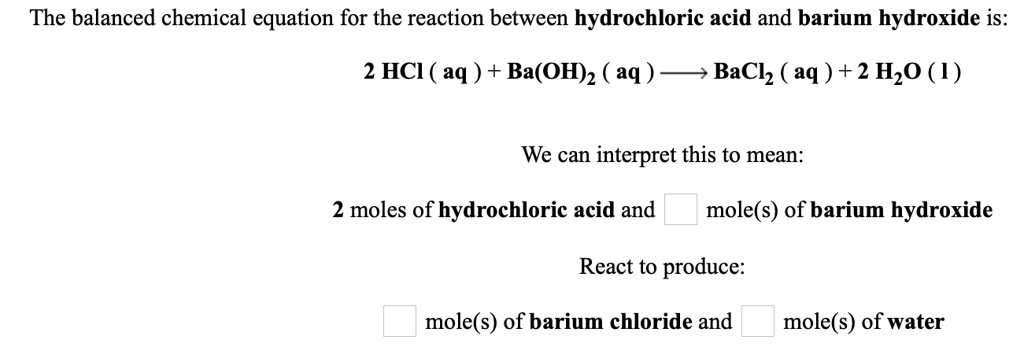
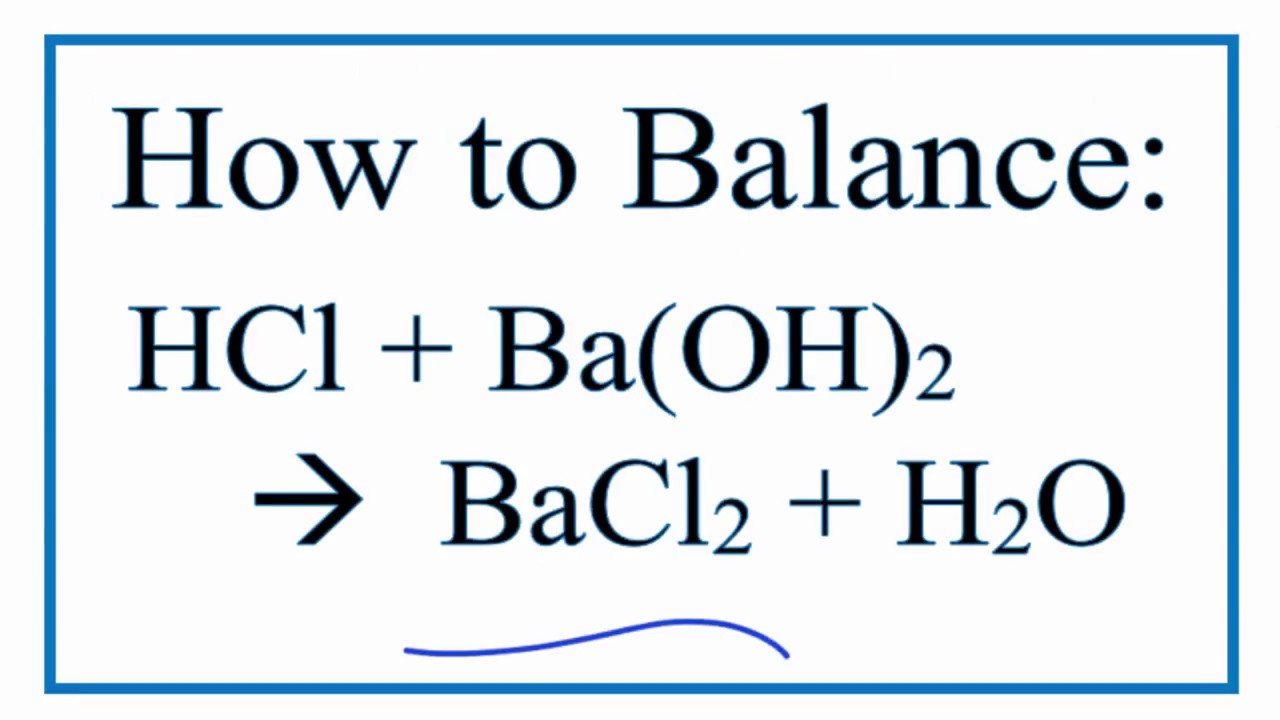
Let's talk about the stars of our show: barium hydroxide (Ba(OH)₂) and hydrochloric acid (HCl). When these two meet, they have a little dance, forming barium chloride (BaCl₂), water (H₂O), and often releasing a bit of heat. The balanced equation for this is: Ba(OH)₂ + 2HCl → BaCl₂ + 2H₂O. See that '2' in front of HCl and H₂O? That's the balancing act! It means for every one molecule of barium hydroxide, you need two molecules of hydrochloric acid to make sure everything is perfectly accounted for. It's like making sure you have enough chairs for everyone at a party.
You might encounter variations of this reaction in different contexts. For instance, if you were to use a different acid, like sulfuric acid (H₂SO₄), you'd get a different set of products and a different balanced equation. Or, if you're looking at neutralization reactions in general, the principle of balancing atoms remains the same, whether it's with strong acids and bases or even weaker ones. This fundamental understanding is key to so many areas of science.
Getting started with understanding these equations is simpler than you might think. You don't need a fancy lab coat! Start by looking up the chemical formulas for common substances. Then, try writing out the reactants (what you start with) and products (what you end up with) for a simple reaction you find interesting. Resources like online chemistry dictionaries or educational websites can be your best friends here. The key is to take it one step at a time and not get overwhelmed. Focus on understanding the symbols and the meaning of those little numbers. It’s about building a foundation of knowledge that you can expand on.
So, there you have it! A peek into the world of balanced chemical equations, using barium hydroxide and hydrochloric acid as our guide. It’s a small glimpse into the organized beauty of chemistry, showing how even simple reactions follow predictable rules. Embracing this understanding can be a really rewarding and enjoyable journey, opening up a new way to see the world around you.
