Balancing Chemical Equations Science Geek Answers

Ever feel like you're juggling a million things at once? Work deadlines, dinner plans, that nagging feeling you forgot to water the plants? Well, imagine a universe where even the tiniest little particles – the building blocks of everything – have to do the same thing! We're talking about balancing chemical equations, and trust me, it's not as scary as it sounds. Think of it as a cosmic game of "who's got what?" to make sure everything is fair and square.
So, what exactly are we balancing here? Picture this: you're baking cookies. You need flour, sugar, eggs, and chocolate chips. If you decide to double your cookie recipe, you can't just magically get twice the ingredients. Nope! You've gotta grab twice the flour, twice the sugar, twice the eggs, and yes, twice the glorious chocolate chips. The universe, in its infinite wisdom, is kind of the same way. When atoms decide to team up and make something new (that's a chemical reaction, by the way!), they can't just disappear or spontaneously generate. They have to stick around!
Let's say we have a super simple reaction where hydrogen (H) and oxygen (O) decide to get together and make water (H₂O). On one side of the equation, we might have two hydrogen atoms floating around, ready to party. But on the other side, where water is formed, we need two hydrogen atoms for every one oxygen atom. Uh oh! We have an oxygen atom chilling by itself, feeling a bit left out. This is where our trusty balancing act comes in.
We can't just chop an oxygen atom in half – that would be downright rude and, frankly, impossible! Instead, we have to make sure we have enough of everything to go around. Think of it like this: if you have a group of kids who want to build with LEGOs, and they need two red bricks for every blue brick to build a cool spaceship, they can't just grab one red brick if they only have one blue brick. They need to make sure the ratio is perfect!
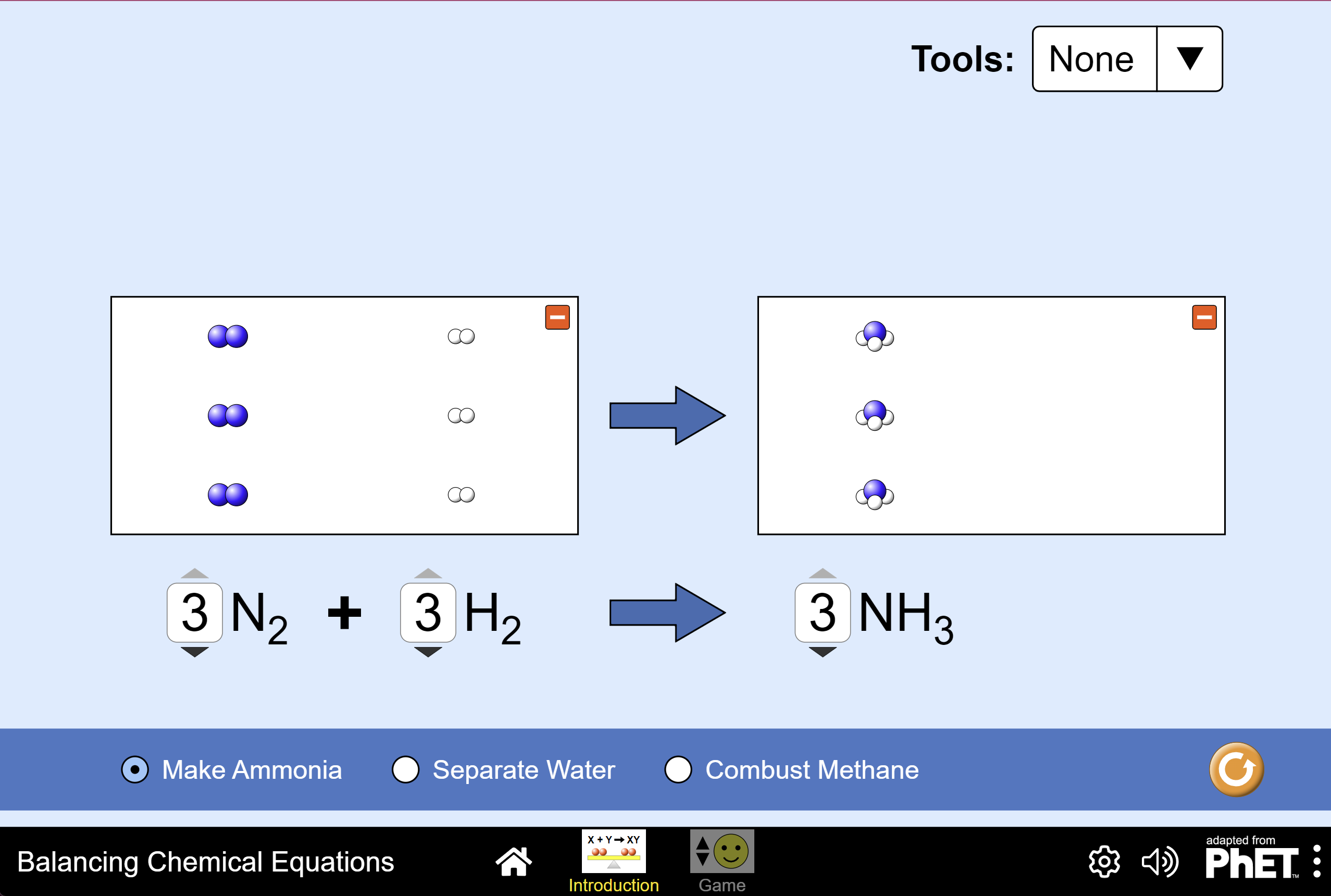
So, back to our water example. If we start with two hydrogen atoms (H₂) and one oxygen molecule (O₂), we're going to have an extra oxygen atom floating around after the reaction. To fix this, we can bring in another molecule of hydrogen (H₂). Now we have four hydrogen atoms. And to make sure we get two water molecules (H₂O), we need two oxygen atoms. Voila! We've balanced the equation. We now have 2 H₂ + O₂ → 2 H₂O. See? Four hydrogen atoms on the left, four on the right. Two oxygen atoms on the left, two on the right. Everyone's happy, everyone's accounted for. It's like a perfectly orchestrated symphony of atoms!
It's all about making sure the number of each type of atom going into the reaction is exactly the same as the number of each type of atom coming out. This fundamental principle is called the Law of Conservation of Mass. It's one of those big, fancy science ideas that basically says, "You can't make something from nothing, and you can't destroy something and have it vanish into thin air." It’s the universe’s ultimate "no funny business" rule!
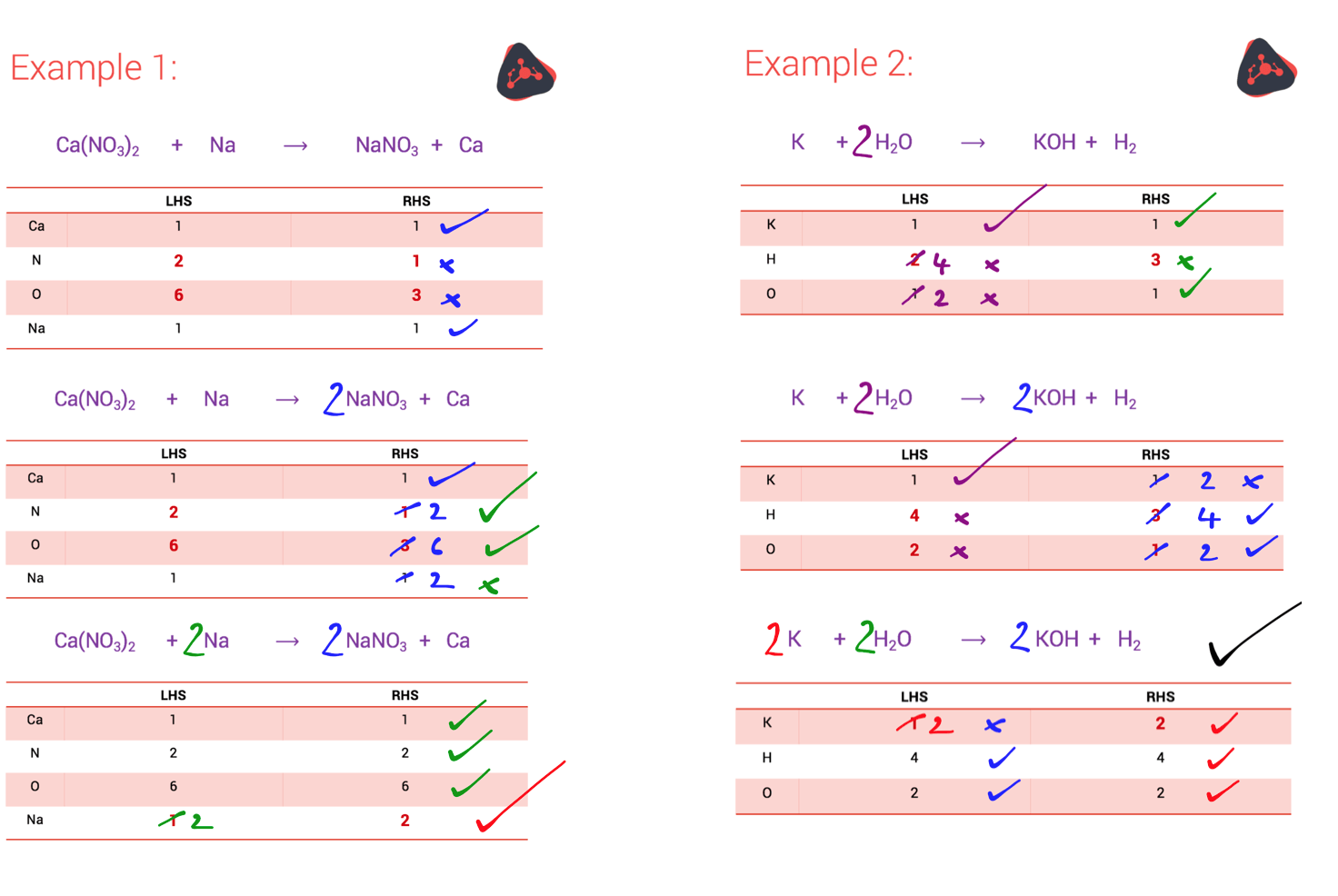
Sometimes, balancing can feel like a puzzle. You might need to add a few more molecules here, a couple more there. It's like trying to fit all your winter clothes into your suitcase for a tropical vacation – a little bit of strategic stuffing and rearranging is required! You'll get a feel for it, you'll start spotting patterns, and before you know it, you'll be a chemical equation balancing ninja.
"Imagine you're a chef, and you're making goulash. You can't just throw in a handful of paprika and hope for the best. You need the right amount of beef, the right amount of onions, the right amount of paprika – everything in perfect proportion to get that delicious, comforting flavor. Chemical equations are the recipes of the universe, and balancing them is like making sure you've got every ingredient measured out just right!"
Don't worry if it feels a bit fiddly at first. Even the most brilliant scientists probably stared at a few unbalanced equations with a furrowed brow in their day. The key is to be patient, to count carefully, and to remember that you're not creating or destroying anything, just rearranging it! You're playing a vital role in understanding how the world around you works, from the air you breathe to the food you eat. You're basically a cosmic accountant, making sure the atomic books always balance. And that, my friends, is pretty darn cool. So next time you see a chemical equation, don't run for the hills! Give it a friendly wave, maybe a little wink, and get ready to make those atoms do a happy dance of perfect proportion. You've got this!
